Beilstein Journal of Organic Chemistry ( IF 2.7 ) Pub Date : 2024-02-19 , DOI: 10.3762/bjoc.20.32 Sharmila Neupane , Marcelo Rodrigues de Amorim , Elizabeth Skellam
Abstract
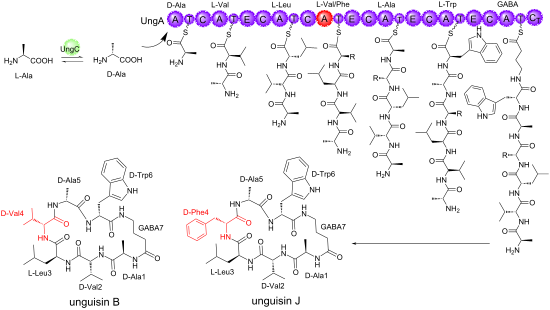
Several under-explored Aspergillus sp. produce intriguing heptapeptides containing a γ-aminobutyric acid (GABA) residue with as yet unknown biological functions. In this study, a new GABA-containing heptapeptide – unguisin J (1) – along with known unguisin B (2) were isolated from a solid culture of Aspergillus heteromorphus CBS 117.55. The structure of compound 1 was elucidated by extensive 1D and 2D NMR spectroscopic analysis including HSQC, HMBC, COSY, and 2D NOESY as well as HRESIMS. The stereochemistry of 1 and 2 was determined by Marfey’s method. A biosynthetic gene cluster (BGC) encoding unguisins B and J was compared to characterized BGCs in other Aspergillus sp. Since the unguisin family of heptapetides incorporate different amino acid residues at different positions of the peptide, the A and C domains of the UngA NRPS were analyzed in an attempt to understand the lack of substrate specificity observed.
Beilstein J. Org. Chem. 2024, 20, 321–330. doi:10.3762/bjoc.20.32



























 京公网安备 11010802027423号
京公网安备 11010802027423号