Abstract
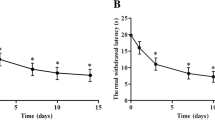
The medial prefrontal cortex (mPFC) is critical for both the sensory and emotional/cognitive components of pain. However, the underlying mechanism remains largely unknown. Here, we examined changes in the transcriptomic profiles in the mPFC of mice with chronic pain using RNA sequencing (RNA-seq) technology. A mouse model of peripheral neuropathic pain was established via chronic constriction injury (CCI) of the sciatic nerve. CCI mice developed sustained mechanical allodynia and thermal hyperalgesia, as well as cognitive impairment four weeks after surgery. RNA-seq was conducted 4 weeks after CCI surgery. Compared with contral group, RNA-seq identified a total 309 and 222 differentially expressed genes (DEGs) in the ipsilateral and contralateral mPFC of CCI model mice, respectively. GO analysis indicated that the functions of these genes were mainly enriched in immune- and inflammation-related processes such as interferon-gamma production and cytokine secretion. KEGG analysis further showed the enrichment of genes involved in the neuroactive ligand–receptor interaction signaling pathway and Parkinson disease pathway that have been reported to be importantly involved in chronic neuralgia and cognitive dysfunction. Our study may provide insights into the possible mechanisms underlying neuropathic pain and pain-related comorbidities.




Similar content being viewed by others
References
Altelaar, A. F., Munoz, J., & Heck, A. J. (2013). Next-generation proteomics: Towards an integrative view of proteome dynamics. Nature Reviews Genetics, 14(1), 35–48. https://doi.org/10.1038/nrg3356
Alvarado, S., Tajerian, M., Millecamps, M., Suderman, M., Stone, L. S., & Szyf, M. (2013). Peripheral nerve injury is accompanied by chronic transcriptome-wide changes in the mouse prefrontal cortex. Molecular Pain, 9, 21. https://doi.org/10.1186/1744-8069-9-21
Arion, D., Corradi, J. P., Tang, S., Datta, D., Boothe, F., He, A., Cacace, A. M., Zaczek, R., Albright, C. F., Tseng, G., & Lewis, D. A. (2015). Distinctive transcriptome alterations of prefrontal pyramidal neurons in schizophrenia and schizoaffective disorder. Molecular Psychiatry, 20(11), 1397–1405. https://doi.org/10.1038/mp.2014.171
Attal, N., Jazat, F., Kayser, V., & Guilbaud, G. (1990). Further evidence for “pain-related” behaviours in a model of unilateral peripheral mononeuropathy. Pain, 41(2), 235–251. https://doi.org/10.1016/0304-3959(90)90022-6
Avigan, P. D., Cammack, K., & Shapiro, M. L. (2020). Flexible spatial learning requires both the dorsal and ventral hippocampus and their functional interactions with the prefrontal cortex. Hippocampus, 30(7), 733–744. https://doi.org/10.1002/hipo.23198
Babenko, V. N., Smagin, D. A., Galyamina, A. G., Kovalenko, I. L., & Kudryavtseva, N. N. (2018). Altered Slc25 family gene expression as markers of mitochondrial dysfunction in brain regions under experimental mixed anxiety/depression-like disorder. BMC Neuroscience, 19(1), 79. https://doi.org/10.1186/s12868-018-0480-6
Baliki, M. N., Chialvo, D. R., Geha, P. Y., Levy, R. M., Harden, R. N., Parrish, T. B., & Apkarian, A. V. (2006). Chronic pain and the emotional brain: Specific brain activity associated with spontaneous fluctuations of intensity of chronic back pain. Journal of Neuroscience, 26(47), 12165–12173. https://doi.org/10.1523/jneurosci.3576-06.2006
Bennett, G. J., & Xie, Y. K. (1988). A peripheral mononeuropathy in rat that produces disorders of pain sensation like those seen in man. Pain, 33(1), 87–107. https://doi.org/10.1016/0304-3959(88)90209-6
Bishnoi, M., Jain, A., Hurkat, P., & Jain, S. K. (2016). Chondroitin sulphate: A focus on osteoarthritis. Glycoconjugate Journal, 33(5), 693–705. https://doi.org/10.1007/s10719-016-9665-3
Cai, W., Yang, T., Liu, H., Han, L., Zhang, K., Hu, X., Zhang, X., Yin, K. J., Gao, Y., Bennett, M. V. L., Leak, R. K., & Chen, J. (2018). Peroxisome proliferator-activated receptor γ (PPARγ): A master gatekeeper in CNS injury and repair. Progress in Neurobiology, 163–164, 27–58. https://doi.org/10.1016/j.pneurobio.2017.10.002
Cao, S., Fisher, D. W., Yu, T., & Dong, H. (2019). The link between chronic pain and Alzheimer’s disease. Journal of Neuroinflammation, 16(1), 204. https://doi.org/10.1186/s12974-019-1608-z
Cardoso-Cruz, H., Lima, D., & Galhardo, V. (2013). Impaired spatial memory performance in a rat model of neuropathic pain is associated with reduced hippocampus-prefrontal cortex connectivity. Journal of Neuroscience, 33(6), 2465–2480. https://doi.org/10.1523/jneurosci.5197-12.2013
Caterina, M. J., Schumacher, M. A., Tominaga, M., Rosen, T. A., Levine, J. D., & Julius, D. (1997). The capsaicin receptor: A heat-activated ion channel in the pain pathway. Nature, 389(6653), 816–824. https://doi.org/10.1038/39807
Chou, C. W., Wong, G. T., Lim, G., McCabe, M. F., Wang, S., Irwin, M. G., & Mao, J. (2011). Peripheral nerve injury alters the expression of NF-κB in the rat’s hippocampus. Brain Research, 1378, 66–71. https://doi.org/10.1016/j.brainres.2011.01.006
Cohen, S. P., Vase, L., & Hooten, W. M. (2021). Chronic pain: An update on burden, best practices, and new advances. Lancet, 397(10289), 2082–2097.
Dawes, J. M., Calvo, M., Perkins, J. R., Paterson, K. J., Kiesewetter, H., Hobbs, C., Kaan, T. K., Orengo, C., Bennett, D. L., & McMahon, S. B. (2011). CXCL5 mediates UVB irradiation-induced pain. Science Translational Medicine, 3(90), 90ra60. https://doi.org/10.1126/scitranslmed.3002193
Dengler-Crish, C. M., Ball, H. C., Lin, L., Novak, K. M., & Cooper, L. N. (2018). Evidence of Wnt/β-catenin alterations in brain and bone of a tauopathy mouse model of Alzheimer’s disease. Neurobiology of Aging, 67, 148–158. https://doi.org/10.1016/j.neurobiolaging.2018.03.021
Descalzi, G., Mitsi, V., Purushothaman, I., Gaspari, S., Avrampou, K., Loh, Y. E., Shen, L., & Zachariou, V. (2017). Neuropathic pain promotes adaptive changes in gene expression in brain networks involved in stress and depression. Science Signaling. https://doi.org/10.1126/scisignal.aaj1549
Doly, S., Martin, P. Y., & Courteix, C. (2021). 5-HT(6) receptor-mTOR: An hyperactive couple in neuropathic pain. Medical Science (paris), 37(5), 547–549. https://doi.org/10.1051/medsci/2021049
Fonseca-Rodrigues, D., Amorim, D., Almeida, A., & Pinto-Ribeiro, F. (2021). Emotional and cognitive impairments in the peripheral nerve chronic constriction injury model (CCI) of neuropathic pain: A systematic review. Behavioural Brain Research, 399, 113008. https://doi.org/10.1016/j.bbr.2020.113008
Ford, B. (2010). Pain in Parkinson’s disease. Movement Disorders, 25(Suppl 1), S98-103. https://doi.org/10.1002/mds.22716
Franceschini, A., Szklarczyk, D., Frankild, S., Kuhn, M., Simonovic, M., Roth, A., Lin, J., Minguez, P., Bork, P., von Mering, C., & Jensen, L. J. (2013). STRING v9.1: Protein-protein interaction networks, with increased coverage and integration. Nucleic Acids Research, 41(1), D808-815. https://doi.org/10.1093/nar/gks1094
Franklin, T. B., Silva, B. A., Perova, Z., Marrone, L., Masferrer, M. E., Zhan, Y., Kaplan, A., Greetham, L., Verrechia, V., Halman, A., Pagella, S., Vyssotski, A. L., Illarionova, A., Grinevich, V., Branco, T., & Gross, C. T. (2017). Prefrontal cortical control of a brainstem social behavior circuit. Nature Neuroscience, 20(2), 260–270. https://doi.org/10.1038/nn.4470
Garcia-Larrea, L., & Peyron, R. (2013). Pain matrices and neuropathic pain matrices: A review. Pain, 154(Suppl 1), S29-s43. https://doi.org/10.1016/j.pain.2013.09.001
Gérard, C., el Mestikawy, S., Lebrand, C., Adrien, J., Ruat, M., Traiffort, E., Hamon, M., & Martres, M. P. (1996). Quantitative RT-PCR distribution of serotonin 5-HT6 receptor mRNA in the central nervous system of control or 5,7-dihydroxytryptamine-treated rats. Synapse (new York, N. Y.), 23(3), 164–173. https://doi.org/10.1002/(sici)1098-2396(199607)23:3%3c164::Aid-syn5%3e3.0.Co;2-6
Ghosh, R., & Tabrizi, S. J. (2018). Clinical features of Huntington’s disease. Advances in Experimental Medicine and Biology, 1049, 1–28. https://doi.org/10.1007/978-3-319-71779-1_1
Hare, B. D., Shinohara, R., Liu, R. J., Pothula, S., DiLeone, R. J., & Duman, R. S. (2019). Optogenetic stimulation of medial prefrontal cortex Drd1 neurons produces rapid and long-lasting antidepressant effects. Nature Communications, 10(1), 223. https://doi.org/10.1038/s41467-018-08168-9
Hu, B., Doods, H., Treede, R. D., & Ceci, A. (2016). Duloxetine and 8-OH-DPAT, but not fluoxetine, reduce depression-like behaviour in an animal model of chronic neuropathic pain. Neuroscience Letters, 619, 162–167. https://doi.org/10.1016/j.neulet.2016.03.019
Huang, J., Gadotti, V. M., Chen, L., Souza, I. A., Huang, S., Wang, D., Ramakrishnan, C., Deisseroth, K., Zhang, Z., & Zamponi, G. W. (2019). A neuronal circuit for activating descending modulation of neuropathic pain. Nature Neuroscience, 22(10), 1659–1668. https://doi.org/10.1038/s41593-019-0481-5
Huang, S., Zhang, Z., Gambeta, E., Xu, S. C., Thomas, C., Godfrey, N., Chen, L., M’Dahoma, S., Borgland, S. L., & Zamponi, G. W. (2020). Dopamine inputs from the ventral tegmental area into the medial prefrontal cortex modulate neuropathic pain-associated behaviors in mice. Cell Reports, 31(12), 107812. https://doi.org/10.1016/j.celrep.2020.107812
Huda, R., Sipe, G. O., Breton-Provencher, V., Cruz, K. G., Pho, G. N., Adam, E., Gunter, L. M., Sullins, A., Wickersham, I. R., & Sur, M. (2020). Distinct prefrontal top-down circuits differentially modulate sensorimotor behavior. Nature Communications, 11(1), 6007. https://doi.org/10.1038/s41467-020-19772-z
Ianov, L., Rani, A., Beas, B. S., Kumar, A., & Foster, T. C. (2016). Transcription profile of aging and cognition-related genes in the medial prefrontal cortex. Front Aging Neuroscience, 8, 113. https://doi.org/10.3389/fnagi.2016.00113
Johnson, P. W., Doe, C. Q., & Lai, S. L. (2018). Drosophila nucleostemin 3 is required to maintain larval neuroblast proliferation. Developmental Biology, 440(1), 1–12. https://doi.org/10.1016/j.ydbio.2018.04.014
Karateev, D. E., Luchikhina, E. L., Makevnina, A. V., & Tangieva, A. R. (2021). Apparatus physiotherapy in the treatment of rheumatic diseases. Voprosy Kurortologii, Fizioterapii i Lechebnoi Fizicheskoi Kultury, 98(2), 31–38. https://doi.org/10.17116/kurort20219802131
Karl, F., Colaço, M. B. N., Schulte, A., Sommer, C., & Üçeyler, N. (2019). Affective and cognitive behavior is not altered by chronic constriction injury in B7–H1 deficient and wildtype mice. BMC Neuroscience, 20(1), 16. https://doi.org/10.1186/s12868-019-0498-4
Kim, J., Lee, S., Kim, J., Ham, S., Park, J. H. Y., Han, S., Jung, Y. K., Shim, I., Han, J. S., Lee, K. W., & Kim, J. (2020). Ca2+-permeable TRPV1 pain receptor knockout rescues memory deficits and reduces amyloid-β and tau in a mouse model of Alzheimer’s disease. Human Molecular Genetics, 29(2), 228–237. https://doi.org/10.1093/hmg/ddz276
Lawlor, P. G. (2002). The panorama of opioid-related cognitive dysfunction in patients with cancer: A critical literature appraisal. Cancer, 94(6), 1836–1853. https://doi.org/10.1002/cncr.10389
Leger, M., Quiedeville, A., Bouet, V., Haelewyn, B., Boulouard, M., Schumann-Bard, P., & Freret, T. (2013). Object recognition test in mice. Nature Protocols, 8(12), 2531–2537. https://doi.org/10.1038/nprot.2013.155
Leite-Almeida, H., Cerqueira, J. J., Wei, H., Ribeiro-Costa, N., Anjos-Martins, H., Sousa, N., Pertovaara, A., & Almeida, A. (2012). Differential effects of left/right neuropathy on rats’ anxiety and cognitive behavior. Pain, 153(11), 2218–2225. https://doi.org/10.1016/j.pain.2012.07.007
Liu, Y., Qin, X., Lu, X., & Jiang, J. (2019). Effects of inhibiting the PI3K/Akt/mTOR signaling pathway on the pain of sciatic endometriosis in a rat model. Canadian Journal of Physiology and Pharmacology, 97(10), 963–970. https://doi.org/10.1139/cjpp-2019-0156
Lu, J., Zhou, W., Dou, F., Wang, C., & Yu, Z. (2021). TRPV1 sustains microglial metabolic reprogramming in Alzheimer’s disease. EMBO Reports, 22(6), e52013. https://doi.org/10.15252/embr.202052013
Mathys, H., Davila-Velderrain, J., Peng, Z., Gao, F., Mohammadi, S., Young, J. Z., Menon, M., He, L., Abdurrob, F., Jiang, X., Martorell, A. J., Ransohoff, R. M., Hafler, B. P., Bennett, D. A., Kellis, M., & Tsai, L. H. (2019). Single-cell transcriptomic analysis of Alzheimer’s disease. Nature, 570(7761), 332–337. https://doi.org/10.1038/s41586-019-1195-2
Maves, T. J., Pechman, P. S., Gebhart, G. F., & Meller, S. T. (1993). Possible chemical contribution from chromic gut sutures produces disorders of pain sensation like those seen in man. Pain, 54(1), 57–69. https://doi.org/10.1016/0304-3959(93)90100-4
Moriarty, O., McGuire, B. E., & Finn, D. P. (2011). The effect of pain on cognitive function: A review of clinical and preclinical research. Progress in Neurobiology, 93(3), 385–404. https://doi.org/10.1016/j.pneurobio.2011.01.002
Ozawa, M., Davis, P., Ni, J., Maguire, J., Papouin, T., & Reijmers, L. (2020). Experience-dependent resonance in amygdalo-cortical circuits supports fear memory retrieval following extinction. Nature Communications, 11(1), 4358. https://doi.org/10.1038/s41467-020-18199-w
Palma-Tortosa, S., Hurtado, O., Pradillo, J. M., Ferreras-Martín, R., García-Yébenes, I., García-Culebras, A., Moraga, A., Moro, M., & Lizasoain, I. (2019). Toll-like receptor 4 regulates subventricular zone proliferation and neuroblast migration after experimental stroke. Brain, Behavior, and Immunity, 80, 573–582. https://doi.org/10.1016/j.bbi.2019.05.002
Restivo, L., Vetere, G., Bontempi, B., & Ammassari-Teule, M. (2009). The formation of recent and remote memory is associated with time-dependent formation of dendritic spines in the hippocampus and anterior cingulate cortex. Journal of Neuroscience, 29(25), 8206–8214. https://doi.org/10.1523/jneurosci.0966-09.2009
Rii, J., Sakamoto, S., Sugiura, M., Kanesaka, M., Fujimoto, A., Yamada, Y., Maimaiti, M., Ando, K., Wakai, K., Xu, M., Imamura, Y., Shindo, N., Hirota, T., Kaneda, A., Kanai, Y., Ikehara, Y., Anzai, N., & Ichikawa, T. (2021). Functional analysis of LAT3 in prostate cancer: Its downstream target and relationship with androgen receptor. Cancer Science, 112(9), 3871–3883. https://doi.org/10.1111/cas.14991
Robinson, M. D., McCarthy, D. J., & Smyth, G. K. (2010). edgeR: A bioconductor package for differential expression analysis of digital gene expression data. Bioinformatics, 26(1), 139–140. https://doi.org/10.1093/bioinformatics/btp616
Saito, R., Smoot, M. E., Ono, K., Ruscheinski, J., Wang, P. L., Lotia, S., Pico, A. R., Bader, G. D., & Ideker, T. (2012). A travel guide to Cytoscape plugins. Nature Methods, 9(11), 1069–1076. https://doi.org/10.1038/nmeth.2212
Scheltens, P., De Strooper, B., Kivipelto, M., Holstege, H., Chételat, G., Teunissen, C. E., Cummings, J., & van der Flier, W. M. (2021). Alzheimer’s disease. Lancet, 397(10284), 1577–1590. https://doi.org/10.1016/s0140-6736(20)32205-4
Sprenger, G. P., Roos, R. A. C., van Zwet, E., Reijntjes, R. H., Achterberg, W. P., & de Bot, S. T. (2021). The prevalence of pain in Huntington’s disease in a large worldwide cohort. Parkinsonism & Related Disorders, 89, 73–78. https://doi.org/10.1016/j.parkreldis.2021.06.015
Tang, Y., Chen, Y., Liu, R., Li, W., Hua, B., & Bao, Y. (2022). Wnt signaling pathways: A role in pain processing. Neuromolecular Medicine. https://doi.org/10.1007/s12017-021-08700-z
Taylor, J. P., McKeith, I. G., Burn, D. J., Boeve, B. F., Weintraub, D., Bamford, C., Allan, L. M., Thomas, A. J., & O’Brien, J. T. (2020). New evidence on the management of Lewy body dementia. Lancet Neurology, 19(2), 157–169. https://doi.org/10.1016/s1474-4422(19)30153-x
Tyrtyshnaia, A. A., Manzhulo, I. V., Sultanov, R. M., & Ermolenko, E. V. (2017). Adult hippocampal neurogenesis in neuropathic pain and alkyl glycerol ethers treatment. Acta Histochemica, 119(8), 812–821. https://doi.org/10.1016/j.acthis.2017.10.007
Villafranca-Faus, M., Vila-Martín, M. E., Esteve, D., Merino, E., Teruel-Sanchis, A., Cervera-Ferri, A., Martínez-Ricós, J., Lloret, A., Lanuza, E., & Teruel-Martí, V. (2021). Integrating pheromonal and spatial information in the amygdalo-hippocampal network. Nature Communications, 12(1), 5286. https://doi.org/10.1038/s41467-021-25442-5
von Loga, I. S., Batchelor, V., Driscoll, C., Burleigh, A., Chia, S. L., Stott, B., Miotla-Zarebska, J., Riley, D., Dell’Accio, F., & Vincent, T. L. (2020). Does pain at an earlier stage of chondropathy protect female mice against structural progression after surgically induced osteoarthritis? Arthritis & Rhematology, 72(12), 2083–2093. https://doi.org/10.1002/art.41421
Wang, G. Q., Cen, C., Li, C., Cao, S., Wang, N., Zhou, Z., Liu, X. M., Xu, Y., Tian, N. X., Zhang, Y., Wang, J., Wang, L. P., & Wang, Y. (2015). Deactivation of excitatory neurons in the prelimbic cortex via Cdk5 promotes pain sensation and anxiety. Nature Communications, 6, 7660. https://doi.org/10.1038/ncomms8660
Wang, Q., & Holst, J. (2015). L-type amino acid transport and cancer: Targeting the mTORC1 pathway to inhibit neoplasia. American Journal of Cancer Research, 5(4), 1281–1294.
Xia, S.-H., Hu, S.-W., Ge, D.-G., Liu, D., Wang, D., Zhang, S., Zhang, Q., Yuan, L., Li, Y.-Q., Yang, J.-X., Wu, P., Zhang, H., Han, M.-H., Ding, H.-L., & Cao, J.-L. (2020). Chronic pain impairs memory formation via disruption of neurogenesis mediated by mesohippocampal brain-derived neurotrophic factor signaling. Biological Psychiatry, 88(8), 597–610. https://doi.org/10.1016/j.biopsych.2020.02.013
Zhang, W. W., Cao, H., Li, Y., Fu, X. J., & Zhang, Y. Q. (2022). Peripheral ablation of type III adenylyl cyclase induces hyperalgesia and eliminates KOR-mediated analgesia in mice. JCI Insight. https://doi.org/10.1172/jci.insight.153191
Funding
This work was supported by the National Natural Science Foundation of China, No. 81971043 and 81771208, STI 2030-Major projects (2021ZD0203203) and Shanghai Municipal Science and Technology Major Project, No. 2018SHZDZX01.
Author information
Authors and Affiliations
Contributions
Q-MZ and Z-RZ did the behavioral tests. X-YH prepared the sample for RNA-seq; Q-MZ and HC analyzed the RNA-seq results. NL participated in data discussion. Y-QZ and HC designed the study. Q-MZ, HC and Y-QZ performed data interpretation, presentation, and writing of the manuscript. HC was responsible for the study.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zheng, QM., Zhou, ZR., Hou, XY. et al. Transcriptome Analysis of the Mouse Medial Prefrontal Cortex in a Chronic Constriction Injury Model. Neuromol Med 25, 375–387 (2023). https://doi.org/10.1007/s12017-023-08742-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12017-023-08742-5