Abstract
Phosphodiesterase 4 inhibitors (PDE4-I), which selectively increase cyclic adenosine monophosphate (cAMP) levels, have shown neuroprotective effects after several neurological injuries inducing blood–brain barrier (BBB) damage including local/focal cerebral ischemia. The present investigated whether roflumilast confers BBB neuroprotection in the hippocampus after transient global cerebral ischemia (TGCI) in rats. TGCI resulted in whole BBB disruption as measured by the increase of Evans blue (EB) and IgG extravasation, neurodegeneration, and downregulation of claudin-5 and endothelial nitric oxide synthase (eNOS) levels in the CA1 hippocampal subfield of ischemic rats. Roflumilast attenuated BBB disruption and restored the levels of eNOS in the CA1 hippocampal area. Moreover, roflumilast increased the levels of B2 cell lymphoma (BcL-2) and neuron-glial antigen-2 (NG2) in the CA1 subfield after global ischemia in rats. The protective effects of roflumilast against TGCI-induced BBB breakdown might involve preservation of BBB integrity, vascularization and angiogenesis, and myelin repair.





Similar content being viewed by others
Availability of Data and Materials
The experimental data will be available when requested.
References
Abdullahi W, Tripathi D, Ronaldson PT (2018) Blood-brain barrier dysfunction in ischemic stroke: targeting tight junctions and transporters for vascular protection. Am J Physiol Cell Physiol 315(3):C343–C356. https://doi.org/10.1152/ajpcell.00095.2018
Anuncibay-Soto B, Pérez-Rodriguez D, Santos-Galdiano M, Font-Belmonte E, Ugidos IF, Gonzalez-Rodriguez P, Regueiro-Purriños M, Fernández-López A (2018) Salubrinal and robenacoxib treatment after global cerebral ischemia. Exploring the interactions between ER stress and inflammation. Biochem Pharmacol 151:26–37. https://doi.org/10.1016/j.bcp.2018.02.029
Bacarin CC, Mori MA, Ferreira ED, Romanini CV, de Oliveira RMW, & Milani H (2013) Fish oil provides robust and sustained memory recovery after cerebral ischemia: influence of treatment regimen. Physiol Behav 119:61–71. https://doi.org/10.1016/j.physbeh.2013.06.001
Belayev L, Busto R, Ikeda M, Rubin LL, Kajiwara A, Morgan L, Ginsberg MD (1998) Protection against blood-brain barrier disruption in focal cerebral ischemia by the type IV phosphodiesterase inhibitor BBB022: a quantitative study. Brain Res 787(2):277–285. https://doi.org/10.1016/s0006-8993(97)01499-6
Belayev L, Busto R, Zhao W, Ginsberg MD (1996) Quantitative evaluation of blood-brain barrier permeability following middle cerebral artery occlusion in rats. Brain Res 739(1–2):88–96. https://doi.org/10.1016/s0006-8993(96)00815-3
Bernardo-Castro et al (2020) Pathophysiology of blood-brain barrier permeability throughout the different stages of ischemic stroke and its implication on hemorrhagic transformation and recovery. Front Neurol 11:594672. https://doi.org/10.3389/fneur.2020.594672
Bieber M, Schuhmann MK, Kollikowski AM, Stegner D, Nieswandt B, Pham M, Stoll G (2021) Targeting platelet glycoprotein VI attenuates progressive ischemic brain damage before recanalization during middle cerebral artery occlusion in mice. Exp Neurol 344:113804. https://doi.org/10.1016/j.expneurol.2021.113804
Block F, Tondar A, Schmidt W, Schwarz M (1997) Delayed treatment with rolipram protects against neuronal damage following global ischemia in rats. Neuroreport 8(17):3829–32. https://doi.org/10.1097/00001756-199712010-00033
Bonato JM, Meyer E, de Mendonça P, Milani H, Prickaerts J, de Oliveira RMW (2021) Roflumilast protects against spatial memory impairments and exerts anti-inflammatory effects after transient global cerebral ischemia. Eur J Neurosci 53(4):1171–1188. https://doi.org/10.1111/ejn.15089
Brouns R, Wauters A, De Surgeloose D, Mariën P, de Deyn PP (2011) Biochemical markers for blood-brain barrier dysfunction in acute ischemic stroke correlate with evolution and outcome. Eur Neurol 65(1):23–31. https://doi.org/10.1159/000321965
Cai N, Xu B, Li X, Qin Y, Li M, Chen K, Xu J, Wang H (2022) Roflumilast, a cyclic nucleotide phosphodiesterase 4 inhibitor, protects against cerebrovascular endothelial injury following cerebral ischemia/reperfusion by activating the Notch1/Hes1 pathway. European journal of pharmacology 926:175027. https://doi.org/10.1016/j.ejphar.2022.175027
Carmichael ST (2016) The 3 Rs of stroke biology: radial, relayed, and regenerative. Neurotherapeutics 13(2):348–359. https://doi.org/10.1007/s13311-015-0408-0
Chen J, Yu H, Zhong J, Feng H, Wang H, Cheng Y, Zou Z, Huang C, Zhou Z, Zheng W, Xu J (2017) The phosphodiesterase-4 inhibitor, FCPR16, attenuates ischemia-reperfusion injury in rats subjected to middle cerebral artery occlusion and reperfusion. Brain Res Bull 137:98–106. https://doi.org/10.1016/j.brainresbull.2017.11.010
Frisch S, Thiel F, Schroeter ML, Jentzsch RT (2017) Apathy and Cognitive Deficits in Patients with Transient Global Ischemia After Cardiac Arrest. Cognitive and Behavioral Neurology : Official Journal of the Society for Behavioral and Cognitive Neurology 30(4):172–175. https://doi.org/10.1097/WNN.0000000000000139
Garthwaite J (1991) Glutamate, nitric oxide and cell-cell signalling in the nervous system. Trends Neurosci 14(2):60–67. https://doi.org/10.1016/0166-2236(91)90022-m
Geri G, Mongardon N, Daviaud F, Empana JP, Dumas F, Cariou A (2014) Neurological consequences of cardiac arrest: where do we stand? Ann Fr Anesth Reanim 33(2):98–101. https://doi.org/10.1016/j.annfar.2013.11.003
Guo X, Yuan J, Li M, Wang M, Lv P (2021) Neuroprotection of intermedin against cerebral ischemia/reperfusion injury through cerebral microcirculation improvement and apoptosis inhibition. Journal of Molecular Neuroscience : MN 71(4):767–777. https://doi.org/10.1007/s12031-020-01697-3
Guo X, Yuan J, Wang J, Cui C, Jiang P (2019) Calcitriol alleviates global cerebral ischemia-induced cognitive impairment by reducing apoptosis regulated by VDR/ERK signaling pathway in rat hippocampus. Brain research 1724:146430. https://doi.org/10.1016/j.brainres.2019.146430
Haglund M, Lindberg E, Englund E (2019) Hippocampus and basal ganglia as potential sentinel sites for ischemic pathology after resuscitated cardiac arrest. Resuscitation 139:230–233. https://doi.org/10.1016/j.resuscitation.2019.04.012
Haley MJ, Lawrence CB (2017) The blood-brain barrier after stroke: structural studies and the role of transcytotic vesicles. Journal of Cerebral Blood Flow and Metabolism : Official Journal of the International Society of Cerebral Blood Flow and Metabolism 37(2):456–470. https://doi.org/10.1177/0271678X16629976
Huang Z, Huang PL, Ma J, Meng W, Ayata C, Fishman MC, Moskowitz MA (1996) Enlarged infarcts in endothelial nitric oxide synthase knockout mice are attenuated by nitro-L-arginine. J Cereb Blood Flow Metab 16(5):981–987. https://doi.org/10.1097/00004647-199609000-00023
Huang ZG, Xue D, Preston E, Karbalai H, Buchan AM (1999) Biphasic opening of the blood-brain barrier following transient focal ischemia: effects of hypothermia. Can J Neurol Sci J Can Des Sci Neurol 26:298–304
Hwang L, Choi IY, Kim SE, Ko IG, Shin MS, Kim CJ, Kim SH, Jin JJ, Chung JY, Yi JW (2013) Dexmedetomidine ameliorates intracerebral hemorrhage-induced memory impairment by inhibiting apoptosis and enhancing brain-derived neurotrophic factor expression in the rat hippocampus. Int J Mol Med 31(5):1047–1056. https://doi.org/10.3892/ijmm.2013.1301
Jiang X, Andjelkovic AV, Zhu L, Yang T, Bennett MVL, Chen J, Keep RF, Shi Y. Bloodbrain barrier dysfunction and recovery after ischemic stroke (2018) Prog Neurobiol 163–164:144–171. https://doi.org/10.1016/j.pneurobio.2017.10.001
Jin X, Riew TR, Kim S, Kim HL, Lee MY (2020) Spatiotemporal profile and morphological changes of NG2 Glia in the CA1 region of the rat hippocampus after transient forebrain ischemia. Experimental Neurobiology 29(1):50–69. https://doi.org/10.5607/en.2020.29.1.50
Jin Z, Ke J, Guo P, Wang Y, Wu H (2019) Quercetin improves blood-brain barrier dysfunction in rats with cerebral ischemia reperfusion via Wnt signaling pathway. American Journal of Translational Research 11(8):4683–4695
Johnston KC, Bruno A, Pauls Q, Hall CE, Barrett KM, Barsan W, Fansler A, Van de Bruinhorst K, Janis S, Durkalski-Mauldin VL, Neurological emergencies treatment trials network and the SHINE trial investigators, (2019) Intensive vs standard treatment of hyperglycemia and functional outcome in patients with acute ischemic stroke: the SHINE randomized clinical trial. JAMA 322(4):326–335. https://doi.org/10.1001/jama.2019.9346
Ju F, Ran Y, Zhu L, Cheng X, Gao H, Xi X, Yang Z, Zhang S (2018) Increased BBB permeability enhances activation of microglia and exacerbates loss of dendritic spines after transient global cerebral ischemia. Front Cell Neurosci 12:236. https://doi.org/10.3389/fncel.2018.00236
Kho AR, Choi BY, Lee SH, Hong DK, Lee SH, Jeong JH, Park KH, Song HK, Choi HC, Suh SW (2018) Effects of protocatechuic acid (PCA) on global cerebral ischemia-induced hippocampal neuronal death. Int J Mol Sci 19(5):1420. https://doi.org/10.3390/ijms19051420
Kirino T (1982) Delayed neuronal death in the gerbil hippocampus following ischemia. Brain Res 239(1):57–69. https://doi.org/10.1016/0006-8993(82)90833-2
Kraft P, Schwarz T, Göb E, Heydenreich N, Brede M, Meuth SG, Kleinschnitz C (2013) The phosphodiesterase-4 inhibitor rolipram protects from ischemic stroke in mice by reducing blood-brain-barrier damage, inflammation and thrombosis. Exp Neurol 247:80–90. https://doi.org/10.1016/j.expneurol.2013.03.026
Kubo K, Nakao S, Jomura S, Sakamoto S, Miyamoto E, Xu Y, Tomimoto H, Inada T, Shingu K (2009) Edaravone, a free radical scavenger, mitigates both gray and white matter damages after global cerebral ischemia in rats. Brain Res 1279:139–146. https://doi.org/10.1016/j.brainres.2009.04.045
Lakics V, Karran EH, Boess FG (2010) Quantitative comparison of phosphodiesterase mRNA distribution in human brain and peripheral tissues. Neuropharmacology 59(6):367–74. https://doi.org/10.1016/j.neuropharm.2010.05.004
Langdon KD, Granter-Button S, Corbett D (2008) Persistent behavioral impairments and neuroinflammation following global ischemia in the rat. Eur J Neurosci 28(11):2310–2318. https://doi.org/10.1111/j.1460-9568.2008.06513.x
Li HZ, Chen JF, Liu M, Shen J (2018) Effect of hyperbaric oxygen on the permeability of the blood-brain barrier in rats with global cerebral ischemia/reperfusion injury. Biomedicine & pharmacotherapy=Biomedecine & pharmacotherapie 108:1725–1730. https://doi.org/10.1016/j.biopha.2018.10.025
Li LX, Cheng YF, Lin HB, Wang C, Xu JP, Zhang HT (2011) Prevention of cerebral ischemia-induced memory deficits by inhibition of phosphodiesterase-4 in rats. Metab Brain Dis 26(1):37–47. https://doi.org/10.1007/s11011-011-9235-0
Li X, Tian X (2021) The protective effects of Pimavanserin against cerebral ischemia-induced brain injury. Bioengineered 12(1):7481–7494. https://doi.org/10.1080/21655979.2021.1978617
Lorberboym M, Lampl Y, Sadeh M (2003) Correlation of 99mTc-DTPA SPECT of the blood-brain barrier with neurologic outcome after acute stroke. Journal of Nuclear Medicine : Official Publication, Society of Nuclear Medicine 44(12):1898–1904
Luo J, Yang L, Yang J, Yang D, Liu BC, Liu D, Liang BM, Liu CT (2018) Efficacy and safety of phosphodiesterase 4 inhibitors in patients with asthma: a systematic review and meta-analysis. Respirology (Carlton, Vic.) 23(5):467–477. https://doi.org/10.1111/resp.13276
Mallon BS, Shick HE, Kidd GJ, Macklin WB (2002) Proteolipid promoter activity distinguishes two populations of NG2-positive cells throughout neonatal cortical development. J Neurosci 1;22(3):876–885. https://doi.org/10.1523/JNEUROSCI.22-03-00876.2002
Moulaert VR, Wachelder EM, Verbunt JA, Wade DT, van Heugten CM (2010) Determinants of quality of life in survivors of cardiac arrest. J Rehabil Med 42(6):553–558. https://doi.org/10.2340/16501977-0547
Mun CH, Lee WT, Park KA, Lee JE (2010) Regulation of endothelial nitric oxide synthase by agmatine after transient global cerebral ischemia in rat brain. Anatomy & Cell Biology 43(3):230–240. https://doi.org/10.5115/acb.2010.43.3.230
Paxinos G, Watson C (1997) The rat brain in stereotaxic coordinates, 5th edition. San Diego Acad Press. https://doi.org/10.1016/0143-4179(83)90049-5
Peng S, Yan HZ, Liu PR, Shi XW, Liu CL, Liu Q, Zhang Y (2018) Phosphodiesterase 4 inhibitor roflumilast protects rat hippocampal neurons from sevoflurane induced injury via modulation of MEK/ERK signaling pathway. Cellular Physiology and Biochemistry : International Journal of Experimental Cellular Physiology, Biochemistry, and Pharmacology 45(6):2329–2337. https://doi.org/10.1159/000488180
Petito CK, Feldmann E, Pulsinelli WA, Plum F (1987) Delayed hippocampal damage in humans following cardiorespiratory arrest. Neurology 37(8):1281–1286. https://doi.org/10.1212/wnl.37.8.1281
Pilchova I, Klacanova K, Chomova M, Tatarkova Z, Dobrota D, Racay P (2015) Possible contribution of proteins of Bcl-2 family in neuronal death following transient global brain ischemia. Cell Mol Neurobiol 35(1):23–31. https://doi.org/10.1007/s10571-014-0104-3
Prakash R, Carmichael ST (2015) Blood-brain barrier breakdown and neovascularization processes after stroke and traumatic brain injury. Curr Opin Neurol 28(6):556–564. https://doi.org/10.1097/WCO.0000000000000248
Pulsinelli WA, Levy DE, Duffy TE (1982) Regional cerebral blood flow and glucose metabolism following transient forebrain ischemia. Ann Neurol 11(5):499–502. https://doi.org/10.1002/ana.410110510
Santagostino SF, Spinazzi M, Radaelli E (2021) Restricted sensitivity of FJC staining to assess neuronal degeneration and death in preclinical mouse studies. Vet Pathol 58(4):643–649. https://doi.org/10.1177/0300985820985290
Santiago A, Soares LM, Schepers M, Milani H, Vanmierlo T, Prickaerts J, Weffort de Oliveira RM (2018) Roflumilast promotes memory recovery and attenuates white matter injury in aged rats subjected to chronic cerebral hypoperfusion. Neuropharmacology 138:360–370. https://doi.org/10.1016/j.neuropharm.2018.06.019
Scalise AA, Kakogiannos N, Zanardi F, Iannelli F, Giannotta M (2021) The blood-brain and gut-vascular barriers: from the perspective of claudins. Tissue Barriers 3;9(3):1926190. https://doi.org/10.1080/21688370.2021.1926190
Schmued LC, Hopkins KJ (2000) Fluoro-Jade: novel fluorochromes for detecting toxicant-induced neuronal degeneration. Toxicol Pathol 28(1):91–99. https://doi.org/10.1177/019262330002800111
Soares LM, De Vry J, Steinbusch HWM, Milani H, Prickaerts J, Weffort de Oliveira RM (2016) Rolipram improves cognition, reduces anxiety- and despair-like behaviors and impacts hippocampal neuroplasticity after transient global cerebral ischemia. Neuroscience 21(326):69–83. https://doi.org/10.1016/j.neuroscience.2016.03.062
Stamenova V, Nicola R, Aharon-Peretz J, Goldsher D, Kapeliovich M, Gilboa A (2018) Long-term effects of brief hypoxia due to cardiac arrest: Hippocampal reductions and memory deficits. Resuscitation 126:65–71. https://doi.org/10.1016/j.resuscitation.2018.02.016
Strbian et al (2008) The blood-brain barrier is continuously open for several weeks following transient focal cerebral ischemia. Neuroscience 153(1):175–181. https://doi.org/10.1016/j.neuroscience.2008.02.012
Syed YA, Baer A, Hofer MP, González GA, Rundle J, Myrta S, Huang JK, Zhao C, Rossner MJ, Trotter MW, Lubec G, Franklin RJ, Kotter MR (2013) Inhibition of phosphodiesterase-4 promotes oligodendrocyte precursor cell differentiation and enhances CNS remyelination. EMBO Mol Med 5(12):1918–1934. https://doi.org/10.1002/emmm.201303123
Veenith TV, Carter EL, Geeraerts T, Grossac J, Newcombe VF, Outtrim J, Gee GS, Lupson V, Smith R, Aigbirhio FI, Fryer TD, Hong YT, Menon DK, Coles JP (2016) Pathophysiologic mechanisms of cerebral ischemia and diffusion hypoxia in traumatic brain injury. JAMA Neurol 73(5):542–550. https://doi.org/10.1001/jamaneurol.2016.0091
Wang H, Zhang FF, Xu Y, Fu HR, Wang XD, Wang L, Chen W, Xu XY, Gao YF, Zhang JG, Zhang HT (2020) The phosphodiesterase-4 inhibitor roflumilast, a potential treatment for the comorbidity of memory loss and depression in Alzheimer’s disease: a preclinical study in APP/PS1 transgenic mice. Int J Neuropsychopharmacol 23(10):700–711. https://doi.org/10.1093/ijnp/pyaa048
Wen H, Liu L, Zhan L, Liang D, Li L, Liu D, Sun W, Xu E (2018) Neuroglobin mediates neuroprotection of hypoxic postconditioning against transient global cerebral ischemia in rats through preserving the activity of Na+/K+ ATPases. Cell Death Dis 9(6):635. https://doi.org/10.1038/s41419-018-0656-0
Whitaker CM, Beaumont E, Wells MJ, Magnuson DS, Hetman M, Onifer SM (2008) Rolipram attenuates acute oligodendrocyte death in the adult rat ventrolateral funiculus following contusive cervical spinal cord injury. Neurosci Lett 438(2):200–204. https://doi.org/10.1016/j.neulet.2008.03.087
Winkler L, Blasig R, Breitkreuz-Korff O, Berndt P, Dithmer S, Helms HC, Puchkov D, Devraj K, Kaya M, Qin Z, Liebner S, Wolburg H, Andjelkovic AV, Rex A, Blasig IE, Haseloff RF (2021) Tight junctions in the blood-brain barrier promote edema formation and infarct size in stroke - ambivalent effects of sealing proteins. Journal of Cerebral Blood Flow and Metabolism : Official Journal of the International Society of Cerebral Blood Flow and Metabolism 41(1):132–145. https://doi.org/10.1177/0271678X20904687
Wu Q, Qi L, Li H, Mao L, Yang M, Xie R, Yang X, Wang J, Zhang Z, Kong J, Sun B (2017) Roflumilast reduces cerebral inflammation in a rat model of experimental subarachnoid hemorrhage. Inflammation 40(4):1245–1253. https://doi.org/10.1007/s10753-017-0567-8
Xu B, Wang T, Xiao J, Dong W, Wen HZ, Wang X, Qin Y, Cai N, Zhou Z, Xu J, Wang H (2019) FCPR03, a novel phosphodiesterase 4 inhibitor, alleviates cerebral ischemia/reperfusion injury through activation of the AKT/GSK3β/ β-catenin signaling pathway. Biochem Pharmacol 163:234–249. https://doi.org/10.1016/j.bcp.2019.02.023
Yang C, Hawkins KE, Doré S, Candelario-Jalil E (2019) Neuroinflammatory mechanisms of blood-brain barrier damage in ischemic stroke. Am J Physiol Cell Physiol 316(2):C135–C153. https://doi.org/10.1152/ajpcell.00136.2018
Yang F, Liu S, Wang SJ, Yu C, Paganini-Hill A, Fisher MJ (2011) Tissue plasminogen activator expression and barrier properties of human brain microvascular endothelial cells. Cellular Physiology and Biochemistry : International Journal of Experimental Cellular Physiology, Biochemistry, and Pharmacology 28(4):631–638. https://doi.org/10.1159/000335785
Yoshioka H, Niizuma K, Katsu M, Sakata H, Okami N, Chan PH (2011) Consistent injury to medium spiny neurons and white matter in the mouse striatum after prolonged transient global cerebral ischemia. J Neurotrauma 28(4):649–660. https://doi.org/10.1089/neu.2010.1662
Zhao H, Yenari MA, Cheng D, Sapolsky RM, Steinberg GK (2003) Bcl-2 overexpression protects against neuron loss within the ischemic margin following experimental stroke and inhibits cytochrome c translocation and caspase-3 activity. J Neurochem 85(4):1026–1036. https://doi.org/10.1046/j.1471-4159.2003.01756.x
Funding
This study was funded by CAPES/NUFFIC Program (number 88881.156529/2017–0.
Author information
Authors and Affiliations
Contributions
JM Bonato conducted the animals’ surgeries and biochemical assays. She wrote the first draft of the manuscript. BA de Mattos and DV Oliveira helped to perform the animals’ surgeries. H Milani analyzed the data and helped with statistical analysis. J Prickaerts and RMW Oliveira conceived the experimental design, planned the experiments, and performed the data workup. They also contributed to writing the manuscript. All authors discussed the results and commented on the manuscript.
Corresponding author
Ethics declarations
Ethical Approval
The experimental procedures conformed to the ethics of this article and are protected by copyright. All rights reserved principles of the Brazilian College of Animal Experimentation (COBEA) and were approved by the local Ethics Committee on Animal Experimentation of the State University of Maringá (CEUA No. 7995230420).
Competing Interests
JP has a proprietary interest in the PDE4-I roflumilast.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
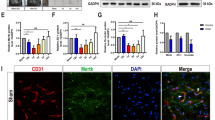
• Transient global cerebral ischemia (TGCI) results in blood-brain barrier (BBB) disruption, neurodegeneration, and a decrease in claudin-5 and eNOS levels in the CA1 hippocampal subfield of rats.
• Roflumilast attenuates BBB disruption and increases the claudin-5 levels in the CA1 hippocampal area of TGCI rats.
• Roflumilast prevents eNOS decreases after TGCI and increases the levels of BcL-2 in the CA1 hippocampus of ischemic rats.
• Roflumilast increases the NG2-glia levels in the CA1 hippocampal area after TGCI in rats.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Bonato, J.M., de Mattos, B.A., Oliveira, D.V. et al. Blood–Brain Barrier Rescue by Roflumilast After Transient Global Cerebral Ischemia in Rats. Neurotox Res 41, 311–323 (2023). https://doi.org/10.1007/s12640-023-00639-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12640-023-00639-2