当前位置:
X-MOL 学术
›
Polym. Chem.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
Enzyme-polymer conjugates with photocleavable linkers for control over protein activity
Polymer Chemistry ( IF 4.6 ) Pub Date : 2024-02-13 , DOI: 10.1039/d3py01339a Mikayla F. Tan 1 , Brock M. Hosier 1 , Neil L. Forsythe 1 , Heather D. Maynard 1
Polymer Chemistry ( IF 4.6 ) Pub Date : 2024-02-13 , DOI: 10.1039/d3py01339a Mikayla F. Tan 1 , Brock M. Hosier 1 , Neil L. Forsythe 1 , Heather D. Maynard 1
Affiliation
|
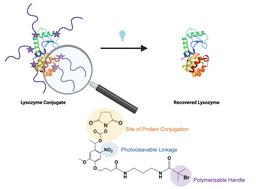
Reversible conjugation of polymers to proteins is important for a variety of applications, for example to control protein activity. Light is often employed as an external trigger to allow for spatio and temporal control over release of a payload. In this report, we demonstrate preparation of photocleavable poly(polyethylene glycol) acrylate-lysozyme (pPEGA-Lys) conjugates via ortho-nitrobenzyl linkages. The conjugates were made by both grafting-to and grafting-from in order to compare and contrast the two synthetic approaches. First, a lysine-reactive ortho-nitrobenzyl atom transfer radical polymerization (ATRP) initiator was synthesized. For the grafting-to strategy, the initiator was employed in the ATRP of PEGA, and the subsequent polymer was conjugated to the lysine residues of lysozyme. For the grafting-from strategy, lysozyme was modified first with the photocleavable initiator, and the purified macroinitiator was then subjected to polymerization conditions to synthesize the protein-polymer conjugate. The polymer was cleaved from the protein via UV light, and activity before and after polymer removal was evaluated, showing 83% recovery. This work provides evidence that reversing conjugation is successful for activity modulation for ortho-nitrobenzyl linked protein-polymer conjugates.
更新日期:2024-02-13



























 京公网安备 11010802027423号
京公网安备 11010802027423号