Abstract
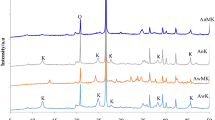
As 1:1 dioctahedral clay minerals, kaolinite and halloysite have similar chemical compositions. However, halloysite often possesses a nanotubular structure and special surface reactivity compared to platy kaolinite. The objective of this current work was to determine the effect of the SiO2/Al2O3 ratio on the microstructure and properties of geopolymers derived from two kinds of kaolin: platy kaolinite and nanotubular halloysite. The chemical structures and compositions of the geopolymers obtained were characterized through X-ray diffraction (XRD), Fourier-transform infrared (FTIR) spectroscopy, and nuclear magnetic resonance (NMR), whereas the microstructural analysis was performed by scanning electron microscopy (SEM), the Brunauer–Emmett–Teller (BET) method, and N2 physisorption analysis. The results indicated that calcined halloysite showed greater geopolymerization reactivity than calcined kaolinite. In addition, the mechanical properties of the clay-based geopolymers depended not only on the SiO2/Al2O3 ratio but also on the morphology of the clay. Crystalline zeolite A and geopolymer were produced after alkali-activation of kaolin with a SiO2/Al2O3 ratio of 2.5; these products possessed porous and heterogeneous microstructures having poor compressive strength. As SiO2/Al2O3 ratios increased to >2.5, geopolymers with compact microstructure and high compressive strength were produced after alkali-activation of kaolin. Notably, at a given condition, halloysite-based geopolymers exhibited greater early compressive strength, more compactness, and more homogeneous microstructure than kaolinite-based geopolymers. This can be attributed to the nanotubular microstructure of halloysite, which can release more Si and Al during alkali activation than platy kaolinite. These results indicated that the various morphologies and microstructures among clays have significant impact on the microstructure and compressive strength of geopolymers.















Similar content being viewed by others
Data Availability
Data will be available on request.
References
Amran, Y. H. M., Alyousef, R., Alabduljabbar, H., & El-Zeadani, M. (2020). Clean production and properties of geopolymer concrete; A review. Journal of Cleaner Production, 251, 119679. https://doi.org/10.1016/j.jclepro.2019.119679
Barrie, E., Cappuyns, V., Vassilieva, E., Adriaens, R., Hollanders, S., Garcés, D., Paredes, C., Pontikes, Y., Elsen, J., & Machiels, L. (2015). Potential of inorganic polymers (geopolymers) made of halloysite and volcanic glass for the immobilisation of tailings from gold extraction in Ecuador. Applied Clay Science, 109–110, 95–106. https://doi.org/10.1016/j.clay.2015.02.025
Bewa, C. N., Tchakouté, H. K., Banenzoué, C., Cakanou, L., Mbakop, T. T., Kamseu, E., & Rüscher, C. H. (2020). Acid-based geopolymers using waste fired brick and different metakaolins as raw materials. Applied Clay Science, 198, 105813. https://doi.org/10.1016/j.clay.2020.105813
Blaise, N. B., Ndigui, B., Emmanuel, Y., Rodrigue, C. K., & Robert, N. (2019). Effect of limestone dosages on some properties of geopolymer from thermally activated halloysite. Construction and Building Materials, 217, 28–35. https://doi.org/10.1016/j.conbuildmat.2019.05.058
Cai, J., Tan, J., & Li, X. (2020). Thermoelectric behaviors of fly ash and metakaolin based geopolymer. Construction and Building Materials, 237, 117757. https://doi.org/10.1016/j.conbuildmat.2019.117757
Chen, K., Wu, D., Xia, L., Cai, Q., & Zhang, Z. (2021). Geopolymer concrete durability subjected to aggressive environments – A review of influence factors and comparison with ordinary Portland cement. Construction and Building Materials, 279, 122496. https://doi.org/10.1016/j.conbuildmat.2021.122496
Davidovits, J. (2011) Geopolymer Chemistry and Applications. 3rd Edition, Institut Geopolymere, Saint-Quentin.
Deng, L., Yuan, P., Liu, D., Annabi-Bergaya, F., Zhou, J., Chen, F., & Liu, Z. (2017). Effects of microstructure of clay minerals, montmorillonite, kaolinite and halloysite, on their benzene adsorption behaviors. Applied Clay Science, 143, 184–191. https://doi.org/10.1016/j.clay.2017.03.035
Duxson, P., Mallicoat, S. W., Lukey, G. C., Kriven, W. M., & Deventer, J. S. J. (2007). The effect of alkali and Si/Al ratio on the development of mechanical properties of metakaolin-based geopolymers. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 292, 8–20. https://doi.org/10.1016/j.colsurfa.2006.05.044
Farhan, N. A., Sheikh, M. N., & Hadi, M. N. S. (2019). Investigation of engineering properties of normal and high strength fly ash based geopolymer and alkali-activated slag concrete compared to ordinary Portland cement concrete. Construction and Building Materials, 196, 26–42. https://doi.org/10.1016/j.conbuildmat.2018.11.083
Ferone, C., Liguori, B., Capasso, I., Colangelo, F., Cioffi, R., Cappelletto, E., & Di Maggio, R. (2015). Thermally treated clay sediments as geopolymer source material. Applied Clay Science, 107, 195–204. https://doi.org/10.1016/j.clay.2015.01.027
Haw, T. T., Hart, F., Rashidi, A., & Pasbakhsh, P. (2020). Sustainable cementitious composites reinforced with metakaolin and halloysite nanotubes for construction and building applications. Applied Clay Science, 188, 105533. https://doi.org/10.1016/j.clay.2020.105533
He, J., Zhang, J., Yu, Y., & Zhang, G. (2012). The strength and microstructure of two geopolymers derived from metakaolin and red mud-fly ash admixture: A comparative study. Construction and Building Materials, 30, 80–91. https://doi.org/10.1016/j.conbuildmat.2011.12.011
He, P., Wang, M., Shuai, F., Jia, D., Shu, Y., Yuan, J., Xu, J., Wang, P., & Yu, Z. (2016). Effects of Si/Al ratio on the structure and properties of metakaolin based geopolymer. Ceramics International, 42, 14416–14422. https://doi.org/10.1016/j.ceramint.2016.06.033
Heah, C. Y., Kamarudin, H., Al Bakri, A. M. M., Bnhussain, M., Luqman, M., Nizar, I. K., Ruzaidi, C. M., & Liew, Y. M. (2012). Study on solids-to-liquid and alkaline activator ratios on kaolin-based geopolymers. Construction and Building Materials, 35, 912–922. https://doi.org/10.1016/j.conbuildmat.2012.04.102
Hollanders, S., Adriaens, R., Skibsted, J., Cizer, Ö., & Elsen, J. (2016). Pozzolanic reactivity of pure calcined clays. Applied Clay Science, 132–133, 552–560. https://doi.org/10.1016/j.clay.2016.08.003
Hounsi, A. D., & Lecomte, G. L. (2013). Kaolin-based geopolymers: Effect of mechanical activation and curing process. Construction and Building Materials, 42, 105–113. https://doi.org/10.1016/j.conbuildmat.2012.12.069
Hu, N., Bernsmeier, D., Grathoff, G. H., & Warr, L. N. (2016). The influence of alkali activatortype, curing temperature and gibbsite on the geopolymerization of an interstratified illite-smectite rich clay from Friedland. Applied Clay Science, 135, 386–393. https://doi.org/10.1016/j.clay.2016.10.021
Izadifar, M., Thissen, P., Steudel, A., Kleeberg, R., Kaufhold, S., Kaltenbach, J., Schuhmann, R., Dehn, F., & Emmerich, K. (2020). Comprehensive examination of dehydroxylation of kaolinite, disordered kaolinite, and dickite: Experimental studies and density functional theory. Clays and Clay Minerals, 68, 319–333. https://doi.org/10.1007/s42860-020-00082-w
Joussein, E., Petit, S. G., Churchman, J., Theng, B. K. G., Righi, D., & Delvaux, B. (2005). Halloysite clay minerals - A review. Clay Minerals, 40, 383–426. https://doi.org/10.1180/0009855054040180
Kaze, C. R., Tchakoute, H. K., Mbakop, T. T., Mache, J. R., Kamseu, E., Melo, U. C., Leonelli, C., & Rahier, H. (2018). Synthesis and properties of inorganic polymers (geopolymers) derived from Cameroon-meta-halloysite. Ceramics International, 44, 18499–18508. https://doi.org/10.1016/j.ceramint.2018.07.070
Kaze, C. R., Alomayri, T., Hasan, A., Tome, S., Lecomte-Nana, G. L., Nemaleu, J. G. D., Tchakoute, H. K., Kamseu, E., Melo, U. C., & Rahier, H. (2020). Reaction kinetics and rheological behaviour of meta-halloysite based geopolymer cured at room temperature: Effect of thermal activation on physicochemical and microstructural properties. Applied Clay Science, 196, 105773. https://doi.org/10.1016/j.clay.2020.105773
Kaze, C. R., Venyite, P., Nana, A., Deutou, J. G. N., Tchakoute, H. K., Rahier, H., Kamseu, E., Melo, U. C., & Leonelli, C. (2020). Meta-halloysite to improve compactness in iron-rich laterite-based alkali activated materials. Materials Chemistry and Physics, 239, 122268. https://doi.org/10.1016/j.matchemphys.2019.122268
Kaze, C. R., Adesina, A., Alomayri, T., Assaedi, H., Kamseu, E., Melo, U. C., Andreola, F., & Leonelli, C. (2021). Characterization, reactivity and rheological behaviour of metakaolin and Meta-halloysite based geopolymer binders. Cleaner Materials, 2, 100025. https://doi.org/10.1016/j.clema.2021.100025
Kaze, C. R., Nana, A., Lecomte-Nana, G. L., Deutou, J. G. N., Kamseu, E., Melo, U. C., Andreola, F., & Leonelli, C. (2022). Thermal behaviour and microstructural evolution of metakaolin and meta-halloysite-based geopolymer binders: A comparative study. Journal of Thermal Analysis and Calorimetry, 147(3), 2055–2071. https://doi.org/10.1007/s10973-021-10555-2
Khalifa, A. Z., Cizer, Ö., Pontikes, Y., Heath, A., Patureau, P., Bernal, S. A., & Marsh, A. T. M. (2020). Advances in alkali-activation of clay minerals. Cement and Concrete Research, 132, 106050. https://doi.org/10.1016/j.cemconres.2020.106050
Li, Y., Chen, M., Song, H., Yuan, P., Liu, D., Zhang, B., & Bu, H. (2020). Methane hydrate formation in the stacking of kaolinite particles with different surface contacts as nanoreactors: A molecular dynamics simulation study. Applied Clay Science, 186, 105439. https://doi.org/10.1016/j.clay.2020.105439
Liew, Y. M., Heah, C. Y., Al Bakri, M. M., & Hussin, K. (2016). Structure and properties of clay-based geopolymer cements: A review. Progress in Materials Science, 83, 595–629. https://doi.org/10.1016/j.pmatsci.2016.08.002
Liu, J., Zha, F., Xu, L., Kang, B., Yang, C., Zhang, W., Zhang, J., & Liu, Z. (2020). Zinc leachability in contaminated soil stabilized/solidified by cement-soda residue under freeze-thaw cycles. Applied Clay Science, 186, 105474. https://doi.org/10.1016/j.clay.2020.105474
Liu, J., Doh, J., Dinh, H. L., Ong, D. E. L., Zi, G., & You, I. (2022). Effect of Si/Al molar ratio on the strength behavior of geopolymer derived from various industrial waste: A current state of the art review. Construction and Building Materials, 329, 127134. https://doi.org/10.1016/j.conbuildmat.2022.127134
Lolli, F., Manzano, H., Provis, J. L., Bignozzi, M. C., & Masoero, E. (2018). Atomistic Simulations of Geopolymer Models: The Impact of Disorder on Structure and Mechanics. ACS Applied Materials & Interfaces, 10, 22809–22820. https://doi.org/10.1021/acsami.8b03873
Ma, Y., Hu, J., & Ye, G. (2013). The pore structure and permeability of alkali activated fly ash. Fuel, 104, 771–780. https://doi.org/10.1016/j.fuel.2012.05.034
Madejová, J., & Komadel, P. (2001). Baseline studies of the clay minerals society source clays: Infrared methods. Clays and Clay Minerals, 49, 372–373. https://doi.org/10.1346/CCMN.2001.0490508
Maia, A. Á. B., Angélica, R. S., de Freitas Neves, R., Pöllmann, H., Straub, C., & Saalwächter, K. (2014). Use of 29Si and 27Al MAS NMR to study thermal activation of kaolinites from Brazilian Amazon kaolin wastes. Applied Clay Science, 87, 189–196. https://doi.org/10.1016/J.CLAY.2013.10.028
Martina, M. C., Lorenzo, L., Giuseppe, C., Giuseppe, C., & Stefana, M. (2022). Halloysite based geopolymers filled with wax microparticles as sustainable building materials with enhanced thermo-mechanical performances. Journal of Environmental Chemical Engineering, 10, 108594. https://doi.org/10.1016/j.jece.2022.108594
Mbey, J. A., Thomas, F., Razafitianamaharavo, A., Caillet, C., & Villiéras, F. (2019). A comparative study of some kaolinites surface properties. Applied Clay Science, 172, 135–145. https://doi.org/10.1016/j.clay.2019.03.005
Medri, V., Fabbri, S., Dedecek, J., Sobalik, Z., Tvaruzkova, Z., & Vaccari, A. (2010). Role of the morphology and the dehydroxylation of metakaolins on geopolymerization. Applied Clay Science, 50, 538–545. https://doi.org/10.1016/j.clay.2010.10.010
Najafi, E. K., Chenari, R. J., & Arabani, M. (2020). The potential use of clay-fly ash geopolymer in the design of active-passive liners: A review. Clays and Clay Minerals, 68, 296–308. https://doi.org/10.1007/s42860-020-00074-w
Nana, A., Ngouné, J., Kaze, R. C., Boubakar, L., Tchounang, S. K., Tchakouté, H. K., Kamseu, E., & Leonelli, C. (2019). Room-temperature alkaline activation of feldspathic solid solutions: Development of high strength geopolymers. Construction and Building Materials, 195, 258–268. https://doi.org/10.1016/j.conbuildmat.2018.11.068
Navid, R., Carsten, K., Carsten, G., Paul, K., & Mehdi, M. (2023). Halloysite reinforced 3D-printable geopolymers. Cement and Concrete Composites, 136, 104894. https://doi.org/10.1016/j.cemconcomp.2022.104894
Nemaleu, J. G. D., Kaze, C. R., Tome, S., Alomayri, T., Assaedi, H., Kamseu, E., Melo, U. C., & Sglavo, V. M. (2021). Powdered banana peel in calcined halloysite replacement on the setting times and engineering properties on the geopolymer binders. Construction and Building Materials, 279, 122480. https://doi.org/10.1016/j.conbuildmat.2021.122480
Nicolas, S. R., Cyr, M., & Escadeillas, G. (2013). Characteristics and applications of flash metakaolins. Applied Clay Science, 83–84, 253–262. https://doi.org/10.1016/j.clay.2013.08.036
Nkwaju, R. Y., Djobo, J. N. Y., Nouping, J. N. F., Huisken, P. W. M., Deutou, J. G. N., & Courard, L. (2019). Iron-rich laterite-bagasse fibers based geopolymer composite: Mechanical, durability and insulating properties. Applied Clay Science, 183, 105333. https://doi.org/10.1016/j.clay.2019.105333
Okada, K., Ōtsuka, N., & Ossaka, J. (1986). Characterization of Spinel Phase Formed in the Kaolin-Mullite Thermal Sequence. Journal of the American Ceramic Society, 69, C-251–C−253. https://doi.org/10.1111/j.1151-2916.1986.tb07353.x
Parker, T. W. (1969). A Classification of Kaolinites by Infrared Spectroscopy. Clay Minerals, 8, 135–141. https://doi.org/10.1180/claymin.1969.008.2.02
Prasad, M. S., Reid, K. J., & Murray, H. H. (1991). Kaolin: Processing, properties and applications. Applied Clay Science, 6, 87–119. https://doi.org/10.1016/0169-1317(91)90001-P
Provis, J. L., Lukey, G. C., & Deventer, J. S. J. (2005). Do geopolymers actually contain nanocrystalline zeolites? A reexamination of existing results. Chemistry of Materials, 17, 3075–3085. https://doi.org/10.1021/cm050230i
Qian, W., Feng, R., Song, S., García, R. E., Estrella, R. M., Patiño, C. L., & Zhang, Y. (2017). Geopolymerization reaction, microstructure and simulation of metakaolin-based geopolymers at extended Si/Al ratios. Cement and Concrete Composites, 79, 45–52. https://doi.org/10.1016/j.cemconcomp.2017.01.014
Rees, C. A., Provis, J. L., Lukey, G. C., & Deventer, J. S. J. (2007). Attenuated total reflectance fourier transform infrared analysis of fly ash geopolymer gel aging. Langmuir, 23, 8170–8179. https://doi.org/10.1021/la700713g
Shekhovtsova, J., Zhernovsky, I., Kovtun, M., Kozhukhova, N., Zhernovskaya, I., & Kearsley, E. (2018). Estimation of fly ash reactivity for use in alkali-activated cements - A step towards sustainable building material and waste utilization. Journal of Cleaner Production, 178, 22–33. https://doi.org/10.1016/j.jclepro.2017.12.270
Singh, B. (1996). Why does halloysite roll? - A new model. Clays and Clay Minerals, 44(2), 191–196. https://doi.org/10.1346/CCMN.1996.0440204
Singh, P. S., Trigg, M., Burgar, I., & Bastow, T. (2005). Geopolymer formation processes at room temperature studied by 29Si and 27Al MAS-NMR. Materials Science and Engineering: A, 396, 392–402. https://doi.org/10.1016/j.msea.2005.02.002
Skibsted, J., & Andersen, M. D. (2013). The effect of alkali ions on the incorporation of aluminum in the calcium silicate hydrate (C–S–H) phase resulting from Portland cement hydration studied by 29Si MAS NMR. Journal of the American Ceramic Society, 96, 651–656. https://doi.org/10.1111/jace.12024
Smith, M. E., Neal, G., Trigg, M. B., & Drennan, J. (1993). Structural characterization of the thermal transformation of halloysite by solid state NMR. Applied Magnetic Resonance, 4, 157–170. https://doi.org/10.1007/BF03162561
Sonuparlak, B., Sarikaya, M., & Aksay, I. A. (2005). Spinel phase formation during the 980°C exothermic reaction in the kaolinite-to-mullite reaction series. Journal of the American Ceramic Society, 70, 837–842. https://doi.org/10.1111/j.1151-2916.1987.tb05637.x
Sun, Z., & Vollpracht, A. (2017). Isothermal calorimetry and in-situ XRD study of the NaOH activated fly ash, metakaolin and slag. Cement and Concrete Research, 103, 110–122. https://doi.org/10.1016/j.cemconres.2017.10.004
Tan, D., Yuan, P., Annabi-Bergaya, F., Dong, F., Liu, D., & He, H. (2015). A comparative study of tubular halloysite and platy kaolinite as carriers for the loading and release of the herbicide amitrole. Applied Clay Science, 114, 190–196. https://doi.org/10.1016/j.clay.2015.05.024
Tchakouté, H. K., Melele, S. J. K., Djamen, A. T., Kaze, C. R., Kamseu, E., Nanseu, C. N. P., Leonelli, C., & Rüscher, C. H. (2020). Microstructural and mechanical properties of poly(sialate-siloxo) networks obtained using metakaolins from kaolin and halloysite as aluminosilicate sources: A comparative study. Applied Clay Science, 186, 105448. https://doi.org/10.1016/j.clay.2020.105448
Tian, Q., Chen, C., Wang, M., Guo, B., Zhang, H., & Sasaki, K. (2021). Effect of Si/Al molar ratio on the immobilization of selenium and arsenic oxyanions in geopolymer. Environmental Pollution, 274, 116509. https://doi.org/10.1016/j.envpol.2021.116509
Tognonvi, T. M., Petlitckaia, S., Gharzouni, A., Fricheteau, M., Texier-Mandoki, N., Bourbon, X., & Rossignol, S. (2020). High-temperature, resistant, argillite-based, alkali-activated materials with improved post-thermal treatment mechanical strength. Clays and Clay Minerals, 68, 211–219. https://doi.org/10.1007/s42860-020-00067-9
Valentini, L. (2018). Modeling Dissolution-Precipitation Kinetics of Alkali-Activated Metakaolin. ACS Omega, 3, 18100–18108. https://doi.org/10.1021/acsomega.8b02380
Walkley, B., & Provis, J. (2019). Solid-state nuclear magnetic resonance spectroscopy of cements. Materials Today Advances, 1, 100007. https://doi.org/10.1016/j.mtadv.2019.100007
Wang, Y. H., Chen, J. Y., Wu, H. D., & Lei, X. R. (2017). Controllable Preparation of Zeolite P1 From Metakaolin-Based Geopolymers via a Hydrothermal Method. Clays and Clay Minerals, 65, 42–51. https://doi.org/10.1346/CCMN.2016.064048
Wang, R., Wang, J., Dong, T., & Ouyang, G. (2020). Structural and mechanical properties of geopolymers made of aluminosilicate powder with different SiO2/Al2O3 ratio: Molecular dynamics simulation and microstructural experimental study. Construction and Building Materials, 240, 117935. https://doi.org/10.1016/j.conbuildmat.2019.117935
Wang, Q., Guo, H., Yu, T., Yuan, P., Deng, L., & Zhang, B. (2022). Utilization of calcium carbide residue as solid alkali for preparing fly ash-based geopolymers: Dependence of compressive strength and microstructure on calcium carbide residue, water content and curing temperature. Materials, 15, 973. https://doi.org/10.3390/ma15030973
Werling, N., Kaltenbach, J., Weidler, P. G., Schuhmann, R., Dehn, F., & Emmerich, K. (2022). Solubility of calcined kaolinite, montmorillonite, and illite in high molar NaOH and suitability as precursors for geopolymers. Clays and Clay Minerals, 70, 270–289. https://doi.org/10.1007/s42860-022-00185-6
White, R. D., Bavykin, D. V., & Walsh, F. C. (2012). The stability of halloysite nanotubes in acidic and alkaline aqueous suspensions. Nanotechnology, 23, 065705. https://doi.org/10.1088/0957-4484/23/6/065705
Yuan, P., Southon, P. D., Liu, Z. W., Green, M. E. R., Hook, J. M., Antill, S. J., & Kepert, C. J. (2008). Functionalization of halloysite clay nanotubes by grafting with gamma-aminopropyltriethoxysilane. Journal of Physical Chemistry C, 112, 15742–15751. https://doi.org/10.1021/JP805657T
Yuan, P., Tan, D., Aannabi-Bergaya, F., Yan, W., Fan, M., Liu, D., & He, H. (2012). Changes in structure, morphology, porosity, and surface activity of mesoporous halloysite nanotubes under heating. Clays and Clay Minerals, 60, 561–573. https://doi.org/10.1346/CCMN.2012.0600602
Yuan, P., Tan, D., & Annabi-Bergaya, F. (2015). Properties and applications of halloysite nanotubes: Recent research advances and future prospects. Applied Clay Science, 112–113, 75–93. https://doi.org/10.1016/j.clay.2015.05.001
Yuan, J. K., He, P. G., Jia, D. C., Yang, C., Zhang, Y., Yan, S., Yang, Z. H., Duan, X. M., Wang, S. J., & Zhou, Y. (2016). Effect of curing temperature and SiO2/K2O molar ratio on the performance of metakaolin-based geopolymers. Ceramics International, 42, 16184–16190. https://doi.org/10.1016/j.ceramint.2016.07.139
Yuan, P., (2016). Chapter 7 - Thermal-Treatment-Induced Deformations and Modifications of Halloysite, in: Yuan, P., Thill, A., Bergaya, F. (Eds.), Developments in Clay Science, 7, 137–166. https://doi.org/10.1016/B978-0-08-100293-3.00007-8.
Zhang, Z. H., Xiao, Y., Huajun, Z., & Yue, C. (2009). Role of water in the synthesis of calcined kaolin-based geopolymer. Applied Clay Science, 43, 218–223. https://doi.org/10.1016/j.clay.2008.09.003
Zhang, Z., Wang, H., Yao, X., & Zhu, Y. (2012a). Effects of halloysite in kaolin on the formation and properties of geopolymers. Cement and Concrete Composites, 34, 709–715. https://doi.org/10.1016/j.cemconcomp.2012.02.003
Zhang, Z. H., Wang, H., Provis, J. L., Bullen, F., Reid, A., & Zhu, Y. C. (2012b). Quantitative kinetic and structural analysis of geopolymers. Part 1. The activation of metakaolin with sodium hydroxide. Thermochimica Acta, 539, 23–33. https://doi.org/10.1016/j.tca.2012.03.021
Zhang, Z. H., Provis, J. L., Wang, H., Bullen, F., & Reid, A. (2013). Quantitative kinetic and structural analysis of geopolymers. Part 2. Thermodynamics of sodium silicate activation of metakaolin. Thermochimica Acta, 565, 163–171. https://doi.org/10.1016/j.tca.2013.01.040
Zhang, Z. H., Zhu, H. J., Zhou, C. H., & Wang, H. (2016). Geopolymer from kaolin in China: An overview. Applied Clay Science, 119, 31–41. https://doi.org/10.1016/j.clay.2015.04.023
Zhang, B., Guo, H., Deng, L., Fan, W., Yu, T., & Wang, Q. (2020a). Undehydrated kaolinite as materials for the preparation of geopolymer through phosphoric acid-activation. Applied Clay Science, 199, 105887. https://doi.org/10.1016/j.clay.2020.105887
Zhang, B., Guo, H., Yuan, P., Li, Y., Wang, Q., Deng, L., & Liu, D. (2020b). Geopolymerization of halloysite via alkali-activation: Dependence of microstructures on precalcination. Applied Clay Science, 185, 105375. https://doi.org/10.1016/j.clay.2019.105375
Zhang, B., Guo, H., Yuan, P., Deng, L., & Liu, D. (2020c). Novel acid-based geopolymer synthesized from nanosized tubular halloysite: The role of precalcination temperature and phosphoric acid concentration. Cement and Concrete Composites, 110, 103601. https://doi.org/10.1016/j.cemconcomp.2020.103601
Zhang, B., Yuan, P., Guo, H., Deng, L., Li, Y., Li, L., Wang, Q., & Liu, D. (2021). Effect of curing conditions on the microstructure and mechanical performance of geopolymers derived from nanosized tubular halloysite. Construction and Building Materials, 268, 121186. https://doi.org/10.1016/j.conbuildmat.2020.121186
Zhang, B., Yu, T., Deng, L., Li, Y., Guo, H., Zhou, J., Li, L., & Yuan, P. (2022). Ion-adsorption type rare earth tailings for preparation of alkali-based geopolymer with capacity for heavy metals immobilization. Cement and Concrete Composites, 134, 104768. https://doi.org/10.1016/j.cemconcomp.2022.104768
Acknowledgements
Financial support by the National Special Support for High-Level Personnel, Basic and Applied Basic Research Foundation of Guangdong Province (Grant No 2023A1515012180), and the National Natural Science Foundation of China (Grant Nos. 52161145405 and 41972045) are gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing Interest
The authors declare that there is no conflict of interest.
Additional information
Associate Editor: Victoria Krupskaya.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhang, B., Yu, T., Guo, H. et al. Effect of the SiO2/Al2O3 Molar Ratio on the Microstructure and Properties of Clay-based Geopolymers: A Comparative Study of Kaolinite-based and Halloysite-based Geopolymers. Clays Clay Miner. 70, 882–902 (2022). https://doi.org/10.1007/s42860-023-00223-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s42860-023-00223-x