当前位置:
X-MOL 学术
›
Org. Lett.
›
论文详情
Our official English website, www.x-mol.net, welcomes your feedback! (Note: you will need to create a separate account there.)
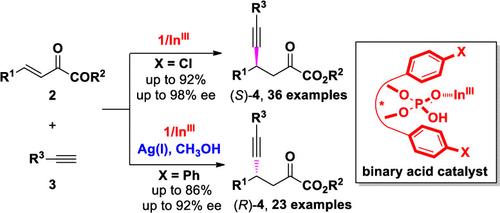
Switchable Enantioselectivity in Conjugate Alkyne Addition of β,γ-Unsaturated α-Keto Esters by Asymmetric Binary Acid Catalysis
Organic Letters ( IF 5.2 ) Pub Date : 2024-04-24 , DOI: 10.1021/acs.orglett.4c01101 Depeng Duan 1 , Mengdie Tang 1 , Man Wang 1 , Huixin Qiu 1 , Ran Song 1 , Daoshan Yang 1 , Jian Lv 1
Organic Letters ( IF 5.2 ) Pub Date : 2024-04-24 , DOI: 10.1021/acs.orglett.4c01101 Depeng Duan 1 , Mengdie Tang 1 , Man Wang 1 , Huixin Qiu 1 , Ran Song 1 , Daoshan Yang 1 , Jian Lv 1
Affiliation
|
Switchable enantioselectivity was uncovered in the enantioselective catalytic conjugate addition of β,γ-unsaturated α-keto esters with terminal alkynes to the chiral Lewis acid complex of In(BF4)3 and chiral phosphoric acid.
中文翻译:

不对称二元酸催化 β,γ-不饱和 α-酮酯共轭炔加成中的可切换对映选择性
在将带有末端炔的 β,γ-不饱和 α-酮酯与 In(BF 4 ) 3和手性磷酸的手性路易斯酸络合物进行对映选择性催化共轭加成时,发现了可切换的对映选择性。
更新日期:2024-04-24
中文翻译:

不对称二元酸催化 β,γ-不饱和 α-酮酯共轭炔加成中的可切换对映选择性
在将带有末端炔的 β,γ-不饱和 α-酮酯与 In(BF 4 ) 3和手性磷酸的手性路易斯酸络合物进行对映选择性催化共轭加成时,发现了可切换的对映选择性。



























 京公网安备 11010802027423号
京公网安备 11010802027423号