Abstract
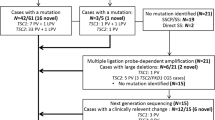
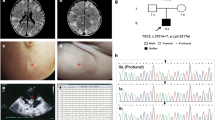
Tuberous sclerosis complex (TSC) is mainly caused by variants in TSC1 and TSC2, which encodes hamartin protein and tuberin protein, respectively. Here, we report clinical and molecular characteristics of 18 families with TSC. High-throughput DNA sequencing was employed to detect variants in all the exons and flanking region of TSC1 and TSC2. TA clone and real-time PCR were performed to verify the pathogenicity of candidate variants. A total of 17 mutations were identified, including 13 mutations in TSC2 and 4 mutations in TSC1. Fifty-six percent (10/18) of the families carried de novo mutations, and 8 of these mutations were not reported previously. Most mutations detected were loss-of-function mutations (15/17). One splice-site mutation (TSC2 c.599 + 5G > A) caused abnormal splicing and was confirmed by in vitro analysis. Facial angiofibromas (94%) and epilepsy (89%) were the most prevalent clinical features in our patients. Treatment with anti-seizure medication (ASM) or in combination with rapamycin results in clinical remission in most patients with TSC-associated seizures (14/15). For genotype–phenotype correlation, patients in our cohort with TSC2 mutations had an earlier onset age and patients with TSC1 showed better response to ASM. Our study has expanded the spectrum of TSC1 and TSC2 causing TSC.


Similar content being viewed by others
References
O’Callaghan FJ, Shiell AW, Osborne JP, Martyn CN (1998) Prevalence of tuberous sclerosis estimated by capture-recapture analysis. Lancet 351:1490. https://doi.org/10.1016/S0140-6736(05)78872-3
Sampson JR, Scahill SJ, Stephenson JB, Mann L, Connor JM (1989) Genetic aspects of tuberous sclerosis in the west of Scotland. J Med Genet 26:28–31
Leung AK, Robson WL (2007) Tuberous sclerosis complex: a review. J Pediatr Health Care 21:108–114. https://doi.org/10.1016/j.pedhc.2006.05.004
Curatolo P, Moavero R, de Vries PJ (2015) Neurological and neuropsychiatric aspects of tuberous sclerosis complex. Lancet Neurol 14:733–745. https://doi.org/10.1016/S1474-4422(15)00069-1
Sancak O, Nellist M, Goedbloed M, Elfferich P, Wouters C, Maat-Kievit A, Zonnenberg B, Verhoef S, Halley D, van den Ouweland A (2005) Mutational analysis of the TSC1 and TSC2 genes in a diagnostic setting: genotype–phenotype correlations and comparison of diagnostic DNA techniques in tuberous sclerosis complex. Eur J Hum Genet 13:731–741. https://doi.org/10.1038/sj.ejhg.5201402
Dabora SL, Jozwiak S, Franz DN, Roberts PS, Nieto A, Chung J, Choy YS, Reeve MP, Thiele E, Egelhoff JC, Kasprzyk-Obara J, Domanska-Pakiela D, Kwiatkowski DJ (2001) Mutational analysis in a cohort of 224 tuberous sclerosis patients indicates increased severity of TSC2, compared with TSC1, disease in multiple organs. Am J Hum Genet 68:64–80. https://doi.org/10.1086/316951
Jones AC, Shyamsundar MM, Thomas MW, Maynard J, Idziaszczyk S, Tomkins S, Sampson JR, Cheadle JP (1999) Comprehensive mutation analysis of TSC1 and TSC2-and phenotypic correlations in 150 families with tuberous sclerosis. Am J Hum Genet 64:1305–1315. https://doi.org/10.1086/302381
Tyburczy ME, Dies KA, Glass J, Camposano S, Chekaluk Y, Thorner AR, Lin L, Krueger D, Franz DN, Thiele EA, Sahin M, Kwiatkowski DJ (2015) Mosaic and intronic mutations in TSC1/TSC2 explain the majority of TSC patients with no mutation identified by conventional testing. PLoS Genet 11:e1005637. https://doi.org/10.1371/journal.pgen.1005637
Jones AC, Daniells CE, Snell RG, Tachataki M, Idziaszczyk SA, Krawczak M, Sampson JR, Cheadle JP (1997) Molecular genetic and phenotypic analysis reveals differences between TSC1 and TSC2 associated familial and sporadic tuberous sclerosis. Hum Mol Genet 6:2155–2161. https://doi.org/10.1093/hmg/6.12.2155
Northrup H, Krueger DA, International Tuberous Sclerosis Complex Consensus G (2013) Tuberous sclerosis complex diagnostic criteria update: recommendations of the 2012 International Tuberous Sclerosis Complex Consensus Conference. Pediatr Neurol 49:243–254. https://doi.org/10.1016/j.pediatrneurol.2013.08.001
van Slegtenhorst M, de Hoogt R, Hermans C, Nellist M, Janssen B, Verhoef S, Lindhout D, van den Ouweland A, Halley D, Young J, Burley M, Jeremiah S, Woodward K, Nahmias J, Fox M, Ekong R, Osborne J, Wolfe J, Povey S, Snell RG, Cheadle JP, Jones AC, Tachataki M, Ravine D, Sampson JR, Reeve MP, Richardson P, Wilmer F, Munro C, Hawkins TL, Sepp T, Ali JB, Ward S, Green AJ, Yates JR, Kwiatkowska J, Henske EP, Short MP, Haines JH, Jozwiak S, Kwiatkowski DJ (1997) Identification of the tuberous sclerosis gene TSC1 on chromosome 9q34. Science 277:805–808
European Chromosome 16 Tuberous Sclerosis C (1993) Identification and characterization of the tuberous sclerosis gene on chromosome 16. Cell 75:1305–1315
Tee AR, Fingar DC, Manning BD, Kwiatkowski DJ, Cantley LC, Blenis J (2002) Tuberous sclerosis complex-1 and -2 gene products function together to inhibit mammalian target of rapamycin (mTOR)-mediated downstream signaling. Proc Natl Acad Sci U S A 99:13571–13576. https://doi.org/10.1073/pnas.202476899
Gao X, Zhang Y, Arrazola P, Hino O, Kobayashi T, Yeung RS, Ru B, Pan D (2002) Tsc tumour suppressor proteins antagonize amino-acid-TOR signalling. Nat Cell Biol 4:699–704. https://doi.org/10.1038/ncb847
Inoki K, Li Y, Zhu T, Wu J, Guan K-L (2002) TSC2 is phosphorylated and inhibited by Akt and suppresses mTOR signalling. Nat Cell Biol 4:648–657. https://doi.org/10.1038/ncb839
Ding Y, Wang J, Zhou S, Zhou Y, Zhang L, Yu L, Wang Y (2020) Genotype and phenotype analysis of Chinese children with tuberous sclerosis complex: a pediatric cohort study. Front Genet 11:204. https://doi.org/10.3389/fgene.2020.00204
Henske EP, Jozwiak S, Kingswood JC, Sampson JR, Thiele EA (2016) Tuberous sclerosis complex. Nat Rev Dis Primers 2:16035. https://doi.org/10.1038/nrdp.2016.35
Chu-Shore CJ, Major P, Camposano S, Muzykewicz D, Thiele EA (2010) The natural history of epilepsy in tuberous sclerosis complex. Epilepsia 51:1236–1241. https://doi.org/10.1111/j.1528-1167.2009.02474.x
Rosset C, Netto CBO, Ashton-Prolla P (2017) TSC1 and TSC2 gene mutations and their implications for treatment in tuberous sclerosis complex: a review. Genet Mol Biol 40:69–79. https://doi.org/10.1590/1678-4685-GMB-2015-0321
Napolioni V, Moavero R, Curatolo P (2009) Recent advances in neurobiology of tuberous sclerosis complex. Brain Dev 31:104–113. https://doi.org/10.1016/j.braindev.2008.09.013
Yang G, Shi ZN, Meng Y, Shi XY, Pang LY, Ma SF, Zhang MN, Wang YY, Zou LP (2017) Phenotypic and genotypic characterization of Chinese children diagnosed with tuberous sclerosis complex. Clin Genet 91:764–768. https://doi.org/10.1111/cge.12920
Rendtorff ND, Bjerregaard B, Frodin M, Kjaergaard S, Hove H, Skovby F, Brondum-Nielsen K, Schwartz M, Danish Tuberous Sclerosis G (2005) Analysis of 65 tuberous sclerosis complex (TSC) patients by TSC2 DGGE, TSC1/TSC2 MLPA, and TSC1 long-range PCR sequencing, and report of 28 novel mutations. Hum Mutat 26:374–383. https://doi.org/10.1002/humu.20227
Alipanahi HYXB (2015) The human splicing code reveals new insights into the genetic determinants of disease. Science 347:144. 0.1126/science.1254806
Coevoets R, Arican S, Hoogeveen-Westerveld M, Simons E, van den Ouweland A, Halley D, Nellist M (2009) A reliable cell-based assay for testing unclassified TSC2 gene variants. Eur J Hum Genet 17:301–310. https://doi.org/10.1038/ejhg.2008.184
Hoogeveen-Westerveld M, Wentink M, van den Heuvel D, Mozaffari M, Ekong R, Povey S, den Dunnen JT, Metcalfe K, Vallee S, Krueger S, Bergoffen J, Shashi V, Elmslie F, Kwiatkowski D, Sampson J, Vidales C, Dzarir J, Garcia-Planells J, Dies K, Maat-Kievit A, van den Ouweland A, Halley D, Nellist M (2011) Functional assessment of variants in the TSC1 and TSC2 genes identified in individuals with tuberous sclerosis complex. Hum Mutat 32:424–435. https://doi.org/10.1002/humu.21451
van Eeghen AM, Nellist M, van Eeghen EE, Thiele EA (2013) Central TSC2 missense mutations are associated with a reduced risk of infantile spasms. Epilepsy Res 103:83–87. https://doi.org/10.1016/j.eplepsyres.2012.07.007
Li S, Zhang Y, Wang Z, Yang Y, Gao W, Li D, Wei J (2018) Genotype-phenotype correlation of patients with tuberous sclerosis complex-associated renal angiomyolipoma: a descriptive study. Hum Pathol 82:61–67. https://doi.org/10.1016/j.humpath.2018.07.017
Maheshwar MM, Cheadle JP, Jones AC, Myring J, Fryer AE, Harris PC, Sampson JR (1997) The GAP-related domain of tuberin, the product of the TSC2 gene, is a target for missense mutations in tuberous sclerosis. Hum Mol Genet 6:1991–1996. https://doi.org/10.1093/hmg/6.11.1991
Dabora SL, Sigalas I, Hall F, Eng C, Vijg J, Kwiatkowski DJ (1998) Comprehensive mutation analysis of TSC1 using two-dimensional DNA electrophoresis with DGGE. Ann Hum Genet 62:491–504. https://doi.org/10.1046/j.1469-1809.1998.6260491.x
Rosset C, Vairo F, Bandeira IC, Correia RL, de Goes FV, da Silva RTB, Bueno LSM, de Miranda Gomes MCS, Galvao HCR, Neri J, Achatz MI, Netto CBO, Ashton-Prolla P (2017) Molecular analysis of TSC1 and TSC2 genes and phenotypic correlations in Brazilian families with tuberous sclerosis. PLoS ONE 12:e0185713. https://doi.org/10.1371/journal.pone.0185713
Zheng LY, Lee YW, Han Y, Tang LL, Cheng YY, Dou JF, Zhou FS, Zheng XD, Wang HY, Wang PG, Gao M (2018) Novel mutations in Chinese Han patients with tuberous sclerosis complex: case series and review of the published work. J Dermatol 45:867–870. https://doi.org/10.1111/1346-8138.14349
Reyna-Fabian ME, Hernandez-Martinez NL, Alcantara-Ortigoza MA, Ayala-Sumuano JT, Enriquez-Flores S, Velazquez-Aragon JA, Varela-Echavarria A, Todd-Quinones CG, Gonzalez-Del Angel A (2020) First comprehensive TSC1/TSC2 mutational analysis in Mexican patients with tuberous sclerosis complex reveals numerous novel pathogenic variants. Sci Rep 10:6589. https://doi.org/10.1038/s41598-020-62759-5
Niida Y, Lawrence-Smith N, Banwell A, Hammer E, Lewis J, Beauchamp RL, Sims K, Ramesh V, Ozelius L (1999) Analysis of both TSC1 and TSC2 for germline mutations in 126 unrelated patients with tuberous sclerosis. Hum Mutat 14:412–422. https://doi.org/10.1002/(SICI)1098-1004(199911)14:5%3c412::AID-HUMU7%3e3.0.CO;2-K
Lee KA, Won HS, Shim JY, Lee PR, Kim A (2013) Molecular genetic, cardiac and neurodevelopmental findings in cases of prenatally diagnosed rhabdomyoma associated with tuberous sclerosis complex. Ultrasound Obstet Gynecol 41:306–311. https://doi.org/10.1002/uog.11227
Acknowledgements
We thank all patients and their family members for their collaboration.
Funding
The study was supported by the Chinese Academy of Medical Sciences (CAMS) Innovation Fund for Medical Sciences (CIFMS) (Grant number: 2021-I2M-1–034, 2021-I2M-1–018, 2016-I2M-1–002), National Natural Science Foundation of China (NSFC; grant numbers 81971293, 81788101), National Key Research and Development Program of China (Grant number: 2016YFC0905100), and Research Fund from CAAE (UCB-2018029).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
This study was approved by Institutional Ethical Review Board of Peking Union Medical College Hospital.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Yin, K., Lin, N., Lu, Q. et al. Genetic analysis of 18 families with tuberous sclerosis complex. Neurogenetics 23, 223–230 (2022). https://doi.org/10.1007/s10048-022-00694-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10048-022-00694-5