Journal of Molecular and Cellular Cardiology ( IF 5 ) Pub Date : 2023-10-02 , DOI: 10.1016/j.yjmcc.2023.09.010 Felix Wiedmann 1 , Amelie Paasche 1 , Jendrik Nietfeld 2 , Manuel Kraft 1 , Anna L Meyer 3 , Gregor Warnecke 3 , Matthias Karck 3 , Norbert Frey 1 , Constanze Schmidt 1
|
Rationale
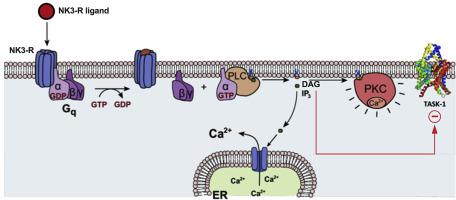
The neurokinin-III receptor was recently shown to regulate atrial cardiomyocyte excitability by inhibiting atrial background potassium currents. TASK-1 (hK2P3.1) two-pore-domain potassium channels, which are expressed atrial-specifically in the human heart, contribute significantly to atrial background potassium currents. As TASK-1 channels are regulated by a variety of intracellular signalling cascades, they represent a promising candidate for mediating the electrophysiological effects of the Gq-coupled neurokinin-III receptor.
Objective
To investigate whether TASK-1 channels mediate the neurokinin-III receptor activation induced effects on atrial electrophysiology.
Methods and results
In Xenopus laevis oocytes, heterologously expressing neurokinin-III receptor and TASK-1, administration of the endogenous neurokinin-III receptor ligands substance P or neurokinin B resulted in a strong TASK-1 current inhibition. This could be reproduced by application of the high affinity neurokinin-III receptor agonist senktide. Moreover, preincubation with the neurokinin-III receptor antagonist osanetant blunted the effect of senktide. Mutagenesis studies employing TASK-1 channel constructs which lack either protein kinase C (PKC) phosphorylation sites or the domain which is regulating the diacyl glycerol (DAG) sensitivity domain of TASK-1 revealed a protein kinase C independent mechanism of TASK-1 current inhibition: upon neurokinin-III receptor activation TASK-1 channels are blocked in a DAG-dependent fashion. Finally, effects of senktide on atrial TASK-1 currents could be reproduced in patch-clamp measurements, performed on isolated human atrial cardiomyocytes.
Conclusions
Heterologously expressed human TASK-1 channels are inhibited by neurokinin-III receptor activation in a DAG dependent fashion. Patch-clamp measurements, performed on human atrial cardiomyocytes suggest that the atrial-specific effects of neurokinin-III receptor activation on cardiac excitability are predominantly mediated via TASK-1 currents.
中文翻译:

神经激肽 III 受体的激活调节人心房 TASK-1 电流
基本原理
最近发现神经激肽-III 受体可以通过抑制心房背景钾电流来调节心房心肌细胞的兴奋性。TASK-1 (hK 2P 3.1) 双孔域钾通道在人类心脏中心房特异性表达,对心房背景钾电流有显着贡献。由于 TASK-1 通道受到多种细胞内信号级联的调节,因此它们是介导 Gq 偶联神经激肽 III 受体电生理效应的有希望的候选者。
客观的
探讨 TASK-1 通道是否介导神经激肽 III 受体激活对心房电生理的影响。
方法和结果
在异源表达神经激肽 III 受体和 TASK-1 的非洲爪蟾卵母细胞中,给予内源性神经激肽 III 受体配体 P 物质或神经激肽 B会导致强烈的 TASK-1 电流抑制。这可以通过应用高亲和力神经激肽-III 受体激动剂senktide来重现。此外,与神经激肽-III受体拮抗剂奥萨尼坦预孵育会减弱senktide的作用。采用缺乏蛋白激酶 C (PKC) 磷酸化位点或调节 TASK-1 二酰基甘油 (DAG) 敏感结构域的 TASK-1 通道构建体的诱变研究揭示了 TASK-1 电流抑制的独立于蛋白激酶 C 的机制:神经激肽 III 受体激活后,TASK-1 通道会以 DAG 依赖性方式被阻断。最后,senktide 对心房 TASK-1 电流的影响可以在膜片钳测量中重现,对分离的人心房心肌细胞进行测量。
结论
异源表达的人类 TASK-1 通道被神经激肽 III 受体激活以 DAG 依赖性方式抑制。对人心房心肌细胞进行的膜片钳测量表明,神经激肽 III 受体激活对心脏兴奋性的心房特异性影响主要是通过 TASK-1 电流介导的。



























 京公网安备 11010802027423号
京公网安备 11010802027423号