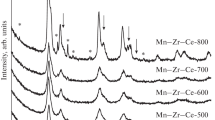
NiO-Al2O3 and Co3O4 oxide catalysts modified with palladium and oxides of rare earth metals (Ce and La) and formed on structured cordierite supports have been studied in the process of methane oxidation at different O2/CH4 ratios. It is shown that methane oxidation proceeds without the formation of CO even at an O2/CH4 ratio of < 2 (in contrast to nickel-containing catalysts) in the presence of cobalt-containing catalysts modified with cerium and lanthanum oxides. The addition of CeO2 promotes Co3O4 dispersion, increases the number of oxygen vacancies, and prevents agglomeration of the active phase of the catalyst at high temperatures. The modification of Co3O4-CeO2/cordierite with palladium (0.1%) enhances its activity due to increased surface oxygen mobility as a result of the interphase interaction of palladium with cobalt oxides and ceria (according to the TPR-H2 and XPS data).


Similar content being viewed by others
References
L. He, Y. Fan, J. Bellettre, J. Yue, and L. Luo, Renew. Sustain. Energy Rev., 119, 109589 (2020).
J. Chen, H. Arandiyan, X. Gao, and J. Li, Catal. Surv. Asia, 19, No. 3, 140-171 (2015).
K. Ghaib, and F. Z. Ben-Fares, Renew. Sustain. Energy Rev., 81, 433-446 (2018).
T. V. Choudhary, S. Banerjee, and V. R. Choudhary, Appl. Catal. A., 234, Nos. 1-2, 1-23 (2002).
F. Liu, Y. Sang, H. Ma, Z. Li, and Z. Gao, J. Nat. Gas Sci. Eng., 41, 1-6 (2017).
A. Boreave, H. Tan, V. Roche, et al., Solid State Ionics, 179, Nos. 21-26, 1071-1075 (2008).
X. Yang, C. Du, Guo Y., et al., J. Rare Earths, 37, No. 7, 714-719 (2019).
N. Sharma, S. Bhatnagar, and S. Jain, Eng. J. Appl. Scopes, 1, No. 2, 70-74 (2016).
S. O. Soloviev, P. I. Kyriienko, and N. O. Popovych, J. Environ. Sci., 24, No. 7, 1327-1333 (2012).
J. Chastain (ed.), Handbook îf X-ray Photoelectron Spectroscopy, Perkin-Elmer Corporation (1992).
N. Mouaddib, C. Feumi-Jantou, E. Garbowski, and M. Primet, Appl. Catal. A., 87, No. 1, 129-144 (1992).
G. Pecchi, P. Reyes, T. Lopez, and R. Gomez, J. Non-Cryst. Solids, 345, 624-627 (2004).
R. J. Farrauto, M. Hobson, T. Kennelly, and E. Waterman, Appl. Catal. A., 81, No. 2, 227-237 (1992).
N. Tsud, V. Johanek, I. Stara, et al., Surf. Sci., 467, Nos. 1-3, 169-176 (2000).
J. Li, J. Zhang , Z. Lei, and B. Chen, Energy Fuels, 26, No. 1, 443-450 (2012).
S. M. Orlyk, S. O. Soloviev, A. Yu. Kapran, et al., Cerium: Chemical Properties, Applications, and Environmental Impact, N. Sherata (ed.), Nova Science Publishers, New York (2017), pp. 43-100.
L. F. Liotta, G. Di Carlo, A. Longo, et al., Catal. Today, 139, No. 3, 174-179 (2008).
G. Zo, Y. Xu, S. Wang, M. Chen, et al., Catal. Sci. Technol., 5, No. 2, 1084-1092 (2015).
X. D. Hou, Y. Zh. Wang, and Y. X. Zhao, Catal. Lett., 123, No. 3, 321-326 (2008).
P. Euzen, J.-H. Le Gal, B. Rebours, and G. Martin, Catal. Today, 47, Nos. 1-4, 19-27 (1999).
K. Persson, A. Ersson, K. Jansson, et al., J. Catal., 231, No. 1, 139-150 (2005).
P. Stefanov, S. Todorova, A. Naydenov, et al., Chem. Eng. J., 266, 329-338 (2015).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Teoretychna ta Eksperymentalna Khimiya, Vol. 58, No. 5, pp. 307-313, September-October, 2022.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Soloviev, S.O., Kosmambetova, G.R., Kyriienko, P.I. et al. Influence of Pd and Rare Earth Metals Oxides (Ce, La) as Modifying Additives in the Co,Ni-Oxide Catalyst Compositions on the Process of Methane Oxidation. Theor Exp Chem 58, 342–349 (2022). https://doi.org/10.1007/s11237-023-09750-z
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11237-023-09750-z