Abstract
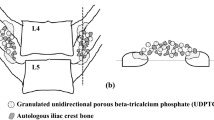
Decalcified bone matrix (DBM) is a widely used alternative material for bone transplantation. In the DBM production process, an effective particle size and the highest utilization rate of raw materials can be achieved only through multiple high-speed circulating comminution. The rat posterolateral lumbar fusion model (PLF) is the most mature small animal model for the initial evaluation of the efficacy of graft materials for bone regeneration and spinal fusion. To evaluate the differences in the in vivo osteogenic effects of DBM pulverization through 1, 5, 9, and 14 high-speed cycles, sixty athymic rats were divided into six groups: single cycling crushing (CC1), 5 cycles of crushing (CC5), 9 cycles of crushing (CC9), 13 cycles of crushing (CC13), autogenous bone graft (ABG) and negative control (NC). Posterolateral lumbar fusion was performed. Six weeks after surgery, the bilateral lumbar fusion of athymic rats was evaluated through manual palpation, X-ray, micro-CT and histological sections. Rank data were tested by the rank-sum test, and nonparametric data were tested by the Kruskal‒Wallis H test. The manual palpation and X-ray results showed that the fusion rate did not significantly differ between the CC1, CC5, CC9, CC13 and ABG groups. However, cavities appeared in CC9 and CC13 on the micro-CT image. The bone mass (BV/TV) of CC1, CC5, CC9 and CC13 was better than that of the ABG group, while almost no osteogenesis was observed in the NC group. Histologically, there was no obvious difference between the four groups except that the CC9 group and CC13 group had more fibrous tissues in the new bone. In conclusion, DMB with different cycling crushing times has no obvious difference in fusion rate of PLF, but it is slightly better than the ABG group.








Similar content being viewed by others
References
Bhamb N, Kanim LEA, Drapeau S, Mohan S, Vasquez E, Shimko D, Mc KW, Bae HW (2019) Comparative efficacy of commonly available human bone graft substitutes as tested for posterolateral fusion in an athymic rat model. Int J Spine Surg 13(5):437–458
Boden SD, Schimandle JH, Hutton WC (1995) An experimental lumbar intertransverse process spinal fusion model. Radiographic, histologic, and biomechanical healing characteristics. Spine (phila Pa 1976) 20(4):412–420
Bomback DA, Grauer JN, Lugo R, Troiano N, Patel T, Friedlaender GE (2004) Comparison of posterolateral lumbar fusion rates of Grafton Putty and OP-1 Putty in an athymic rat model. Spine (phila Pa 1976) 29(15):1612–1617
Brecevich AT, Kiely PD, Yoon BV, Nguyen JT, Cammisa FP, Abjornson C (2017) Efficacy comparison of Accell Evo3 and Grafton demineralized bone matrix putties against autologous bone in a rat posterolateral spine fusion model. Spine J 17(6):855–862
Chung CG, James AW, Asatrian G, Chang L, Nguyen A, Le K, Bayani G, Lee R, Stoker D, Zhang X, Ting K, Péault B, Soo C (2014) Human perivascular stem cell-based bone graft substitute induces rat spinal fusion. Stem Cells Transl Med 3(10):1231–1241
Driscoll JA, Lubbe R, Jakus AE, Chang K, Haleem M, Yun C, Singh G, Schneider AD, Katchko KM, Soriano C, Newton M, Maerz T, Li X, Baker K, Hsu WK, Shah RN, Stock SR, Hsu EL (2020) 3D-printed ceramic-demineralized bone matrix hyperelastic bone composite scaffolds for spinal fusion. Tissue Eng Part A 26(3–4):157–166
Grauer JN, Bomback DA, Lugo R, Troiano NW, Patel TC, Friedlaender GE (2004) Posterolateral lumbar fusions in athymic rats: characterization of a model. Spine J 4(3):281–286
Gruskin E, Doll BA, Futrell FW, Schmitz JP, Hollinger JO (2012) Demineralized bone matrix in bone repair: history and use. Adv Drug Deliv Rev 64(12):1063–1077
Hayashi T, Lord EL, Suzuki A, Takahashi S, Scott TP, Phan K, Tian H, Daubs MD, Shiba K, Wang JC (2016) A comparison of commercially available demineralized bone matrices with and without human mesenchymal stem cells in a rodent spinal fusion model. J Neurosurg Spine 25(1):133–137
Hu T, Abbah SA, Toh SY, Wang M, Lam RW, Naidu M, Bhakta G, Cool SM, Bhakoo K, Li J, Goh JC, Wong HK (2015) Bone marrow-derived mesenchymal stem cells assembled with low-dose BMP-2 in a three-dimensional hybrid construct enhances posterolateral spinal fusion in syngeneic rats. Spine J 15(12):2552–2563
Jakus AE, Rutz AL, Jordan SW, Kannan A, Mitchell SM, Yun C, Koube KD, Yoo SC, Whiteley HE, Richter CP, Galiano RD, Hsu WK, Stock SR, Hsu EL, Shah RN (2016) Hyperelastic “bone”:A highly versatile, growth factor-free, osteoregenerative, scalable, and surgically friendly biomaterial. Sci Transl Med 8(358):358ra127
Kanayama M, Cunningham BW, Weis JC, Parker LM, Kaneda K, McAfee PC (1997) Maturation of the posterolateral spinal fusion and its effect on load-sharing of spinal instrumentation. An in vivo sheep model. J Bone Joint Surg Am 79(11):1710–1720
Lee S, Zhang X, Shen J, James AW, Chung CG, Hardy R, Li C, Girgius C, Zhang Y, Stoker D, Wang H, Wu BM, Peault B, Ting K, Soo C (2015) Brief report: human perivascular stem cells and nel-like protein-1 synergistically enhance spinal fusion in osteoporotic rats. Stem Cells 33(10):3158–3163
Li W, Lee M, Whang J, Siu RK, Zhang X, Liu C, Wu BM, Wang JC, Ting K, Soo C (2010) Delivery of lyophilized Nell-1 in a rat spinal fusion model. Tissue Eng Part A 16(9):2861–2870
Lu SS, Zhang X, Soo C, Hsu T, Napoli A, Aghaloo T, Wu BM, Tsou P, Ting K, Wang JC (2007) The osteoinductive properties of Nell-1 in a rat spinal fusion model. Spine J 7(1):50–60
Group MR 2005,2004. US markets for orthopaedic biomaterials
Martin GJ Jr, Boden SD, Titus L, Scarborough NL (1999) New formulations of demineralized bone matrix as a more effective graft alternative in experimental posterolateral lumbar spine arthrodesis. Spine (phila Pa 1976) 24(7):637–645
Miyazaki M, Morishita Y, He W, Hu M, Sintuu C, Hymanson HJ, Falakassa J, Tsumura H, Wang JC (2009) A porcine collagen-derived matrix as a carrier for recombinant human bone morphogenetic protein-2 enhances spinal fusion in rats. Spine J 9(1):22–30
Montgomery SR, Nargizyan T, Meliton V, Nachtergaele S, Rohatgi R, Stappenbeck F, Jung ME, Johnson JS, Aghdasi B, Tian H, Weintraub G, Inoue H, Atti E, Tetradis S, Pereira RC, Hokugo A, Alobaidaan R, Tan Y, Hahn TJ, Wang JC, Parhami F (2014) A novel osteogenic oxysterol compound for therapeutic development to promote bone growth: activation of hedgehog signaling and osteogenesis through smoothened binding. J Bone Miner Res 29(8):1872–1885
Peterson B, Whang PG, Iglesias R, Wang JC, Lieberman JR (2004) Osteoinductivity of commercially available demineralized bone matrix. Preparations in a spine fusion model. J Bone Joint Surg Am 86(10):2243–2250
Plantz MA, Minardi S, Lyons JG, Greene AC, Ellenbogen DJ, Hallman M, Yamaguchi JT, Jeong S, Yun C, Jakus AE, Blank KR, Havey RM, Muriuki M, Patwardhan AG, Shah RN, Hsu WK, Stock SR, Hsu EL (2021) Osteoinductivity and biomechanical assessment of a 3D printed demineralized bone matrix-ceramic composite in a rat spine fusion model. Acta Biomater 127:146–158
Qiu QQ, Shih MS, Stock K, Panzitta T, Murphy PA, Roesch SC, Connor J (2007) Evaluation of DBM/AM composite as a graft substitute for posterolateral lumbar fusion. J Biomed Mater Res B Appl Biomater 82(1):239–245
Russell N, Walsh WR, Lovric V, Kim P, Chen JH, Larson MJ, Vizesi F (2020) In-vivo performance of seven commercially available demineralized bone matrix fiber and putty products in a rat posterolateral fusion model. Front Surg 7:10
Sandhu HS, Khan SN, Suh DY, Boden SD (2001) Demineralized bone matrix, bone morphogenetic proteins, and animal models of spine fusion: an overview. Eur Spine J 10(Suppl 2(Suppl2)):S122–S131
Schimandle JH, Boden SD (1994a) Spine update. Animal use in spinal research. Spine (phila Pa 1976) 19(21):2474–2477
Schimandle JH, Boden SD (1994b) Spine update. The use of animal models to study spinal fusion. Spine (phila Pa 1976) 19(17):1998–2006
Zhang H, Yang L, Yang XG, Wang F, Feng JT, Hua KC, Li Q, Hu YC (2019) Demineralized bone matrix carriers and their clinical applications: an overview. Orthop Surg 11(5):725–737
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors have no relevant financial interests to disclose. No funds, grants, or other support was receive.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Shao-Lin Ji, Xiao-Dan Zhao and Li-Min Wang have contributed to the work equally and should be regarded as co-first authors.
Jing-Yu Zhang and Yong-Cheng Hu have contributed to the work equally and should be regarded as co-corresponding author.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ji, SL., Zhao, XD., Wang, LM. et al. Comparison of demineralized bone matrix with different cycling crushing times in posterolateral fusion model of athymic rats. Cell Tissue Bank 24, 747–758 (2023). https://doi.org/10.1007/s10561-023-10086-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10561-023-10086-0