Abstract
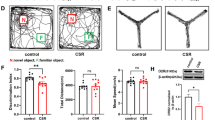
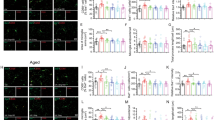
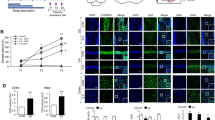
Sleep deprivation causes significant memory impairment in healthy adults. Extensive research has focused on identifying the biological mechanisms underlying memory impairment. Microglia-mediated synaptic elimination plays an indispensable role in sleep deprivation. Here, the potential role of the CD33/TREM2 signaling pathway in modulating memory decline during chronic sleep restriction (CSR) was evaluated. In this study, adult male C57BL/6 mice were sleep-restricted using an automated sleep deprivation apparatus for 20 h per day for 7 days. The Y-maze test revealed that spontaneous alternation was significantly reduced in CSR mice compared with control mice. The percentage of exploratory preference for the novel object in CSR mice was significantly decreased compared with that in control mice. These memory deficits correlated with aberrant microglial activation and increased phagocytic ability. Moreover, in CSR mice, the CD33 protein level in hippocampal tissue was significantly downregulated, but the TREM2 protein level was increased. In BV2 microglial cells, downregulation of CD33 increased TREM2 expression and improved microglial phagocytosis. Then, the sialic ligand monosialo-ganglioside 1 (GM1, 20 mg/kg, i.p.) was administered to mice once a day during CSR. Our results further showed that GM1 activated CD33 and consequently disturbed TREM2-mediated microglial phagocytosis. Finally, GM1 reversed CSR-induced synaptic loss and memory impairment via the CD33/TREM2 signaling pathway in the CA1 region of the hippocampus. This study provides novel evidence that activating CD33 and/or inhibiting TREM2 activity represent potential therapies for sleep loss-induced memory deficits through the modulation of microglial phagocytosis.






Similar content being viewed by others
Data Availability
The data that support the funding of this study are available from the corresponding author upon reasonable request.
Abbreviations
- AD:
-
Alzheimer’s disease
- AZ:
-
Active zone
- CD33:
-
Siglec-3
- CFH:
-
Regulatory complement factor H
- CON:
-
Control
- CSR:
-
Chronic sleep restriction
- DAM:
-
Disease-associated microglia
- GM1:
-
Monosialo-ganglioside 1
- NOR:
-
Novel objective recognition
- PSD95:
-
Postsynaptic protein 95
- ROI:
-
Polygonal region of interest
- SC:
-
Synaptic cleft
- SHP-1/2:
-
Tyrosine phosphatase-1/2
- siRNA:
-
Small interfering RNA
- Syk:
-
Spleen tyrosine kinase
- SYP:
-
Synaptophysin
- TREM2:
-
Triggering receptor expressed on myeloid cells 2
References
Arganda-Carreras, I., Fernández-González, R., Muñoz-Barrutia, A., & Ortiz-De-Solorzano, C. (2010). 3D reconstruction of histological sections: Application to mammary gland tissue. Microscopy Research and Technique, 73(11), 1019–1029. https://doi.org/10.1002/jemt.20829
Bandyopadhyay, A., & Sigua, N. L. (2019). What is sleep deprivation? American Journal of Respiratory and Critical Care Medicine, 199(6), P11–P12. https://doi.org/10.1164/rccm.1996p11
Bellesi, M., de Vivo, L., Chini, M., Gilli, F., Tononi, G., & Cirelli, C. (2017). Sleep Loss Promotes Astrocytic Phagocytosis and Microglial Activation in Mouse Cerebral Cortex. Journal of Neuroscience, 37(21), 5263–5273. https://doi.org/10.1523/JNEUROSCI.3981-16.2017
Bellesi, M., de Vivo, L., Tononi, G., & Cirelli, C. (2015). Effects of sleep and wake on astrocytes: Clues from molecular and ultrastructural studies. BMC Biology, 13, 66. https://doi.org/10.1186/s12915-015-0176-7
Bhattacherjee, A., Rodrigues, E., Jung, J., Luzentales-Simpson, M., Enterina, J. R., Galleguillos, D., St Laurent, C. D., Nakhaei-Nejad, M., Fuchsberger, F. F., Streith, L., Wang, Q., Kawasaki, N., Duan, S., Bains, A., Paulson, J. C., Rademacher, C., Giuliani, F., Sipione, S., & Macauley, M. S. (2019). Repression of phagocytosis by human CD33 is not conserved with mouse CD33. Communications Biology, 2, 450. https://doi.org/10.1038/s42003-019-0698-6
Bolsius, Y. G., Meerlo, P., Kas, M. J., Abel, T., & Havekes, R. (2021). Sleep deprivation reduces the density of individual spine subtypes in a branch-specific fashion in CA1 neurons. Journal of Sleep Research, 31(1), e13438. https://doi.org/10.1111/jsr.13438
Cai, X., Li, Y., Zheng, X., Hu, R., Li, Y., Xiao, L., & Wang, Z. (2021). Propofol suppresses microglial phagocytosis through the downregulation of MFG-E8. Journal of Neuroinflammation, 18(1), 18. https://doi.org/10.1186/s12974-020-02061-3
Claude, J., Linnartz-Gerlach, B., Kudin, A. P., Kunz, W. S., & Neumann, H. (2013). Microglial CD33-related Siglec-E inhibits neurotoxicity by preventing the phagocytosis-associated oxidative burst. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience, 33(46), 18270–18276. https://doi.org/10.1523/JNEUROSCI.2211-13.2013
Clinton, J. M., Davis, C. J., Zielinski, M. R., Jewett, K. A., & Krueger, J. M. (2011). Biochemical regulation of sleep and sleep biomarkers. Journal of Clinical Sleep Medicine : JCSM : Official Publication of the American Academy of Sleep Medicine, 7(5 Suppl), S38–S42. https://doi.org/10.5664/JCSM.1360
Delpech, J.-C., Wei, L., Hao, J., Yu, X., Madore, C., Butovsky, O., & Kaffman, A. (2016). Early life stress perturbs the maturation of microglia in the developing hippocampus. Brain, Behavior, and Immunity, 57, 79–93. https://doi.org/10.1016/j.bbi.2016.06.006
Fernandes, C., Rocha, N. B. F., Rocha, S., Herrera-Solís, A., Salas-Pacheco, J., García-García, F., Murillo-Rodríguez, E., Yuan, T.-F., Machado, S., & Arias-Carrión, O. (2015). Detrimental role of prolonged sleep deprivation on adult neurogenesis. Frontiers in Cellular Neuroscience, 9, 140. https://doi.org/10.3389/fncel.2015.00140
Filipello, F., Morini, R., Corradini, I., Zerbi, V., Canzi, A., Michalski, B., Erreni, M., Markicevic, M., Starvaggi-Cucuzza, C., Otero, K., Piccio, L., Cignarella, F., Perrucci, F., Tamborini, M., Genua, M., Rajendran, L., Menna, E., Vetrano, S., Fahnestock, M., … Matteoli, M. (2018). The microglial innate immune receptor TREM2 is required for synapse elimination and normal brain connectivity. Immunity, 48(5), 979–991.e978 https://doi.org/10.1016/j.immuni.2018.04.016
Fricker, M., Neher, J. J., Zhao, J.-W., Théry, C., Tolkovsky, A. M., & Brown, G. C. (2012). MFG-E8 mediates primary phagocytosis of viable neurons during neuroinflammation. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience, 32(8), 2657–2666. https://doi.org/10.1523/JNEUROSCI.4837-11.2012
Griciuc, A., Federico, A. N., Natasan, J., Forte, A. M., McGinty, D., Nguyen, H., Volak, A., LeRoy, S., Gandhi, S., Lerner, E. P., Hudry, E., Tanzi, R. E., & Maguire, C. A. (2020). Gene therapy for Alzheimer’s disease targeting CD33 reduces amyloid beta accumulation and neuroinflammation. Hum Mol Genet, 29(17), 2920–2935. https://doi.org/10.1093/hmg/ddaa179
Griciuc, A., Patel, S., Federico, A., Choi, S., Innes, B., Oram, M., Cereghetti, G., McGinty, D., Anselmo, A., Sadreyev, R. I., Hickman, S. E., El Khoury, J., Colonna, M., & Tanzi, R. E. (2019). TREM2 acts downstream of CD33 in modulating microglial pathology in Alzheimer’s disease. Neuron, 103(5), 820–835.e827. https://doi.org/10.1016/j.neuron.2019.06.010
Griciuc, A., Serrano-Pozo, A., Parrado, A. R., Lesinski, A. N., Asselin, C. N., Mullin, K., Hooli, B., Choi, S. H., Hyman, B. T., & Tanzi, R. E. (2013). Alzheimer’s disease risk gene CD33 inhibits microglial uptake of amyloid beta. Neuron, 78(4), 631–643. https://doi.org/10.1016/j.neuron.2013.04.014
Havekes, R., Park, A. J., Tudor, J. C., Luczak, V. G., Hansen, R. T., Ferri, S. L., Bruinenberg, V. M., Poplawski, S. G., Day, J. P., Aton, S. J., Radwańska, K., Meerlo, P., Houslay, M. D., Baillie, G. S., & Abel, T. (2016). Sleep deprivation causes memory deficits by negatively impacting neuronal connectivity in hippocampal area CA1. eLife, 5, e13424. https://doi.org/10.7554/eLife.13424
Hughes, A., & Appel, B. (2020). Microglia phagocytose myelin sheaths to modify developmental myelination. Nature Neuroscience, 23(9), 1055–1066. https://doi.org/10.1038/s41593-020-0654-2
Karch, C., & Goate, A. (2015). Alzheimer’s disease risk genes and mechanisms of disease pathogenesis. Biological Psychiatry, 77(1), 43–51. https://doi.org/10.1016/j.biopsych.2014.05.006
Kraeuter, A.-K., Guest, P. C., & Sarnyai, Z. (2019). The Y-maze for assessment of spatial working and reference memory in mice. Methods in Molecular Biology (Clifton, N.J.), 1916, 105–111. https://doi.org/10.1007/978-1-4939-8994-2_10
Lian, H., Roy, E., & Zheng, H. (2016). Microglial phagocytosis assay. Bio-Protocol, 6(21), e1988. https://doi.org/10.1007/978-1-4939-8994-2_10
Liao, H., Klaus, C., & Neumann, H. (2020). Control of innate immunity by sialic acids in the nervous tissue. International Journal of Molecular Sciences, 21(15), 5494. https://doi.org/10.3390/ijms21155494
Lueptow, L. M. (2017). Novel object recognition test for the investigation of learning and memory in mice. Journal of Visualized Experiments (JoVE), 126, 55718. https://doi.org/10.3791/55718
Maquet, P. (2001). The role of sleep in learning and memory. Science (New York, N.Y.), 294(5544), 1048–1052. https://doi.org/10.1126/science.1062856
Maret, S., Faraguna, U., Nelson, A. B., Cirelli, C., & Tononi, G. (2011). Sleep and waking modulate spine turnover in the adolescent mouse cortex. Nature Neuroscience, 14(11), 1418–1420. https://doi.org/10.1038/nn.2934
Min, K.-J., Pyo, H.-K., Yang, M.-S., Ji, K.-A., Jou, I., & Joe, E.-H. (2004). Gangliosides activate microglia via protein kinase C and NADPH oxidase. Glia, 48(3), 197–206. https://doi.org/10.1002/glia.20069
Myers, D. R., Abram, C. L., Wildes, D., Belwafa, A., Welsh, A. M. N., Schulze, C. J., Choy, T. J., Nguyen, T., Omaque, N., Hu, Y., Singh, M., Hansen, R., Goldsmith, M. A., Quintana, E., Smith, J. A. M., & Lowell, C. A. (2020). Shp1 loss enhances macrophage effector function and promotes anti-tumor immunity. Frontiers In Immunology, 11, 576310. https://doi.org/10.3389/fimmu.2020.576310
Neher, J. J., Neniskyte, U., Zhao, J.-W., Bal-Price, A., Tolkovsky, A. M., & Brown, G. C. (2011). Inhibition of microglial phagocytosis is sufficient to prevent inflammatory neuronal death. Journal of Immunology (Baltimore, M.d.), 1950, 186(8), 4973–4983. https://doi.org/10.4049/jimmunol.1003600
Neniskyte, U., Neher, J. J., & Brown, G. C. (2011). Neuronal death induced by nanomolar amyloid β is mediated by primary phagocytosis of neurons by microglia. The Journal of Biological Chemistry, 286(46), 39904–39913. https://doi.org/10.1074/jbc.M111.267583
Paolicelli, R., Bolasco, G., Pagani, F., Maggi, L., Scianni, M., Panzanelli, P., Giustetto, M., Ferreira, T. A., Guiducci, E., Dumas, L., Ragozzino, D., & Gross, C. T. (2011). Synaptic pruning by microglia is necessary for normal brain development. Science (New York, N.Y.), 333(6048), 1456–1458. https://doi.org/10.1126/science.1202529
Poliani, P. L., Wang, Y., Fontana, E., Robinette, M. L., Yamanishi, Y., Gilfillan, S., & Colonna, M. (2015). TREM2 sustains microglial expansion during aging and response to demyelination. The Journal of Clinical Investigation, 125(5), 2161–2170. https://doi.org/10.1172/JCI77983
Qu, W., & Li, L. (2020). Loss of TREM2 confers resilience to synaptic and cognitive impairment in aged mice. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience, 40(50), 9552–9563. https://doi.org/10.1523/JNEUROSCI.2193-20.2020
Ramesh, V., Nair, D., Zhang, S. X. L., Hakim, F., Kaushal, N., Kayali, F., Wang, Y., Li, R. C., Carreras, A., & Gozal, D. (2012). Disrupted sleep without sleep curtailment induces sleepiness and cognitive dysfunction via the tumor necrosis factor-α pathway. Journal of Neuroinflammation, 9, 91. https://doi.org/10.1186/1742-2094-9-91
Ramesha, S., Rayaprolu, S., & Rangaraju, S. (2020). Flow cytometry approach to characterize phagocytic properties of acutely-isolated adult microglia and brain macrophages in vitro. Journal of visualized experiments (JoVE). https://doi.org/10.3791/61467
Rangaraju, S., Dammer, E. B., Raza, S. A., Rathakrishnan, P., Xiao, H., Gao, T., Duong, D. M., Pennington, M. W., Lah, J. J., Seyfried, N. T., & Levey, A. I. (2018). Identification and therapeutic modulation of a pro-inflammatory subset of disease-associated-microglia in Alzheimer’s disease. Molecular Neurodegeneration, 13(1), 24. https://doi.org/10.1186/s13024-018-0254-8
Ransohoff, R. M., & Perry, V. H. (2009). Microglial physiology: Unique stimuli, specialized responses. Annual Review of Immunology, 27, 119–145. https://doi.org/10.1146/annurev.immunol.021908.132528
Raven, F., Van der Zee, E. A., Meerlo, P., & Havekes, R. (2018). The role of sleep in regulating structural plasticity and synaptic strength: Implications for memory and cognitive function. Sleep Medicine Reviews, 39, 3–11. https://doi.org/10.1016/j.smrv.2017.05.002
Salminen, A., Kaarniranta, K., & Kauppinen, A. (2021). Hypoxia/ischemia impairs CD33 (Siglec-3)/TREM2 signaling: Potential role in Alzheimer’s pathogenesis. Neurochemistry International, 150, 105186. https://doi.org/10.1016/j.neuint.2021.105186
Schafer, D. P., Lehrman, E. K., Kautzman, A. G., Koyama, R., Mardinly, A. R., Yamasaki, R., Ransohoff, R. M., Greenberg, M. E., Barres, B. A., & Stevens, B. (2012). Microglia sculpt postnatal neural circuits in an activity and complement-dependent manner. Neuron, 74(4), 691–705. https://doi.org/10.1016/j.neuron.2012.03.026
Schmidt, C. Q., HipgraveEderveen, A. L., Harder, M. J., Wuhrer, M., Stehle, T., & Blaum, B. S. (2018). Biophysical analysis of sialic acid recognition by the complement regulator Factor H. Glycobiology, 28(10), 765–773. https://doi.org/10.1093/glycob/cwy061
Schneider, J. S., Aras, R., Williams, C. K., Koprich, J. B., Brotchie, J. M., & Singh, V. (2019). GM1 ganglioside modifies alpha-synuclein toxicity and is neuroprotective in a rat alpha-synuclein model of Parkinson’s disease. Science and Reports, 9(1), 8362. https://doi.org/10.1038/s41598-019-42847-x
Spano, G. M., Banningh, S. W., Marshall, W., de Vivo, L., Bellesi, M., Loschky, S. S., Tononi, G., & Cirelli, G. (2019). Sleep deprivation by exposure to novel objects increases synapse density and axon-spine interface in the hippocampal CA1 region of adolescent mice. The Journal of Neuroscience: The Official Journal of the Society for Neuroscience, 39(34), 6613–6625. https://doi.org/10.1523/JNEUROSCI.0380-19.2019
Takahashi, K., Rochford, C. D. P., & Neumann, H. (2005). Clearance of apoptotic neurons without inflammation by microglial triggering receptor expressed on myeloid cells-2. The Journal of Experimental Medicine, 201(4), 647–657. https://doi.org/10.1084/jem.20041611
Tapp, Z. M., Kumar, J. E., Witcher, K. G., Atluri, R. R., Velasquez, J. A., O’Neil, S. M., Dziabis, J. E., Bray, C. E., Sheridan, J. F., Godbout, J. P., & Kokiko-Cochran, O. N. (2020). Sleep disruption exacerbates and prolongs the inflammatory response to traumatic brain injury. Journal of Neurotrauma, 37(16), 1829–1843. https://doi.org/10.1089/neu.2020.7010
Terry, R., Masliah, E., Salmon, D., Butters, N., DeTeresa, R., Hill, R., Hansen, L. A., & Katzman, R. (1991). Physical basis of cognitive alterations in Alzheimer’s disease: Synapse loss is the major correlate of cognitive impairment. Annals of Neurology, 30(4), 572–580. https://doi.org/10.1002/ana.410300410
Tononi, G., & Cirelli, C. (2014). Sleep and the price of plasticity: From synaptic and cellular homeostasis to memory consolidation and integration. Neuron, 81(1), 12–34. https://doi.org/10.1016/j.neuron.2013.12.025
Tremblay, M. -È., Lowery, R. L., & Majewska, A. K. (2010). Microglial interactions with synapses are modulated by visual experience. PLoS Biology, 8(11), e1000527. https://doi.org/10.1371/journal.pbio.1000527
Tuan, L. H., & Lee, L. J. (2019). Microglia-mediated synaptic pruning is impaired in sleep-deprived adolescent mice. Neurobiology of Disease, 130, 104517. https://doi.org/10.1016/j.nbd.2019.104517
Tynan, R. J., Naicker, S., Hinwood, M., Nalivaiko, E., Buller, K. M., Pow, D. V., Day, T. A., & Walker, F. R. (2010). Chronic stress alters the density and morphology of microglia in a subset of stress-responsive brain regions. Brain, Behavior, and Immunity, 24(7), 1058–1068. https://doi.org/10.1016/j.bbi.2010.02.001
Wadhwa, M., Prabhakar, A., Ray, K., Roy, K., Kumari, P., Jha, P. K., Kishore, K., Kumar, S., & Panjwani, U. (2010). Inhibiting the microglia activation improves the spatial memory and adult neurogenesis in rat hippocampus during 48 h of sleep deprivation. Journal of Neuroinflammation, 14(1), 222. https://doi.org/10.1186/s12974-017-0998-z
Wang, B. (2009). Sialic acid is an essential nutrient for brain development and cognition. Annual Review of Nutrition, 29, 177–222. https://doi.org/10.1146/annurev.nutr.28.061807.155515
Wheaton, A. G., & Claussen, A. H. (2021). Short sleep duration among infants, children, and adolescents aged 4 months–17 years—United States, 2016–2018. MMWR. Morbidity and Mortality Weekly Report, 70(38), 1315–1321. https://doi.org/10.15585/mmwr.mm7038a1
Yang, G., & Gan, W.-B. (2012). Sleep contributes to dendritic spine formation and elimination in the developing mouse somatosensory cortex. Developmental Neurobiology, 72(11), 1391–1398. https://doi.org/10.1002/dneu.20996
Yanguas-Casás, N., Crespo-Castrillo, A., Arevalo, M., & Garcia-Segura, L. (2020). Aging and sex: Impact on microglia phagocytosis. Aging Cell, 19(8), e13182. https://doi.org/10.1111/acel.13182
Zhai, Q., Li, F., Chen, X., Jia, J., Sun, S., Zhou, D., Ma, L., Jiang, T., Bai, F., Xiong, L., & Wang, Q. (2017). Triggering receptor expressed on myeloid cells 2, a novel regulator of immunocyte phenotypes. Confers Neuroprotection by Relieving Neuroinflammation. Anesthesiology, 127(1), 98–110. https://doi.org/10.1097/aln.0000000000001628
Zhang, K., Lian, N., Ding, R., Guo, C., Dong, X., Li, Y., Wei, L., Jiao, Q., Yu, Y., & Shen, H. (2020). Sleep deprivation aggravates cognitive impairment by the alteration of hippocampal neuronal activity and the density of dendritic spine in isoflurane-exposed mice. Frontiers in Behavioral Neuroscience, 14, 589176. https://doi.org/10.3389/fnbeh.2020.589176
Zhao, B., Liu, P., Wei, M., Li, Y., Liu, J., Ma, L., Shang, S., Jiang, Y., Huo, K., Wang, J., & Qu, Q. (2019). Chronic sleep restriction induces Aβ accumulation by disrupting the balance of Aβ production and clearance in rats. Neurochemical Research, 44(4), 859–873. https://doi.org/10.1007/s11064-019-02719-2
Acknowledgements
We would also like to acknowledge the support of Yan Li and Ying Zhang on this project who shared equipment and experimental techniques.
Funding
This work was supported by funds from the National Natural Science Foundation of China (Grant Nos. 81901237, 81774113, 81974540), Beijing, China.
Author information
Authors and Affiliations
Contributions
ST, QW, and QZ conceived and designed this study. ST, HG, and JS performed the experiments, and ST and QZ drafted the manuscript. NL and YZ conducted data analysis, and LY and MW helped to revise the manuscript. And all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that there are no conflict of interest.
Consent to Participate
Not applicable.
Consent for Publication
Not applicable.
Consent to Animal
All experiments were carried out by institutional guidelines and approved by the Animal Care and Use Committee of Xi’an Jiaotong University, Xi’an, Shaanxi Province, China.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tan, S., Gao, H., Sun, J. et al. CD33/TREM2 Signaling Mediates Sleep Deprivation-Induced Memory Impairment by Regulating Microglial Phagocytosis. Neuromol Med 25, 255–271 (2023). https://doi.org/10.1007/s12017-023-08733-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-023-08733-6