Hellenic Journal of Cardiology ( IF 4.1 ) Pub Date : 2023-10-05 , DOI: 10.1016/j.hjc.2023.09.019 Dimitrios Bampatsias 1 , Foteini Theodorakakou 1 , Alexandros Briasoulis 1 , Georgios Georgiopoulos 1 , Anna Dimoula 1 , Vasileios Papantoniou 2 , Ioannis Papantoniou 2 , Chaido Skiadaresi 2 , Pipitsa Valsamaki 3 , Evangelos Repasos 1 , Ioannis Petropoulos 1 , Dimitrios Delialis 1 , Alexandra Papathoma 4 , Georgios Koutsis 5 , Maria-Eirini Tselegkidi 1 , Kimon Stamatelopoulos 1 , Efstathios Kastritis 1
|
Background
Clinical characteristics and outcomes of patients with transthyretin amyloidosis cardiomyopathy (ATTR-CM) vary by region, necessitating the acquisition of country-specific evidence for proper management.
Methods
This is an observational study including sequential patients presenting in the Amyloidosis Reference Center of Greece, from 01/2014 to 12/2022. ATTR-CM was diagnosed by positive scintigraphy and exclusion of light-chain amyloidosis or positive biopsy typing. Genetic testing was performed in all cases.
Results
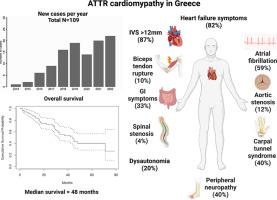
One-hundred and nine ATTR-CM patients were included (median age, 81 years) of which 15 carried TTR mutations (27% Val30Met). Most patients (82%) presented with heart failure and 59% with atrial fibrillation, while 10% had aortic stenosis. Importantly, 78 (71.6%) had clinically significant extracardiac manifestations (45% musculoskeletal disorder, 40% peripheral neuropathy and 33% gastrointestinal symptoms). Sixty-five (60%) received disease-specific treatment with tafamidis. Estimated median survival was 48 months; advanced NYHA class, National Amyloidosis Center stage, eGFR<45 ml/kg/1.73m2, NT-pro-BNP>5000 pg/mL were associated with worse survival, while tafamidis treatment was associated with improved survival in patients with IVS≥ 12 mm.
Discussion
These are the first data describing the characteristics, management, and outcomes of patients with ATTR-CM in Greece, which could influence local guidelines.
Short title
Transthyretin cardiomyopathy in Greece
中文翻译:

希腊转甲状腺素蛋白淀粉样变性心肌病:国家转诊中心的临床见解
背景
转甲状腺素蛋白淀粉样变性心肌病 (ATTR-CM) 患者的临床特征和结果因地区而异,因此需要获取特定国家的证据以进行适当的治疗。
方法
这是一项观察性研究,包括 2014 年 1 月至 2022 年 12 月在希腊淀粉样变性参考中心就诊的连续患者。ATTR-CM 通过阳性闪烁扫描并排除轻链淀粉样变性或阳性活检分型来诊断。所有病例均进行了基因检测。
结果
纳入 109 名 ATTR-CM 患者(中位年龄 81 岁),其中 15 名携带TTR突变(27% Val30Met)。大多数患者 (82%) 出现心力衰竭,59% 出现房颤,10% 出现主动脉瓣狭窄。重要的是,78 例(71.6%)有临床上显着的心外表现(45% 为肌肉骨骼疾病,40% 为周围神经病变,33% 为胃肠道症状)。六十五人 (60%) 接受了 Tafamidis 的疾病特异性治疗。预计中位生存期为 48 个月;NYHA 分级晚期、国家淀粉样变性中心分期、eGFR<45 ml/kg/1.73m 2、NT-pro-BNP>5000 pg/mL 与 IVS ≥ 12 患者的生存率较差相关,而 tafamidis 治疗与生存率改善相关毫米。
讨论
这些是描述希腊 ATTR-CM 患者特征、治疗和结果的第一批数据,可能会影响当地指南。
短标题
希腊转甲状腺素蛋白心肌病



























 京公网安备 11010802027423号
京公网安备 11010802027423号