Abstract
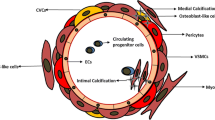
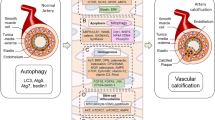
Vascular calcification is an abnormal process in which bone specific hydroxyapatite crystals are actively deposited on the vascular wall mediated by phenotypic differentiated smooth muscle cells and other mesenchymal cells under various pathological conditions. It is one of the important characteristics in the occurrence and development of atherosclerosis, prevalent in patients with type 2 diabetes and advanced chronic kidney disease, especially those requiring maintenance hemodialysis, with severely threatening human health. Previous studies have shown that the early diagnosis and control of vascular calcification is of great significance for cardiovascular risk stratification, prevention of acute cardiovascular events, which can greatly improve the prognosis and quality of life of patients. Galectins are a family of lectin superfamily. It is widely distributed in various animals and plays an important role in many physiological and pathological processes, such as cell adhesion, apoptosis, inflammatory response, tumor metastasis and so on. Many biomarker-and association-related studies and Preclinical-mechanistic studies have suggested that galactose-specific lectin-3 (galectin-3) plays an important role in vascular calcification and vascular intimal calcification (VIC) calcification induced by Wnt/βcatenin signaling pathway, NF-κB signaling pathway and ERK1/2 signaling pathway. This paper mainly expounds the role and mechanism of galectin-3 in vascular calcification under different pathological conditions including atherosclerosis, diabetes and chronic kidney disease.

Similar content being viewed by others
Abbreviations
- AAC:
-
Abdominal aortic calcification
- AVs:
-
Aortic valves
- BMC:
-
Bone marrow cell
- CAD:
-
Coronary artery disease
- CCS:
-
Chronic coronary syndrome
- CTD:
-
Carboxyl terminal domain
- ECM:
-
Extracellular matrix
- ESRD:
-
End-stage renal disease
- EVs:
-
Extracellular vesicles
- GLDs:
-
Glucose and lipid metabolism disorders
- MCP:
-
Modified citrus pectin
- MHD:
-
Maintenance hemodialysis
- MMP:
-
Matrix metalloproteinase
- ND:
-
N-terminal domain
- TXNIP:
-
Thioredoxin-interacting protein
- VIC:
-
Vascular intimal calcification
- VICS:
-
Valve stromal cells
- VMC:
-
Vascular medial calcification
- WT:
-
Wild-type
References
Lee, S.J., Lee, I.K., Jeon, J.H.: Vascular calcification-new insights into its mechanism. Int. J. Mol. Sci. 21(8) (2020)
Ibarrola, J., Martínez-Martínez, E., Sádaba, J.R., Arrieta, V., García-Peña, A., Álvarez, V., et al.: Beneficial effects of galectin-3 blockade in vascular and aortic valve alterations in an experimental pressure overload model. Int. J. Mol. Sci. 18(8) (2017)
Ozturk, D., Celik, O., Satilmis, S., Aslan, S., Erturk, M., Cakmak, H.A., et al.: Association between serum galectin-3 levels and coronary atherosclerosis and plaque burden/structure in patients with type 2 diabetes mellitus. Coron. Artery Dis. 26(5), 396–401 (2015)
Sádaba, J.R., Martínez-Martínez, E., Arrieta, V., Álvarez, V., Fernández-Celis, A., Ibarrola, J., et al.: Role for galectin-3 in calcific aortic valve stenosis. J. Am. Heart Assoc. 5(11) (2016)
Menini, S., Iacobini, C., Ricci, C., Blasetti Fantauzzi, C., Salvi, L., Pesce, C.M., et al.: The galectin-3/RAGE dyad modulates vascular osteogenesis in atherosclerosis. Cardiovasc. Res. 100(3), 472–480 (2013)
Luo, J., Wang, S., Liu, X., Zheng, Q., Wang, Z., Huang, Y., et al.: Galectin-3 promotes calcification of human aortic valve interstitial cells via the NF-kappa B signaling pathway. Cardiovasc. Diagn. Ther. 12(2), 196–207 (2022)
Ho, M.K.: Springer TA. Mac-2, a novel 32,000 Mr mouse macrophage subpopulation-specific antigen defined by monoclonal antibodies. J. Immunol. (Baltimore, Md : 1950). 128(3), 1221–8 (1982)
Cowles, E.A., Agrwal, N., Anderson, R.L., Wang, J.L.: Carbohydrate-binding protein 35. Isoelectric points of the polypeptide and a phosphorylated derivative. J. Biol. Chem. 265(29), 17706–12 (1990)
Dong, R., Zhang, M., Hu, Q., Zheng, S., Soh, A., Zheng, Y., et al.: Galectin-3 as a novel biomarker for disease diagnosis and a target for therapy (Review). Int. J. Mol. Med. 41(2), 599–614 (2018)
Pugliese, G., Iacobini, C., Ricci, C., Blasetti Fantauzzi, C., Menini, S.: Galectin-3 in diabetic patients. Clin. Chem. Lab. Med. 52(10), 1413–1423 (2014)
Newlaczyl, A.U., Yu, L.G.: Galectin-3–a jack-of-all-trades in cancer. Cancer Lett. 313(2), 123–128 (2011)
Cano-Megías, M., Bouarich, H., Guisado-Vasco, P., Pérez Fernández, M., de Arriba-de la Fuente, G., Álvarez-Sanz, C., et al.: Coronary artery calcification in patients with diabetes mellitus and advanced chronic kidney disease. Endocrinol. Diabetes Nutr. 66(5), 297–304 (2019)
Yahagi, K., Kolodgie, F.D., Lutter, C., Mori, H., Romero, M.E., Finn, A.V., et al.: Pathology of human coronary and carotid artery atherosclerosis and vascular calcification in diabetes mellitus. Arterioscler. Thromb. Vasc. Biol. 37(2), 191–204 (2017)
Zhang, L., Sun, H., Liu, S., Gao, J., Xia, J.: Glycemic variability is associated with vascular calcification by the markers of endoplasmic reticulum stress-related apoptosis, Wnt1, galectin-3 and BMP-2. Diabetol. Metab. Syndr. 11, 67 (2019)
Disthabanchong, S., Srisuwarn, P.: Mechanisms of vascular calcification in kidney disease. Adv. Chronic Kidney Dis. 26(6), 417–426 (2019)
Wu, M., Rementer, C., Giachelli, C.M.: Vascular calcification: an update on mechanisms and challenges in treatment. Calcif. Tissue Int. 93(4), 365–373 (2013)
Sun, Z., Zhang, L., Li, L., Shao, C., Liu, J., Zhou, M., et al.: Galectin-3 mediates cardiac remodeling caused by impaired glucose and lipid metabolism through inhibiting two pathways of activating Akt. Am. J. Physiol. Heart Circ. Physiol. 320(1), H364–H380 (2021)
Langley, S.R., Willeit, K., Didangelos, A., Matic, L.P., Skroblin, P., Barallobre-Barreiro, J., et al.: Extracellular matrix proteomics identifies molecular signature of symptomatic carotid plaques. J. Clin. Investig. 127(4), 1546–1560 (2017)
Papaspyridonos, M., McNeill, E., de Bono, J.P., Smith, A., Burnand, K.G., Channon, K.M., et al.: Galectin-3 is an amplifier of inflammation in atherosclerotic plaque progression through macrophage activation and monocyte chemoattraction. Arterioscler. Thromb. Vasc. Biol. 28(3), 433–440 (2008)
Nachtigal, M., Al-Assaad, Z., Mayer, E.P., Kim, K., Monsigny, M.: Galectin-3 expression in human atherosclerotic lesions. Am. J. Pathol. 152(5), 1199–1208 (1998)
Iacobini, C., Blasetti Fantauzzi, C., Bedini, R., Pecci, R., Bartolazzi, A., Amadio, B., et al.: Galectin-3 is essential for proper bone cell differentiation and activity, bone remodeling and biomechanical competence in mice. Metab. Clin. Exp. 83, 149–58 (2018)
Lisowska, A., Knapp, M., Tycińska, A., Motybel, E., Kamiński, K., Święcki, P., et al.: Predictive value of Galectin-3 for the occurrence of coronary artery disease and prognosis after myocardial infarction and its association with carotid IMT values in these patients: A mid-term prospective cohort study. Atherosclerosis 246, 309–317 (2016)
Falcone, C., Lucibello, S., Mazzucchelli, I., Bozzini, S., D’Angelo, A., Schirinzi, S., et al.: Galectin-3 plasma levels and coronary artery disease: a new possible biomarker of acute coronary syndrome. Int. J. Immunopathol. Pharmacol. 24(4), 905–913 (2011)
Sun, Z., Wang, Z., Li, L., Yan, J., Shao, C., Bao, Z., et al.: RAGE/galectin-3 yields intraplaque calcification transformation via sortilin. Acta Diabetol. 56(4), 457–472 (2019)
Kadoglou, N.P., Sfyroeras, G.S., Spathis, A., Gkekas, C., Gastounioti, A., Mantas, G., et al.: Galectin-3, carotid plaque vulnerability, and potential effects of statin therapy. Eur. J. Vasc. Endovasc. Surg. 49(1), 4–9 (2015)
Jing, L., Li, L., Ren, X., Sun, Z., Bao, Z., Yuan, G., et al.: Role of sortilin and matrix vesicles in Nε-Carboxymethyl-Lysine-Induced diabetic atherosclerotic calcification. Diabet. Metab. Synd. Ob. 13, 4141–4151 (2020)
Mori, H., Torii, S., Kutyna, M., Sakamoto, A., Finn, A.V., Virmani, R.: Coronary artery calcification and its progression: What does it really mean? JACC Cardiovasc. Imaging 11(1), 127–142 (2018)
Tian, L., Wang, Y., Zhang, R.: Galectin-3 induces vascular smooth muscle cells calcification via AMPK/TXNIP pathway. Aging 14(12), 5086–5096 (2022)
Kalantar-Zadeh, K., Jafar, T.H., Nitsch, D., Neuen, B.L., Perkovic, V.: Chronic kidney disease. Lancet (London, England). 398(10302), 786–802 (2021)
Desmedt, V., Desmedt, S., Delanghe, J.R., Speeckaert, R., Speeckaert, M.M.: Galectin-3 in renal pathology: More than just an innocent bystander. Am. J. Nephrol. 43(5), 305–317 (2016)
Boutin, L., Depret, F., Gayat, E., Legrand, M., Chadjichristos, C.E.: Galectin-3 in kidney diseases: from an old protein to a new therapeutic target. Int. J. Mol. Sci. 23(6) (2022)
Chen, T.K., Knicely, D.H., Grams, M.E.: Chronic kidney disease diagnosis and management: A review. JAMA 322(13), 1294–1304 (2019)
Drechsler, C., Delgado, G., Wanner, C., Blouin, K., Pilz, S., Tomaschitz, A., et al.: Galectin-3, renal function, and clinical outcomes: Results from the LURIC and 4D Studies. J. Am. Soc. Nephrol. 26(9), 2213–2221 (2015)
Wang, Z., Chen, Z., Ma, X., Yu, H., Chen, X.: The predictive value of serum galectin 3 for abdominal aortic calcification in maintenance hemodialysis patients: A prospective cohort study. Hemodialysis international International Symposium on Home Hemodialysis. 24(2), 212–220 (2020)
Zhang, Q., Yin, K., Ni, Z.: Galectin-3 and abdominal aortic calcification in patients on hemodialysis. Vascular medicine (London, England). 25(6), 575–576 (2020)
Tumlin, J.A., Costanzo, M.R., Chawla, L.S., Herzog, C.A., Kellum, J.A., McCullough, P.A., et al.: Cardiorenal syndrome type 4: insights on clinical presentation and pathophysiology from the eleventh consensus conference of the Acute Dialysis Quality Initiative (ADQI). Contrib. Nephrol. 182, 158–173 (2013)
Iseri, K., Dai, L., Chen, Z., Qureshi, A.R., Brismar, T.B., Stenvinkel, P., et al.: Bone mineral density and mortality in end-stage renal disease patients. Clin. Kidney J. 13(3), 307–321 (2020)
Lanzer, P., Boehm, M., Sorribas, V., Thiriet, M., Janzen, J., Zeller, T., et al.: Medial vascular calcification revisited: review and perspectives. Eur. Heart J. 35(23), 1515–1525 (2014)
Wang, Z., Jiang, Y., Liu, N., Ren, L., Zhu, Y., An, Y., et al.: Advanced glycation end-product Nε-carboxymethyl-Lysine accelerates progression of atherosclerotic calcification in diabetes. Atherosclerosis 221(2), 387–396 (2012)
Gu, W., Wang, Z., Sun, Z., Bao, Z., Zhang, L., Geng, Y., et al.: Role of NFATc1 in the bone-vascular axis calcification paradox. J. Cardiovasc. Pharmacol. 75(3), 200–207 (2020)
Aksan, G., Gedikli, Ö., Keskin, K., Nar, G., İnci, S., Yıldız, S.S., et al.: Is galectin-3 a biomarker, a player-or both-in the presence of coronary atherosclerosis? Journal of investigative medicine : the official publication of the American Federation for Clinical Research. 64(3), 764–770 (2016)
MacKinnon, A.C., Liu, X., Hadoke, P.W., Miller, M.R., Newby, D.E., Sethi, T.: Inhibition of galectin-3 reduces atherosclerosis in apolipoprotein E-deficient mice. Glycobiology 23(6), 654–663 (2013)
Święcki, P., Sawicki, R., Knapp, M., Kamiński, K.A., Ptaszyńska-Kopczyńska, K., Sobkowicz, B., et al.: Galectin-3 as the prognostic factor of adverse cardiovascular events in long-term follow up in patients after myocardial infarction-a pilot study. J. Clin. Med. 9(6) (2020)
Martín-Reyes, R., Franco-Peláez, J.A., Lorenzo, Ó., González-Casaus, M.L., Pello, A.M., Aceña, Á., et al.: Plasma levels of monocyte chemoattractant protein-1, n-Terminal Fragment of brain natriuretic peptide and calcidiol are independently associated with the complexity of coronary artery disease. PLoS ONE 11(5), e0152816 (2016)
Oyenuga, A., Folsom, A.R., Fashanu, O., Aguilar, D., Ballantyne, C.M.: Plasma galectin-3 and sonographic measures of carotid atherosclerosis in the atherosclerosis risk in communities study. Angiology 70(1), 47–55 (2019)
Varasteh, Z., De Rose, F., Mohanta, S., Li, Y., Zhang, X., Miritsch, B., et al.: Imaging atherosclerotic plaques by targeting Galectin-3 and activated macrophages using ((89)Zr)-DFO- Galectin3-F(ab’)(2) mAb. Theranostics. 11(4), 1864–1876 (2021)
Funding
This work was supported as follows the National Natural Science Foundation of China (82070455); Jiangsu Provincial Health Commission Project (M2020016); the Open Project Program of Guangxi Key Laboratory of Centre of Diabetic Systems Medicine (GKLCDSM-20210101–02); Postgraduate Research&Practice Innovation Program of Jiangsu Province (KYCX20_3051).
Author information
Authors and Affiliations
Contributions
All authors contributed to the conception and wrote the review, critically reviewed all parts of the manuscript, accepted its final version prior to submission, and account for its content.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Cai, Y., Sun, Z., Shao, C. et al. Role of galectin-3 in vascular calcification. Glycoconj J 40, 149–158 (2023). https://doi.org/10.1007/s10719-023-10106-x
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10719-023-10106-x