Abstract
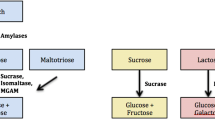
Congenital sucrase–isomaltase deficiency (CSID) is a rare autosomal carbohydrate malabsorption disorder caused by mutations in the sucrase–isomaltase gene. While the prevalence of CSID is high in the indigenous populations of Alaska and Greenland, it is imprecise and ambiguous in the Turkish pediatric population. In this cross-sectional case–control study, which is retrospective in nature, next-generation sequencing (NGS) results obtained from records of 94 pediatric patients with chronic nonspecific diarrhea were reviewed. Demographic characteristics, clinical symptoms and treatment responses of those diagnosed with CSID were evaluated. We identified one new, homozygous frame-shift mutation and 10 other heterozygous mutations. Two cases were from the same family and nine were from different families. While the median age at onset of symptoms was 6 months (0–12), median age at diagnosis was 60 months (18–192) with a median delay of 5 years and 5 months (10 months -15 years and 5 months) in diagnosis. Clinical symptoms included diarrhea (100%), abdominal pain (54.5%), vomiting after consuming sucrose (27.2%), diaper dermatitis (36.3%) and growth retardation (81%). Our clinical study revealed that sucrase-isomaltase deficiency may have been underdiagnosed in patients with chronic diarrhea in Turkey. In addition, the frequency of heterozygous mutation carriers was significantly higher than that of homozygous mutation carriers and those with a heterozygous mutations responded well to the treatment.
Similar content being viewed by others
References
Antonowicz I., Lloyd-Still J. D., Khaw K. T. and Shwachman H. 1972 Congenital sucrase-isomaltase deficiency. Observations over a period of 6 years. Pediatrics 49, 847–853.
Bell R., Draper H. and Bergan J. G. 1973 Sucrose, lactose, and glucose intolerance in northern Alaskan Eskimos. Am. J. Clin. Nutr. 26, 1185–1190.
Capalbo A., Valero R. A., Jimenez-Almazan J., Pardo P. M., Fabiani M., Jime´nez D. et al. 2019 Optimizing clinical exome design and parallel gene-testing for recessive genetic conditions in preconception carrier screening: Translational research genomic data from 14,125 exomes. PLoS Genet. 15, e1008409.
Chiruvella V., Cheema A., Arshad H. M. S., Chan J. T. and Yap J. E. L. 2021 Sucrase-isomaltase deficiency causing persistent bloating and diarrhea in an adult female. Cureus 13, e14349.
Ceyhan-Birsoy O., Murry J. B., Machini K., Lebo M. S., Yu T. W., Fayer S. et al. 2019 interpretation of genomic sequencing results in healthy and Ill newborns: results from the BabySeq project. Am. J. Hum. Genet. 104, 76–93.
Ellestad-Sayad J., Haworth J. and Hildes J. 1978 Disaccharide malabsorption and dietary patterns in two Canadian Eskimo communities. Am. J. Clin. Nutr. 31, 1473–1478.
Garcia-Etxebarria K., Zheng T., Bonfiglio F., Bujanda L., Dlugosz A., Lindberg G. et al. 2018 Increased prevalence of rare sucrase-isomaltase pathogenic variants in irritable bowel syndrome patients. Clin. Gastroenterol. Hepatol. 16, 1673–1676.
Gericke B., Amiri M., Scott C. R. and Naim H. Y. 2017 Molecular pathogenicity of novel sucrase-isomaltase mutations found in congenital sucrase-isomaltase deficiency patients. Biochim. Biophys. Acta Mol. Basis Dis. 1863, 817–826.
Gupta S. K., Chong S. K. and Fitzgerald J. F. 1999 Disaccharidase Activities in children: normal values and comparison based on symptoms and histologic changes. J. Pediatr. Gastroenterol. Nutr. 28, 246–251.
Haberman Y., Di Segni A., Loberman-Nachum N., Barel O., Kunik V., Eyal E. et al. 2017 Congenital sucrase-isomaltase deficiency: a novel compound heterozygous mutation causing aberrant protein localization. J. Pediatr. Gastroenterol. Nutr. 64, 770–776.
Karakoyun M., Kilicoglu E., Sahan Y., Baran M., Unal F. and Aydogdu S. 2015 Our cases with sucrase isomaltase deficiency. J. Gastroenterol. Dig. Sys. 5, 354–358.
Karnsaku W., Luginbuehl U., Hahn D., Sterchi E., Avery S., Sen P. et al. 2002 Disaccharidase activities in dyspeptic children: biochemical and molecular investigations of maltase-glucoamylase activity. J. Pediatr. Gastroenterol. Nutr. 35, 551–556.
Marcadier J. L., Boland M., Scott C. R., Kheirie Issa K., Wu Z., McIntyre A. D. et al. 2015 Congenital sucrase-isomaltase deficiency: identification of a common Inuit founder mutation. Can. Med. Assoc. J. 187, 102–107.
Reinshagen K., Keller K. M., Haase B., Leeb T., Naim H. Y. and Zimmer K. P. 2008 Mosaic pattern of sucrase isomaltase deficiency in two brothers. Pediatr. Res. 63, 79–83.
Robayo-Torres C. C., Diaz-Sotomayor M., Hamaker B. R., Baker S. S., Chumpitazi B. P., Opekun A. R. et al. 2018 13C-labeled-starch breath test in congenital sucrase-isomaltase deficiency. J. Pediatr. Gastroenterol. Nutr. 66, 61–64.
Roberto Q. C., Claudia R. T., Zihua A., Bruce H., Andrea Q. and Gary D. B. 2007 Luminal Substrate “Brake” on Mucosal Maltase-glucoamylase Activity Regulates Total Rate of Starch Digestion to Glucose. Journal of Pediatric Gastroenterology and Nutrition 45, 32–43.
Treem W. R. 2012 Clinical aspects and treatment of congenital sucrase-isomaltase deficiency. J. Pediatr. Gastroenterol. Nutr. 55, 7–13.
Treem W. R. 2009 Congenital sucrase-isomaltase deficiency (CSID) in the era of Sucraid. J. Pediatr. Gastroenterol. Nutr. 53, 85–88.
Weijers H. A., de Va K. J., Mossel D. A. and Dicke W. K. 1960 Diarrhoea caused by deficiency of sugar-splitting enzymes. Lancet 2, 296–297.
Zhou J., Zhao Y., Qian X., Cheng Y., Cai H., Chen M. et al. 2021 Two novel mutations in the SI gene associated with congenital sucrase-isomaltase deficiency: a case report in China. Front. Pediatr. 9, 731–716.
Author information
Authors and Affiliations
Corresponding author
Additional information
Corresponding editor: Shrish Tiwari
Rights and permissions
About this article
Cite this article
Taskin, D.G., Civan, H.A., SarI, E.E. et al. Prevalence of congenital sucrase-isomaltase deficiency in Turkey may be much higher than the estimates. J Genet 102, 31 (2023). https://doi.org/10.1007/s12041-023-01428-8
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12041-023-01428-8