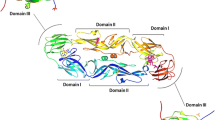
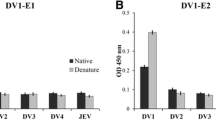
Abstract
Dengue is one of the most frequently transmitted viral infections globally which creates a serious burden to the healthcare system in many countries in the tropical and subtropical regions. To date, no vaccine has demonstrated balanced protection against the four dengue serotypes. Dengvaxia as the only vaccine that has been licensed for use in endemic areas has shown an increased risk in dengue-naïve vaccines to develop severe dengue. A crucial element in protection from dengue infection is the neutralizing antibody responses. Therefore, the identification of protective linear B-cell epitopes can guide vaccine design and facilitate the development of monoclonal antibodies as dengue therapeutics. This review summarizes the identification of dengue B-cell epitopes within the envelope (E) protein of dengue that can be incorporated into peptide vaccine constructs. These epitopes have been identified through approaches such as bioinformatics, three-dimensional structure analysis of antibody-dengue complexes, mutagenesis/alanine scanning and escape mutant studies. Additionally, the therapeutic potential of monoclonal antibodies targeting the E protein of dengue is reviewed. This can provide a basis for the design of future dengue therapies.
Similar content being viewed by others
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
Murray NEA, Quam MB, Wilder-Smith A (2013) Epidemiology of dengue: past, present and future prospects. Clin Epidemiol 5:299
Kalayanarooj S (2011) Clinical manifestations and management of dengue/DHF/DSS. Trop Med Health 39:83
Reich NG, Shrestha S, King AA, Rohani P, Lessler J, Kalayanarooj S, Yoon I-K, Gibbons RV, Burke DS, Cummings DAT (2013) Interactions between serotypes of dengue highlight epidemiological impact of cross-immunity. J R Soc Interface 10:20130414
Dejnirattisai W, Jumnainsong A, Onsirisakul N, Fitton P, Vasanawathana S, Limpitikul W, Puttikhunt C, Edwards C, Duangchinda T, Supasa S, Chawansuntati K, Malasit P, Mongkolsapaya J, Screaton G (2010) Cross-reacting antibodies enhance dengue virus infection in humans. Science (New York, N.Y.) 328:745
Recker M, Vannice K, Hombach J, Jit M, Simmons CP (2016) Assessing dengue vaccination impact: model challenges and future directions. Vaccine 34:4461
Ramanathan B, Poh CL, Kirk K, McBride WJH, Aaskov J, Grollo L (2016) Synthetic B-cell epitopes eliciting cross-neutralizing antibodies: strategies for future dengue vaccine. PLoS ONE 11:e0155900
Capeding MR, Tran NH, Hadinegoro SRS, Ismail HIHJM, Chotpitayasunondh T, Chua MN, Luong CQ, Rusmil K, Wirawan DN, Nallusamy R, Pitisuttithum P, Thisyakorn U, Yoon I-K, van der Vliet D, Langevin E, Laot T, Hutagalung Y, Frago C, Boaz M, Wartel TA, Tornieporth NG, Saville M, Bouckenooghe A (2014) Clinical efficacy and safety of a novel tetravalent dengue vaccine in healthy children in Asia: a phase 3, randomised, observer-masked, placebo-controlled trial. Lancet (London, England) 384:1358
Villar L, Dayan GH, Arredondo-García JL, Rivera DM, Cunha R, Deseda C, Reynales H, Costa MS, Morales-Ramírez JO, Carrasquilla G, Rey LC, Dietze R, Luz K, Rivas E, Miranda Montoya MC, Cortés Supelano M, Zambrano B, Langevin E, Boaz M, Tornieporth N, Saville M, Noriega F (2015) Efficacy of a tetravalent dengue vaccine in children in Latin America. N Engl J Med 372:113
Rosa BR, da Cunha AJLA, Medronho RA (2019) Efficacy, immunogenicity and safety of a recombinant tetravalent dengue vaccine (CYD-TDV) in children aged 2–17 years: systematic review and meta-analysis. BMJ Open 9:e019368
Guzman MG, Gubler DJ, Izquierdo A, Martinez E, Halstead SB (2016) Dengue infection. Nat Rev Dis Primers 2:16055
Falgout B, Markoff L (1995) Evidence that flavivirus NS1-NS2A cleavage is mediated by a membrane-bound host protease in the endoplasmic reticulum. J Virol 69:7232
Gromowski GD, Barrett ND, Barrett ADT (2008) Characterization of dengue virus complex-specific neutralizing epitopes on envelope protein domain III of dengue 2 virus. J Virol 82:8828
Modis Y, Ogata S, Clements D, Harrison SC (2003) A ligand-binding pocket in the dengue virus envelope glycoprotein. Proc Natl Acad Sci USA 100:6986
Rodenhuis-Zybert IA, Wilschut J, Smit JM (2010) Dengue virus life cycle: viral and host factors modulating infectivity. Cell Mol Life Sci 67:2773
Anasir MI, Ramanathan B, Poh CL (2020) Structure-based design of antivirals against envelope glycoprotein of dengue virus. Viruses 12:367
Mukhopadhyay S, Kuhn RJ, Rossmann MG (2005) A structural perspective of the flavivirus life cycle. Nat Rev Microbiol 3:13
Perera-Lecoin M, Meertens L, Carnec X, Amara A (2013) Flavivirus entry receptors: an update. Viruses 6:69
Roehrig JT, Butrapet S, Liss NM, Bennett SL, Luy BE, Childers T, Boroughs KL, Stovall JL, Calvert AE, Blair CD, Huang CY-H (2013) Mutation of the dengue virus type 2 envelope protein heparan sulfate binding sites or the domain III lateral ridge blocks replication in vero cells prior to membrane fusion. Virology 441:114
Roehrig JT, Bolin RA, Kelly RG (1998) Monoclonal antibody mapping of the envelope glycoprotein of the dengue 2 virus, Jamaica. Virology 246:317
Gromowski GD, Barrett ADT (2007) Characterization of an antigenic site that contains a dominant, type-specific neutralization determinant on the envelope protein domain III (ED3) of dengue 2 virus. Virology 366:349
Thullier P, Demangel C, Bedouelle H, Mégret F, Jouan A, Deubel V, Mazié J-C, Lafaye P (2001) Mapping of a dengue virus neutralizing epitope critical for the infectivity of all serotypes: insight into the neutralization mechanism. J Gen Virol 82:1885
Wahala WM, Claire H, Siritorn B, White LJ, de Silva AM (2012) Recombinant dengue type 2 viruses with altered e protein domain III epitopes are efficiently neutralized by human immune sera. J Virol 86:4019
Sukupolvi-Petty S, Austin SK, Purtha WE, Oliphant T, Nybakken GE, Schlesinger JJ, Roehrig JT, Gromowski GD, Barrett AD, Fremont DH, Diamond MS (2007) Type- and subcomplex-specific neutralizing antibodies against domain III of dengue virus type 2 envelope protein recognize adjacent epitopes. J Virol 81:12816
Pokidysheva E, Zhang Y, Battisti J, Bator C, Chipman P, Xiao C, Gregorio GG, Hendrickson W, Kuhn R, Rossmann M (2006) Cryo-EM reconstruction of dengue virus in complex with the carbohydrate recognition domain of DC-SIGN. Cell 124:485
Alen MMF, Dallmeier K, Balzarini J, Neyts J, Schols D (2012) Crucial role of the N-glycans on the viral E-envelope glycoprotein in DC-SIGN-mediated dengue virus infection. Antiviral Res 96:280
Modis Y, Ogata S, Clements D, Harrison SC (2004) Structure of the dengue virus envelope protein after membrane fusion. Nature 427:313
Klein DE, Choi JL, Harrison SC (2013) Structure of a dengue virus envelope protein late-stage fusion intermediate. J Virol 87:2287
Liao M, Sánchez-San Martín C, Zheng A, Kielian M (2010) In Vitro reconstitution reveals key intermediate states of trimer formation by the dengue virus membrane fusion protein. J Virol 84:5730
Byk LA, Iglesias NG, De Maio FA, Gebhard LG, Rossi M, Gamarnik AV (2016) Dengue virus genome uncoating requires ubiquitination. MBio 7:e00804-16
Reid DW, Campos RK, Child JR, Zheng T, Chan KWK, Bradrick SS, Vasudevan SG, Garcia-Blanco MA, Nicchitta CV (2018) Dengue virus selectively annexes endoplasmic reticulum-associated translation machinery as a strategy for co-opting host cell protein synthesis. J Virol 92:e01766-17
Perera R, Kuhn RJ (2008) Structural proteomics of dengue virus. Curr Opin Microbiol 11:369
Moi ML, Takasaki T, Kurane I (2016) Human antibody response to dengue virus: implications for dengue vaccine design. Trop Med Health 44:1
Wahala WMPB, de Silva AM (2011) The human antibody response to dengue virus infection. Viruses 3:2374
Kulkarni R (2020) Antibody-dependent enhancement of viral infections. In: Dynamics of immune activation in viral diseases
Chau TNB, Quyen NTH, Thuy TT, Tuan NM, Hoang DM, Dung NTP, Lien LB, Quy NT, Hieu NT, Hieu LTM, Hien TT, Hung NT, Farrar J, Simmons CP (2008) Dengue in Vietnamese infants-results of infection-enhancement assays correlate with age-related disease epidemiology, and cellular immune responses correlate with disease severity. J Infect Dis 198:516
Chau TNB, Hieu NT, Anders KL, Wolbers M, Lien LB, Hieu LTM, Hien TT, Hung NT, Farrar J, Whitehead S, Simmons CP (2009) Dengue virus infections and maternal antibody decay in a prospective birth cohort study of Vietnamese infants. J Infect Dis 200:1893
Anasir MI, Poh CL (2019) Structural vaccinology for viral vaccine design. Front Microbiol 10:738
Goncalvez AP, Engle RE, St-Claire M, Purcell RH, Lai C-J (2007) Monoclonal antibody-mediated enhancement of dengue virus infection in vitro and in vivo and strategies for prevention. Proc Natl Acad Sci USA 104:9422
Katzelnick LC, Gresh L, Halloran ME, Mercado JC, Kuan G, Gordon A, Balmaseda A, Harris E (2017) Antibody-dependent enhancement of severe dengue disease in humans. Science (New York, N.Y.) 358:929
Kirkpatrick BD, Durbin AP, Pierce KK, Carmolli MP, Tibery CM, Grier PL, Hynes N, Diehl SA, Elwood D, Jarvis AP, Sabundayo BP, Lyon CE, Larsson CJ, Jo M, Lovchik JM, Luke CJ, Walsh MC, Fraser EA, Subbarao K, Whitehead SS (2015) Robust and balanced immune responses to all 4 dengue virus serotypes following administration of a single dose of a live attenuated tetravalent dengue vaccine to healthy, flavivirus-naive adults. J Infect Dis 212:702
Durbin AP, Kirkpatrick BD, Pierce KK, Carmolli MP, Tibery CM, Grier PL, Hynes N, Opert K, Jarvis AP, Sabundayo BP, McElvany BD, Sendra EA, Larsson CJ, Jo M, Lovchik JM, Luke CJ, Walsh MC, Fraser EA, Subbarao K, Whitehead SS (2016) A 12-month-interval dosing study in adults indicates that a single dose of the National Institute of allergy and infectious diseases tetravalent dengue vaccine induces a robust neutralizing antibody response. J Infect Dis 214:832
Kirkpatrick BD, Whitehead SS, Pierce KK, Tibery CM, Grier PL, Hynes NA, Larsson CJ, Sabundayo BP, Talaat KR, Janiak A, Carmolli MP, Luke CJ, Diehl SA, Durbin AP (2016) the live attenuated dengue vaccine TV003 elicits complete protection against dengue in a human challenge model. Sci Transl Med 8:330ra36
Whitehead SS, Durbin AP, Pierce KK, Elwood D, McElvany BD, Fraser EA, Carmolli MP, Tibery CM, Hynes NA, Jo M, Lovchik JM, Larsson CJ, Doty EA, Dickson DM, Luke CJ, Subbarao K, Diehl SA, Kirkpatrick BD (2017) In a randomized trial, the live attenuated tetravalent dengue vaccine TV003 is well-tolerated and highly immunogenic in subjects with flavivirus exposure prior to vaccination. PLoS Negl Trop Dis 11:e0005584
Kallas EG, Precioso AR, Palacios R, Thomé B, Braga PE, Vanni T, Campos LMA, Ferrari L, Mondini G, da Graça Salomão M, da Silva A, Espinola HM, do Prado Santos J, Santos CLS, Timenetsky MCST, Miraglia JL, Gallina NMF, Weiskopf D, Sette A, Goulart R, Salles RT, Maestri A, Sallum AME, Farhat SCL, Sakita NK, Ferreira JCOA, Silveira CGT, Costa PR, Raw I, Whitehead SS, Durbin AP, Kalil J (2020) Safety and immunogenicity of the tetravalent, live-attenuated dengue vaccine butantan-DV in adults in Brazil: a two-step, double-blind, randomised placebo-controlled phase 2 trial. Lancet Infect Dis 20:839
Durbin AP, Kirkpatrick BD, Pierce KK, Elwood D, Larsson CJ, Lindow JC, Tibery C, Sabundayo BP, Shaffer D, Talaat KR, Hynes NA, Wanionek K, Carmolli MP, Luke CJ, Murphy BR, Subbarao K, Whitehead SS (2013) A single dose of any of four different live attenuated tetravalent dengue vaccines is safe and immunogenic in flavivirus-naive adults: a randomized, double-blind clinical trial. J Infect Dis 207:957
Danko JR, Kochel T, Teneza-Mora N, Luke TC, Raviprakash K, Sun P, Simmons M, Moon JE, De La Barrera R, Martinez LJ, Thomas SJ, Kenney RT, Smith L, Porter KR (2018) Safety and immunogenicity of a tetravalent dengue DNA vaccine administered with a cationic lipid-based adjuvant in a phase 1 clinical trial. Am J Trop Med Hyg 98:849
Porter KR, Ewing D, Chen L, Wu S-J, Hayes CG, Ferrari M, Teneza-Mora N, Raviprakash K (2012) Immunogenicity and protective efficacy of a vaxfectin-adjuvanted tetravalent dengue DNA vaccine. Vaccine 30:336
Manoff SB, Sausser M, Falk Russell A, Martin J, Radley D, Hyatt D, Roberts CC, Lickliter J, Krishnarajah J, Bett A, Dubey S, Finn T, Coller B-A (2019) Immunogenicity and safety of an investigational tetravalent recombinant subunit vaccine for dengue: results of a phase I randomized clinical trial in flavivirus-naïve adults. Hum Vaccines Immunother 15:2195
Wilder-Smith A (2020) Dengue vaccine development by the year 2020: challenges and prospects. Curr Opin Virol 43:71
Nivarthi UK, Swanstrom J, Delacruz MJ, Patel B, Durbin AP, Whitehead SS, Kirkpatrick BD, Pierce KK, Diehl SA, Katzelnick L, Baric RS, de Silva AM (2021) A tetravalent live attenuated dengue virus vaccine stimulates balanced immunity to multiple serotypes in humans. Nat Commun 12:1102
Osorio JE, Wallace D, Stinchcomb DT, Recombinant A (2016) Chimeric tetravalent dengue vaccine candidate based on a dengue virus serotype 2 backbone. Expert Rev Vaccines 15:497
Biswal S, Reynales H, Saez-Llorens X, Lopez P, Borja-Tabora C, Kosalaraksa P, Sirivichayakul C, Watanaveeradej V, Rivera L, Espinoza F, Fernando L, Dietze R, Luz K, Venâncio da Cunha R, Jimeno J, López-Medina E, Borkowski A, Brose M, Rauscher M, LeFevre I, Bizjajeva S, Bravo L, Wallace D (2019) Efficacy of a tetravalent dengue vaccine in healthy children and adolescents. N Engl J Med 381:2009
Biswal S, Borja-Tabora C, Martinez Vargas L, Velásquez H, Theresa Alera M, Sierra V, Rodriguez-Arenales EJ, Yu D, Wickramasinghe VP, Duarte Moreira EJ, Fernando AD, Gunasekera D, Kosalaraksa P, Espinoza F, López-Medina E, Bravo L, Tuboi S, Hutagalung Y, Garbes P, Escudero I, Rauscher M, Bizjajeva S, LeFevre I, Borkowski A, Saez-Llorens X, Wallace D (2020) Efficacy of a tetravalent dengue vaccine in healthy children aged 4–16 years: a randomised, placebo-controlled, phase 3 trial. Lancet (London, England) 395:1423
Soria-Guerra RE, Nieto-Gomez R, Govea-Alonso DO, Rosales-Mendoza S (2015) An overview of bioinformatics tools for epitope prediction: implications on vaccine development. J Biomed Inform 53:405
Buus S, Rockberg J, Forsström B, Nilsson P, Uhlen M, Schafer-Nielsen C (2012) High-resolution mapping of linear antibody epitopes using ultrahigh-density peptide microarrays. Mol Cell Proteom MCP 11:1790
Ali M, Pandey RK, Khatoon N, Narula A, Mishra A, Prajapati VK (2017) Exploring dengue genome to construct a multi-epitope based subunit vaccine by utilizing immunoinformatics approach to battle against dengue infection. Sci Rep 7:9232
Tahir RA, Wu H, Rizwan MA, Jafar TH, Saleem S, Sehgal SA (2018) Immunoinformatics and molecular docking studies reveal potential epitope-based peptide vaccine against DENV-NS3 protein. J Theor Biol 459:162
Li S, Peng L, Zhao W, Zhong H, Zhang F, Yan Z, Cao H (2011) Synthetic peptides containing B- and T-cell epitope of dengue virus-2 E domain III provoked B- and T-cell responses. Vaccine 29:3695
Sánchez-Burgos G, Ramos-Castañeda J, Cedillo-Rivera R, Dumonteil E (2010) Immunogenicity of novel dengue virus epitopes identified by bioinformatic analysis. Virus Res 153:113
Muthusamy K, Gopinath K, Nandhini D (2016) Computational prediction of immunodominant antigenic regions & potential protective epitopes for dengue vaccination. Indian J Med Res 144:587
Chan Y, Jazayeri SD, Ramanathan B, Poh CL (2020) Enhancement of tetravalent immune responses to highly conserved epitopes of a dengue peptide vaccine conjugated to polystyrene nanoparticles. Vaccines 8:417
Durham ND, Agrawal A, Waltari E, Croote D, Zanini F, Fouch M, Davidson E, Smith O, Carabajal E, Pak JE, Doranz BJ, Robinson M, Sanz AM, Albornoz LL, Rosso F, Einav S, Quake SR, McCutcheon KM, Goo L (2019) Broadly neutralizing human antibodies against dengue virus identified by single B cell transcriptomics. Elife 8:e52384
Deng Y-Q, Dai J-X, Ji G-H, Jiang T, Wang H-J, Yang H, Tan W-L, Liu R, Yu M, Ge B-X, Zhu Q-Y, Qin E-D, Guo Y-J, Qin C-F (2011) A broadly flavivirus cross-neutralizing monoclonal antibody that recognizes a novel epitope within the fusion loop of E protein. PLoS ONE 6:e16059
Rodenhuis-Zybert IA, Moesker B, da Silva Voorham JM, van der Ende-Metselaar H, Diamond MS, Wilschut J, Smit JM (2011) A fusion-loop antibody enhances the infectious properties of immature flavivirus particles. J Virol 85:11800
Li J, Watterson D, Chang C-W, Che X-Y, Li X-Q, Ericsson DJ, Qiu L-W, Cai J-P, Chen J, Fry SR, Cheung STM, Cooper MA, Young PR, Kobe B (2018) Structural and functional characterization of a cross-reactive dengue virus neutralizing antibody that recognizes a cryptic epitope. Structure (London, England: 1993) 26:51
Cockburn JJB, Navarro Sanchez ME, Fretes N, Urvoas A, Staropoli I, Kikuti CM, Coffey LL, Arenzana Seisdedos F, Bedouelle H, Rey FA (2012) Mechanism of dengue virus broad cross-neutralization by a monoclonal antibody. Structure (London, England: 1993) 20:303
Midgley CM, Flanagan A, Tran HB, Dejnirattisai W, Chawansuntati K, Jumnainsong A, Wongwiwat W, Duangchinda T, Mongkolsapaya J, Grimes JM, Screaton GR (2012) Structural analysis of a dengue cross-reactive antibody complexed with envelope domain III reveals the molecular basis of cross-reactivity. J Immunol (Baltimore, Md: 1950) 188:4971
Renner M, Flanagan A, Dejnirattisai W, Puttikhunt C, Kasinrerk W, Supasa P, Wongwiwat W, Chawansuntati K, Duangchinda T, Cowper A, Midgley CM, Malasit P, Huiskonen JT, Mongkolsapaya J, Screaton GR, Grimes JM (2018) Characterization of a potent and highly unusual minimally enhancing antibody directed against dengue virus. Nat Immunol 19:1248
Rouvinski A, Dejnirattisai W, Guardado-Calvo P, Vaney M-C, Sharma A, Duquerroy S, Supasa P, Wongwiwat W, Haouz A, Barba-Spaeth G, Mongkolsapaya J, Rey FA, Screaton GR (2017) Covalently linked dengue virus envelope glycoprotein dimers reduce exposure of the immunodominant fusion loop epitope. Nat Commun 8:15411
Rouvinski A, Guardado-Calvo P, Barba-Spaeth G, Duquerroy S, Vaney M-C, Kikuti CM, Navarro Sanchez ME, Dejnirattisai W, Wongwiwat W, Haouz A, Girard-Blanc C, Petres S, Shepard WE, Desprès P, Arenzana-Seisdedos F, Dussart P, Mongkolsapaya J, Screaton GR, Rey FA (2015) Recognition determinants of broadly neutralizing human antibodies against dengue viruses. Nature 520:109
Robbiani DF, Bozzacco L, Keeffe JR, Khouri R, Olsen PC, Gazumyan A, Schaefer-Babajew D, Avila-Rios S, Nogueira L, Patel R, Azzopardi SA, Uhl LFK, Saeed M, Sevilla-Reyes EE, Agudelo M, Yao K-H, Golijanin J, Gristick HB, Lee YE, Hurley A, Caskey M, Pai J, Oliveira T, Wunder EAJ, Sacramento G, Nery NJ, Orge C, Costa F, Reis MG, Thomas NM, Eisenreich T, Weinberger DM, de Almeida ARP, West APJ, Rice CM, Bjorkman PJ, Reyes-Teran G, Ko AI, MacDonald MR, Nussenzweig MC (2017) Recurrent potent human neutralizing antibodies to Zika virus in Brazil and Mexico. Cell 169:597
Austin SK, Dowd KA, Shrestha B, Nelson CA, Edeling MA, Johnson S, Pierson TC, Diamond MS, Fremont DH (2012) Structural basis of differential neutralization of DENV-1 genotypes by an antibody that recognizes a cryptic epitope. PLoS Pathog 8:e1002930
Cockburn JJB, Navarro Sanchez ME, Goncalvez AP, Zaitseva E, Stura EA, Kikuti CM, Duquerroy S, Dussart P, Chernomordik LV, Lai C-J, Rey FA (2012) Structural insights into the neutralization mechanism of a higher primate antibody against dengue virus. EMBO J 31:767
Lok S-M, Kostyuchenko V, Nybakken GE, Holdaway HA, Battisti AJ, Sukupolvi-Petty S, Sedlak D, Fremont DH, Chipman PR, Roehrig JT, Diamond MS, Kuhn RJ, Rossmann MG (2008) Binding of a neutralizing antibody to dengue virus alters the arrangement of surface glycoproteins. Nat Struct Mol Biol 15:312
Chen W-H, Chou F-P, Wang Y-K, Huang S-C, Cheng C-H, Wu T-K (2017) Characterization and epitope mapping of dengue virus type 1 specific monoclonal antibodies. Virology Journal 14:189
Edeling MA, Austin SK, Shrestha B, Dowd KA, Mukherjee S, Nelson CA, Johnson S, Mabila MN, Christian EA, Rucker J, Pierson TC, Diamond MS, Fremont DH (2014) Potent dengue virus neutralization by a therapeutic antibody with low monovalent affinity requires bivalent engagement. PLoS Pathog 10:e1004072
Wu K-P, Wu C-W, Tsao Y-P, Kuo T-W, Lou Y-C, Lin C-W, Wu S-C, Cheng J-W (2003) Structural basis of a flavivirus recognized by its neutralizing antibody: solution structure of the domain III of the Japanese encephalitis virus envelope protein. J Biol Chem 278:46007
Volk DE, Beasley DWC, Kallick DA, Holbrook MR, Barrett ADT, Gorenstein DG (2004) solution structure and antibody binding studies of the envelope protein domain III from the New York Strain of West Nile Virus. J Biol Chem 279:38755
Cui X, Wu Y, Fan D, Gao N, Ming Y, Wang P, An J (2018) Peptides P4 and P7 derived from e protein inhibit entry of dengue virus serotype 2 via interacting with Β3 integrin. Antivir Res 155:20
Wahala WMPB, Huang C, Butrapet S, White LJ, de Silva AM (2012) Recombinant dengue type 2 viruses with altered e protein domain III epitopes are efficiently neutralized by human immune sera. J Virol 86:4019
Wahala WMPB, Kraus AA, Haymore LB, Accavitti-Loper MA, de Silva AM (2009) Dengue virus neutralization by human immune sera: role of envelope protein domain III-reactive antibody. Virology 392:103
Keeffe JR, Van Rompay KKA, Olsen PC, Wang Q, Gazumyan A, Azzopardi SA, Schaefer-Babajew D, Lee YE, Stuart JB, Singapuri A, Watanabe J, Usachenko J, Ardeshir A, Saeed M, Agudelo M, Eisenreich T, Bournazos S, Oliveira TY, Rice CM, Coffey LL, MacDonald MR, Bjorkman PJ, Nussenzweig MC, Robbiani DF (2018) A combination of two human monoclonal antibodies prevents Zika virus escape mutations in non-human primates. Cell Rep 25:1385
Shrestha B, Brien JD, Sukupolvi-Petty S, Austin SK, Edeling MA, Kim T, O’Brien KM, Nelson CA, Johnson S, Fremont DH, Diamond MS (2010) The development of therapeutic antibodies that neutralize homologous and heterologous genotypes of dengue virus type 1. PLoS Pathog 6:e1000823
Men R, Yamashiro T, Goncalvez AP, Wernly C, Schofield DJ, Emerson SU, Purcell RH, Lai C-J (2004) Identification of chimpanzee fab fragments by repertoire cloning and production of a full-length humanized immunoglobulin G1 antibody that is highly efficient for neutralization of dengue type 4 virus. J Virol 78:4665
de Alwis R, Smith SA, Olivarez NP, Messer WB, Huynh JP, Wahala WMPB, White LJ, Diamond MS, Baric RS, Crowe JEJ, de Silva AM (2012) Identification of human neutralizing antibodies that bind to complex epitopes on dengue virions. Proc Natl Acad Sci USA 109:7439
Fibriansah G, Tan JL, Smith SA, de Alwis AR, Ng T-S, Kostyuchenko VA, Ibarra KD, Wang J, Harris E, de Silva A, Crowe JEJ, Lok S-M (2014) A potent anti-dengue human antibody preferentially recognizes the conformation of e protein monomers assembled on the virus surface. EMBO Mol Med 6:358
Fibriansah G, Ibarra KD, Ng T-S, Smith SA, Tan JL, Lim X-N, Ooi JSG, Kostyuchenko VA, Wang J, de Silva AM, Harris E, Crowe JEJ, Lok S-M (2015) Cryo-EM structure of an antibody that neutralizes dengue virus type 2 by locking e protein dimers. Science (New York, N.Y.) 349:88
Fibriansah G, Tan JL, Smith SA, de Alwis R, Ng T-S, Kostyuchenko VA, Jadi RS, Kukkaro P, de Silva AM, Crowe JE, Lok S-M (2015) A highly potent human antibody neutralizes dengue virus serotype 3 by binding across three surface proteins. Nat Commun 6:6341
Teoh EP, Kukkaro P, Teo EW, Lim APC, Tan TT, Yip A, Schul W, Aung M, Kostyuchenko VA, Leo YS, Chan SH, Smith KGC, Chan AHY, Zou G, Ooi EE, Kemeny DM, Tan GK, Ng JKW, Ng ML, Alonso S, Fisher D, Shi P-Y, Hanson BJ, Lok S-M, MacAry PA (2012) The structural basis for serotype-specific neutralization of dengue virus by a human antibody. Sci Transl Med 4:139ra83
Li X-Q, Qiu L-W, Chen Y, Wen K, Cai J-P, Chen J, Pan Y-X, Li J, Hu D-M, Huang Y-F, Liu L-D, Ding X-X, Guo Y-H, Che X-Y (2013) Dengue virus envelope domain III immunization elicits predominantly cross-reactive, poorly neutralizing antibodies localized to the AB loop: implications for dengue vaccine design. J Gen Virol 94:2191
Dejnirattisai W, Wongwiwat W, Supasa S, Zhang X, Dai X, Rouvinski A, Jumnainsong A, Edwards C, Quyen NTH, Duangchinda T, Grimes JM, Tsai W-Y, Lai C-Y, Wang W-K, Malasit P, Farrar J, Simmons CP, Zhou ZH, Rey FA, Mongkolsapaya J, Screaton GR (2015) A new class of highly potent, broadly neutralizing antibodies isolated from viremic patients infected with dengue virus. Nat Immunol 16:170
Yamanaka A, Kotaki T, Konishi E (2013) A mouse monoclonal antibody against dengue virus type 1 mochizuki strain targeting envelope protein domain II and displaying strongly neutralizing but not enhancing activity. J Virol 87:12828
Thullier P, Lafaye P, Mégret F, Deubel V, Jouan A, Mazié JC (1999) A recombinant fab neutralizes dengue virus in vitro. J Biotechnol 69:183
Sanchez-Trincado JL, Gomez-Perosanz M, Reche PA (2017) Fundamentals and methods for T- and B-cell epitope prediction. J Immunol Res 2017:2680160
Li L, Meng W, Horton M, DiStefano DR, Thoryk EA, Pfaff JM, Wang Q, Salazar GT, Barnes T, Doranz BJ, Bett AJ, Casimiro DR, Vora KA, An Z, Zhang N (2019) Potent neutralizing antibodies elicited by dengue vaccine in rhesus macaque target diverse epitopes. PLoS Pathog 15:e1007716
Xu M, Zuest R, Velumani S, Tukijan F, Toh YX, Appanna R, Tan EY, Cerny D, MacAry P, Wang C-I, Fink K (2017) A potent neutralizing antibody with therapeutic potential against all four serotypes of dengue virus. NPJ Vaccines 2:2
Hu D, Zhu Z, Li S, Deng Y, Wu Y, Zhang N, Puri V, Wang C, Zou P, Lei C, Tian X, Wang Y, Zhao Q, Li W, Prabakaran P, Feng Y, Cardosa J, Qin C, Zhou X, Dimitrov DS, Ying T (2019) A broadly neutralizing germline-like human monoclonal antibody against dengue virus envelope domain III. PLoS Pathog 15:e1007836
Injampa S, Muenngern N, Pipattanaboon C, Benjathummarak S, Boonha K, Hananantachai H, Wongwit W, Ramasoota P, Pitaksajjakul P (2017) Generation and characterization of cross neutralizing human monoclonal antibody against 4 serotypes of dengue virus without enhancing activity. PeerJ 5:e4021
Smith SA, de Alwis AR, Kose N, Harris E, Ibarra KD, Kahle KM, Pfaff JM, Xiang X, Doranz BJ, de Silva AM, Austin SK, Sukupolvi-Petty S, Diamond MS, Crowe JEJ (2013) The potent and broadly neutralizing human dengue virus-specific monoclonal antibody 1C19 reveals a unique cross-reactive epitope on the Bc loop of domain II of the envelope protein. MBio 4:e00873
Simmons M, Putnak R, Sun P, Burgess T, Marasco WA (2016) Antibody prophylaxis against dengue virus 2 infection in non-human primates. Am J Trop Med Hyg 95:1148
Ong EZ, Budigi Y, Tan HC, Robinson LN, Rowley KJ, Winnett A, Hobbie S, Shriver Z, Babcock GJ, Ooi EE (2017) Preclinical evaluation of VIS513, a therapeutic antibody against dengue virus, in non-human primates. Antivir Res 144:44
Funding
This work was supported by the Ministry of Education, Malaysia (FRGS/1/2020/SKK06/SYUC/03/1) and (FRGS/1/2021/SKK0/SYUC/01/1).
Author information
Authors and Affiliations
Contributions
MIA wrote the manuscript. CLP provided the critical revision and final approval of the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Not applicable.
Ethics approval
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Edited by Volker Lohmann.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Anasir, M.I., Poh, C.L. Discovery of B-cell epitopes for development of dengue vaccines and antibody therapeutics. Med Microbiol Immunol 211, 1–18 (2022). https://doi.org/10.1007/s00430-021-00726-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00430-021-00726-1