Abstract
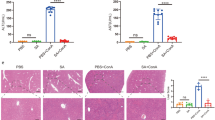
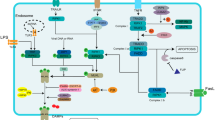
The disease sepsis is caused by an infection that damages organs. Liver injury, with ferroptosis playing a key role, is an early sign of sepsis. G protein-coupled receptor 116 (GPR116) is essential in the maintenance of functional homeostasis in various systems of the body and has been proven to play a protective role in septic lung injury. However, it’s role in septic liver injury remains unclear. In this study, we found that hepatic ferroptosis during sepsis was accompanied by GPR116 upregulation. Hepatocyte-specific GPR116 gene deletion can prevent hepatic ferroptosis, thereby alleviating sepsis-induced liver dysfunction and improving mouse survival, which was verified in vivo. Mechanistically, GPR116 aggravated mitochondrial damage and lipid peroxidation in hepatocytes by inhibiting system Xc–/GSH/GPX4 in overexpression experiments. In conclusion, we have identified GPR116 as a vital mediator of ferroptosis in sepsis-induced liver injury. It is thus an attractive therapeutic target in sepsis.






Similar content being viewed by others
Data availability
All data related to this paper may also be requested from the corresponding authors.
Abbreviations
- ALT :
-
Alanine aminotransferase
- AST :
-
Aspartate aminotransferase
- ALD :
-
Alcoholic liver disease
- AML12 :
-
Alpha mouse liver 12
- CLP :
-
Cecal ligation and puncture
- DILI :
-
Drug-induced liver injury
- ELISA :
-
Enzyme-linked immunosorbent assay
- GPX4 :
-
Glutathione peroxidase 4
- GPCRs :
-
G protein-coupled receptors
- GPR116/ADGRF5 :
-
G protein-coupled receptor 116/Adhesion G protein-coupled receptor F5
- GSH :
-
Glutathione
- GSH-PX :
-
Glutathione peroxidase
- H&E :
-
Haematoxylin and eosin
- IL-6 :
-
Interleukin-6
- IRI :
-
Ischemia/reperfusion-related injury
- MDA :
-
Malondialdehyde
- NAFLD :
-
Nonalcoholic fatty liver disease
- NET :
-
Neutrophil extracellular trap
- RIPA :
-
Radioimmunoprecipitation assay
- SD :
-
Standard deviation
- TEM :
-
Transmission electron microscope
- TNF-α :
-
Umor necrosis factor-α
- SLC7A11 :
-
Solute carrier family 7 member 11
- SLC3A2 :
-
Solute carrier family 3 member 2
- WT :
-
Wild-type
- 4-HNE :
-
4-Hydroxy-2-noneal
References
Ariestanti DM, Ando H, Hirose S, Nakamura N. Targeted disruption of Ig-Hepta/Gpr116 Causes emphysema-like symptoms that are associated with alveolar macrophage activation. J Biol Chem. 2015;290(17):11032–40. https://doi.org/10.1074/jbc.M115.648311.
Bridges JP, Ludwig M-G, Mueller M, Kinzel B, Sato A, Xu Y, Ikegami M. Orphan G protein-coupled receptor GPR116 regulates pulmonary surfactant pool size. Amer J Respir Cell Molec Biol. 2013;49(3):348–57. https://doi.org/10.1165/rcmb.2012-0439OC.
Cecconi M, Evans L, Levy M, Rhodes A. Sepsis and septic shock. Lancet. 2018;392(10141):75–87. https://doi.org/10.1016/S0140-6736(18)30696-2.
Chen J, Li X, Ge C, Min J, Wang F. The multifaceted role of ferroptosis in liver disease. Cell Death Differ. 2022;29(3):467–80. https://doi.org/10.1038/s41418-022-00941-0.
Chen X, Kang R, Kroemer G, Tang D. Ferroptosis in infection, inflammation, and immunity. J Exp Med. 2021a;218(6):e20210518. https://doi.org/10.1084/jem.20210518.
Chen X, Kang R, Kroemer G, Tang D. Organelle-specific regulation of ferroptosis. Cell Death Differ. 2021b;28(10):2843–56. https://doi.org/10.1038/s41418-021-00859-z.
Chen X, Li J, Kang R, Klionsky DJ, Tang D. Ferroptosis: machinery and regulation. Autophagy. 2021c;17(9):2054–81. https://doi.org/10.1080/15548627.2020.1810918.
Dixon SJ, Lemberg KM, Lamprecht MR, Skouta R, Zaitsev EM, Gleason CE, Stockwell BR. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell. 2012;149(5):1060–72. https://doi.org/10.1016/j.cell.2012.03.042.
Gan B. Mitochondrial regulation of ferroptosis. J Cell Biol. 2021;220(9):e202105043. https://doi.org/10.1083/jcb.202105043.
Georgiadi A, Lopez-Salazar V, Merahbi RE, Karikari RA, Ma X, Mourão A, Herzig S. Orphan GPR116 mediates the insulin sensitizing effects of the hepatokine FNDC4 in adipose tissue. Nat Commun. 2021;12(1):2999. https://doi.org/10.1038/s41467-021-22579-1.
Hamann J, Aust G, Araç D, Engel FB, Formstone C, Fredriksson R, Schiöth HB. International Union of Basic and Clinical Pharmacology. XCIV. Adhesion G protein-coupled receptors. Pharmacol Rev. 2015;67(2):338–67. https://doi.org/10.1124/pr.114.009647.
Hauser AS, Attwood MM, Rask-Andersen M, Schiöth HB, Gloriam DE. Trends in GPCR drug discovery: new agents, targets and indications. Nat Rev Drug Discovery. 2017;16(12):829–42. https://doi.org/10.1038/nrd.2017.178.
Jiang X, Stockwell BR, Conrad M. Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol. 2021;22(4):266–82. https://doi.org/10.1038/s41580-020-00324-8.
Lelubre C, Vincent J-L. Mechanisms and treatment of organ failure in sepsis. Nat Rev Nephrol. 2018;14(7):417–27. https://doi.org/10.1038/s41581-018-0005-7.
Li J, Lu K, Sun F, Tan S, Zhang X, Sheng W, Han W. Panaxydol attenuates ferroptosis against LPS-induced acute lung injury in mice by Keap1-Nrf2/HO-1 pathway. J Trans Med. 2021;19(1):96. https://doi.org/10.1186/s12967-021-02745-1.
Li N, Wang W, Zhou H, Wu Q, Duan M, Liu C, Tang Q. Ferritinophagy-mediated ferroptosis is involved in sepsis-induced cardiac injury. Free Radic Biol Med. 2020;160:303–18. https://doi.org/10.1016/j.freeradbiomed.2020.08.009.
Liu J, Xia X, Huang P. xCT: a critical molecule that links cancer metabolism to redox signaling. Mol Ther. 2020;28(11):2358–66. https://doi.org/10.1016/j.ymthe.2020.08.021.
Liu P, Feng Y, Li H, Chen X, Wang G, Xu S, Zhao L. Ferrostatin-1 alleviates lipopolysaccharide-induced acute lung injury via inhibiting ferroptosis. Cell Mol Biol Lett. 2020;25:10. https://doi.org/10.1186/s11658-020-00205-0.
Luo L, Mo G, Huang D. Ferroptosis in hepatic ischemia‑reperfusion injury: regulatory mechanisms and new methods for therapy (Review). Mol Med Rep. 2021;23(3):225. https://doi.org/10.3892/mmr.2021.11864.
Miyata T, Nagy LE. Programmed cell death in alcohol-associated liver disease. Clin Mol Hepatol. 2020;26(4):618–25. https://doi.org/10.3350/cmh.2020.0142.
Niu B, Lei X, Xu Q, Ju Y, Xu D, Mao L, Li X. Protecting mitochondria via inhibiting VDAC1 oligomerization alleviates ferroptosis in acetaminophen-induced acute liver injury. Cell Biol Toxicol. 2022;38(3):505–30. https://doi.org/10.1007/s10565-021-09624-x.
Park SJ, Cho SS, Kim KM, Yang JH, Kim JH, Jeong EH, Ki SH. Protective effect of sestrin2 against iron overload and ferroptosis-induced liver injury. Toxicol Appl Pharmacol. 2019;379:114665. https://doi.org/10.1016/j.taap.2019.114665.
Prescott HC, Angus DC. Enhancing recovery from sepsis: a review. JAMA. 2018;319(1):62–75. https://doi.org/10.1001/jama.2017.17687.
Qiongyue Z, Xin Y, Meng P, Sulin M, Yanlin W, Xinyi L, Xuemin S. Post-treatment with irisin attenuates acute kidney injury in sepsis mice through anti-ferroptosis via the SIRT1/Nrf2 pathway. Front Pharmacol. 2022;13:857067. https://doi.org/10.3389/fphar.2022.857067.
Rudd KE, Johnson SC, Agesa KM, Shackelford KA, Tsoi D, Kievlan DR, Naghavi M. Global, regional, and national sepsis incidence and mortality, 1990–2017: analysis for the Global Burden of Disease Study. Lancet. 2020;395(10219):200–11. https://doi.org/10.1016/S0140-6736(19)32989-7.
Strnad P, Tacke F, Koch A, Trautwein C. Liver - guardian, modifier and target of sepsis. Nat Rev Gastroenterol Hepatol. 2017;14(1):55–66. https://doi.org/10.1038/nrgastro.2016.168.
Tang D, Chen X, Kang R, Kroemer G. Ferroptosis: molecular mechanisms and health implications. Cell Res. 2021;31(2):107–25. https://doi.org/10.1038/s41422-020-00441-1.
Tang D, Kang R, Berghe TV, Vandenabeele P, Kroemer G. The molecular machinery of regulated cell death. Cell Res. 2019;29(5):347–64. https://doi.org/10.1038/s41422-019-0164-5.
Wang D, Yin Y, Yao Y. Advances in sepsis-associated liver dysfunction. Burns Trauma. 2014;2(3):97–105. https://doi.org/10.4103/2321-3868.132689.
Wang J, Zhu Q, Li R, Zhang J, Ye X, Li X. YAP1 protects against septic liver injury via ferroptosis resistance. Cell Biosci. 2022;12(1):163. https://doi.org/10.1186/s13578-022-00902-7.
Wang T, Wang Y, Xiang Q, Lin S, Jin P, Wang J, Bian J. Deletion of G protein-coupled receptor 116 enhances neutrophil function and aggravates lung injury in mice. Genes Dis. 2022. https://doi.org/10.1016/j.gendis.2022.08.017.
Wang W, Qiao Y, Li Z. New insights into modes of GPCR activation. Trends Pharmacol Sci. 2018;39(4):367–86. https://doi.org/10.1016/j.tips.2018.01.001.
Wei S, Bi J, Yang L, Zhang J, Wan Y, Chen X, Wu R. Serum irisin levels are decreased in patients with sepsis, and exogenous irisin suppresses ferroptosis in the liver of septic mice. Clin Transl Med. 2020;10(5):e173. https://doi.org/10.1002/ctm2.173.
Wu J, Wang Y, Jiang R, Xue R, Yin X, Wu M, Meng Q. Ferroptosis in liver disease: new insights into disease mechanisms. Cell Death Discovery. 2021;7(1):276. https://doi.org/10.1038/s41420-021-00660-4.
Yan J, Li S, Li S. The role of the liver in sepsis. Int Rev Immunol. 2014;33(6):498–510. https://doi.org/10.3109/08830185.2014.889129.
Yang MY, Hilton MB, Seaman S, Haines DC, Nagashima K, Burks CM, St Croix B. Essential regulation of lung surfactant homeostasis by the orphan G protein-coupled receptor GPR116. Cell Rep. 2013;3(5):1457–64. https://doi.org/10.1016/j.celrep.2013.04.019.
Zaidman NA, Tomilin VN, HassanzadehKhayyat N, Damarla M, Tidmore J, Capen DE, Pluznick JL. Adhesion-GPCR Gpr116 (ADGRF5) expression inhibits renal acid secretion. Proc Nat Acad Sci USA. 2020;117(42):26470–81. https://doi.org/10.1073/pnas.2007620117.
Zhang H, Liu J, Zhou Y, Qu M, Wang Y, Guo K, Miao C. Neutrophil extracellular traps mediate m6A modification and regulates sepsis-associated acute lung injury by activating ferroptosis in alveolar epithelial cells. Int J Biol Sci. 2022;18(8):3337–57. https://doi.org/10.7150/ijbs.69141.
Zhang H, Zhang E, Hu H. Role of ferroptosis in non-alcoholic fatty liver disease and its implications for therapeutic strategies. Biomedicines. 2021;9(11). https://doi.org/10.3390/biomedicines9111660
Zhang YY, Ning BT. Signaling pathways and intervention therapies in sepsis. Signal Transduct Target Ther. 2021;6(1):407. https://doi.org/10.1038/s41392-021-00816-9.
Zhao Y, Li Y, Zhang R, Wang F, Wang T, Jiao Y. The role of erastin in ferroptosis and its prospects in cancer therapy. Onco Targets Ther. 2020;13:5429–41. https://doi.org/10.2147/OTT.S254995.
Zheng J, Conrad M. The metabolic underpinnings of ferroptosis. Cell Metab. 2020;32(6):920–37. https://doi.org/10.1016/j.cmet.2020.10.011.
Acknowledgements
Thanks to the partners for their contributions.
Funding
This work was supported by grants from the National Natural Science Foundation of China (81871579, 82272205, and 82072220).
Author information
Authors and Affiliations
Contributions
Ying Wang: design of experiments, experimental studies, data cleansing and analysis, edit the first draft and revision. Ting Wang, Qian Xiang: experimental studies and manuscript revision. Na Li, Jun Wang, Yan Zhang: experimental guidance and suggestions for improvement. Jiahao Liu: data analysis. Tao Yang, Jinjun Bian: experimental guidance and suggestions for improvement, manuscript revision.
Corresponding author
Ethics declarations
Ethics approval
All animal studies were approved by the Ethical Committee of Naval Medical University.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, Y., Wang, T., Xiang, Q. et al. GPR116 promotes ferroptosis in sepsis-induced liver injury by suppressing system Xc–/GSH/GPX4. Cell Biol Toxicol 39, 3015–3030 (2023). https://doi.org/10.1007/s10565-023-09815-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-023-09815-8