Abstract
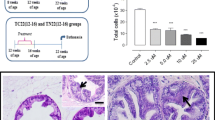
Delayed cancer progression in the ventral prostate of the Transgenic Adenocarcinoma of the Mouse Prostate (TRAMP) model has been previously reported upon celecoxib and nintedanib co-administration. Herein, we sought to further investigate the effects of these drugs association in some of their direct molecular targets (COX-2, VEGF and VEGFR-2) and in reactive stroma markers (TGF-β, αSMA, vimentin and pro-collagen 1) in the dorsolateral prostate, looking for lobe-specific responses. Male TRAMP mice were treated with celecoxib (10 mg/Kg, i.o.) and/or nintedanib (15 mg/Kg, i.o.) for 6 weeks and prostate was harvested for morphological and protein expression analyses. Results showed that combined therapy resulted in unique antitumor effects in dorsolateral prostate, especially due to the respective stromal or epithelial antiproliferative actions of these drugs, which altogether led to a complete inversion in high-grade (HGPIN) versus low-grade (LGPIN) premalignant lesion incidences in relation to controls. At the molecular level, this duality in drug action was paralleled by the differential down/upregulation of TGF-β signaling by celecoxib/nintedanib, thus leading to associated changes in stroma composition towards regression or quiescence, respectively. Additionally, combined therapy was able to promote decreased expression of inflammatory (COX-2) and angiogenesis (VEGF/VEGFR-2) mediators. Overall, celecoxib and nintedanib association provided enhanced antitumor effects in TRAMP dorsolateral as compared to former registers in ventral prostate, thus demonstrating lobe-specific responses of this combined chemoprevention approach. Among these responses, we highlight the ability in promoting TGF-β signaling and its associated stromal maturation/stabilization, thus yielding a more quiescent stromal milieu and resulting in greater epithelial proliferation impairment.








Similar content being viewed by others
References
Abdollahi A, Lipson KE, Sckell A et al (2003) Combined therapy with direct and indirect angiogenesis inhibition results in enhanced antiangiogenic and antitumor effects. Cancer Res 63:8890–8898
Ali A, Kulik G (2021) Signaling pathways that control apoptosis in prostate cancer. Cancers (basel). https://doi.org/10.3390/cancers13050937
Alves LF, da Silva RF, Cagnon VHA (2018) Nintedanib effects on delaying cancer progression and decreasing COX-2 and IL-17 in the prostate anterior lobe in TRAMP mice. Tissue Cell 50:96–103. https://doi.org/10.1016/j.tice.2017.12.008
Awasthi N, Schwarz RE (2015) Profile of nintedanib in the treatment of solid tumors: the evidence to date. Onco Targets Ther 8:3691–3701
Ayala G, Tuxhorn JA, Wheeler TM et al (2003) Reactive stroma as a predictor of biochemical-free recurrence in prostate cancer. Clin Cancer Res 9:4792–4801
Battegay EJ, Raines EW, Seifert RA et al (1990) TGF-β induces bimodal proliferation of connective tissue cells via complex control of an autocrine PDGF loop. Cell 63:515–524. https://doi.org/10.1016/0092-8674(90)90448-N
Benjamin LE, Golijanin D, Itin A et al (1999) Selective ablation of immature blood vessels in established human tumors follows vascular endothelial growth factor withdrawal. J Clin Invest 103:159–165. https://doi.org/10.1172/JCI5028
Bergers G, Javaherian K, Lo K-M et al (1999) Effects of angiogenesis inhibitors on multistage carcinogenesis in mice. Science (1979) 284:808–812. https://doi.org/10.1126/science.284.5415.808
Berman-Booty LD, Sargeant AM, Rosol TJ et al (2011) A review of the existing grading schemes and a proposal for a modified grading scheme for prostatic lesions in TRAMP mice. Toxicol Pathol 40:5–17. https://doi.org/10.1177/0192623311425062
Bhowmick NA, Chytil A, Plieth D et al (2004) TGF-ß signaling in fibroblasts modulates the oncogenic potential of adjacent epithelia. Science (1979) 303:848–851. https://doi.org/10.1126/science.1090922
Bicaklioglu G, Pirhan D, Yazir Y et al (2022) Evaluation of nintedanib as a new postoperative antiscarring agent in experimental extraocular muscle surgery. Int J Ophthalmol 15:914–923
Bill R, Fagiani E, Zumsteg A et al (2015) Nintedanib is a highly effective therapeutic for neuroendocrine carcinoma of the pancreas (PNET) in the Rip1Tag2 transgenic mouse model. Clin Cancer Res 21:4856–4867. https://doi.org/10.1158/1078-0432.CCR-14-3036
Björkerud S (1991) Effects of transforming growth factor-beta 1 on human arterial smooth muscle cells in vitro. Arterioscler Thromb 11:892–902. https://doi.org/10.1161/01.ATV.11.4.892
Brizzolara A, Benelli R, Venè R et al (2017) The ErbB family and androgen receptor signaling are targets of Celecoxib in prostate cancer. Cancer Lett 400:9–17. https://doi.org/10.1016/j.canlet.2017.04.025
Cai J, Jiang WG, Mansel RE (1999) Phosphorylation and disorganization of vascular-endothelial Cadherin in interaction between breast cancer and vascular endothelial cells
Cândido EM, Fávaro WJ, Montico F et al (2012) Senescence and steroid hormone receptor reactivities in accessory sex glands of elderly rats (Sprague-Dawley) following exogenous hormonal therapy. Tissue Cell 44:227–237. https://doi.org/10.1016/j.tice.2012.03.007
Carmeliet P, Jain RK (2000) Angiogenesis in cancer and other diseases. Nature 407:249–257. https://doi.org/10.1038/35025220
Cha B-K, Kim Y-S, Hwang K-E et al (2016) Celecoxib and sulindac inhibit TGF-β1-induced epithelial-mesenchymal transition and suppress lung cancer migration and invasion via downregulation of sirtuin 1. Oncotarget 7:57213–57227
Chávez E, Segovia J, Shibayama M et al (2010) Antifibrotic and fibrolytic properties of celecoxib in liver damage induced by carbon tetrachloride in the rat. Liver Int 30:969–978. https://doi.org/10.1111/j.1478-3231.2010.02256.x
Chen H, Qian Z, Zhang S et al (2021) Silencing COX-2 blocks PDK1/TRAF4-induced AKT activation to inhibit fibrogenesis during skeletal muscle atrophy. Redox Biol 38:101774. https://doi.org/10.1016/j.redox.2020.101774
Chiaverotti T, Couto SS, Donjacour A et al (2008) Dissociation of epithelial and neuroendocrine carcinoma lineages in the transgenic adenocarcinoma of mouse prostate model of prostate cancer. Am J Pathol 172:236–246. https://doi.org/10.2353/ajpath.2008.070602
Chipuk JE, Cornelius SC, Pultz NJ et al (2002) The androgen receptor represses transforming growth factor-β Signaling through interaction with Smad3 *. J Biol Chem 277:1240–1248. https://doi.org/10.1074/jbc.M108855200
Cogan JG, Strauch AR, Sun S et al (1995) Plasticity of vascular smooth muscle alpha-actin gene transcription: characterization of multiple, single-, and double-strand specific DNA-binding proteins in myoblasts and fibroblasts. J Biol Chem 270:11310–11321. https://doi.org/10.1074/jbc.270.19.11310
Cutolo M, Gotelli E, Montagna P et al (2021) Nintedanib downregulates the transition of cultured systemic sclerosis fibrocytes into myofibroblasts and their pro-fibrotic activity. Arthritis Res Ther 23:205. https://doi.org/10.1186/s13075-021-02555-2
da Silva RF, Nogueira-Pangrazi E, Kido LA et al (2017) Nintedanib antiangiogenic inhibitor effectiveness in delaying adenocarcinoma progression in Transgenic Adenocarcinoma of the Mouse Prostate (TRAMP). J Biomed Sci 24:31. https://doi.org/10.1186/s12929-017-0334-z
da Silva RF, Dhar D, Raina K et al (2018a) Nintedanib inhibits growth of human prostate carcinoma cells by modulating both cell cycle and angiogenesis regulators. Sci Rep 8:9540. https://doi.org/10.1038/s41598-018-27831-1
da Silva RF, Banzato TP, Alves LF et al (2020) Antiangiogenic therapy with Nintedanib affects hypoxia, angiogenesis and apoptosis in the ventral prostate of TRAMP animals. Cell Tissue Res 379:407–420. https://doi.org/10.1007/s00441-019-03091-x
Dallas SL, Zhao S, Cramer SD et al (2005) Preferential production of latent transforming growth factor β-2 by primary prostatic epithelial cells and its activation by prostate-specific antigen. J Cell Physiol 202:361–370. https://doi.org/10.1002/jcp.20147
Danielpour D (2005) Functions and regulation of transforming growth factor-beta (TGF-β) in the prostate. Eur J Cancer 41:846–857. https://doi.org/10.1016/j.ejca.2004.12.027
de Marzo AM, Platz EA, Sutcliffe S et al (2007) Inflammation in prostate carcinogenesis. Nat Rev Cancer 7:256–269. https://doi.org/10.1038/nrc2090
Dermond O, Rüegg C (2001) Inhibition of tumor angiogenesis by non-steroidal anti-inflammatory drugs: emerging mechanisms and therapeutic perspectives. Drug Resist Updates 4:314–321. https://doi.org/10.1054/drup.2001.0219
Epstein Shochet G, Wollin L, Shitrit D (2018) Fibroblast–matrix interplay: Nintedanib and pirfenidone modulate the effect of IPF fibroblast-conditioned matrix on normal fibroblast phenotype. Respirology 23:756–763. https://doi.org/10.1111/resp.13287
Esser S, Lampugnani MG, Corada M et al (1998) Vascular endothelial growth factor induces VE-cadherin tyrosine phosphorylation in endothelial cells. J Cell Sci 111:1853–1865. https://doi.org/10.1242/jcs.111.13.1853
Folkman J (2006) Angiogenesis. Annu Rev Med 57:1–18. https://doi.org/10.1146/annurev.med.57.121304.131306
Funahashi Y, O’Malley KJ, Kawamorita N et al (2014) Upregulation of androgen-responsive genes and transforming growth factor-β1 cascade genes in a rat model of non-bacterial prostatic inflammation. Prostate 74:337–345. https://doi.org/10.1002/pros.22668
Funahashi Y, Wang Z, O’Malley KJ et al (2015) Influence of E. coli-induced prostatic inflammation on expression of androgen-responsive genes and transforming growth factor beta 1 cascade genes in rats. Prostate 75:381–389. https://doi.org/10.1002/pros.22924
Gaengel K, Genové G, Armulik A, Betsholtz C (2009) Endothelial-mural cell signaling in vascular development and angiogenesis. Arterioscler Thromb Vasc Biol 29:630–638. https://doi.org/10.1161/ATVBAHA.107.161521
Gerdes MJ, Dang TD, Larsen M, Rowley DR (1998) Transforming growth factor-β1 induces nuclear to cytoplasmic distribution of androgen receptor and inhibits androgen response in prostate smooth muscle cells*. Endocrinology 139:3569–3577. https://doi.org/10.1210/endo.139.8.6138
Gingrich JR, Barrios RJ, Foster BA, Greenberg NM (1999) Pathologic progression of autochthonous prostate cancer in the TRAMP model. Prostate Cancer Prostatic Dis 2:70–75. https://doi.org/10.1038/sj.pcan.4500296
Greenberg JI, Cheresh DA (2009) VEGF as an inhibitor of tumor vessel maturation: implications for cancer therapy. Expert Opin Biol Ther 9:1347–1356. https://doi.org/10.1517/14712590903208883
Greenberg NM, DeMayo F, Finegold MJ et al (1995) Prostate cancer in a transgenic mouse. Proc Natl Acad Sci 92:3439–3443. https://doi.org/10.1073/pnas.92.8.3439
Greenberg JI, Shields DJ, Barillas SG et al (2008) A role for VEGF as a negative regulator of pericyte function and vessel maturation. Nature 456:809–813. https://doi.org/10.1038/nature07424
Grivas N, Goussia A, Stefanou D, Giannakis D (2016) Microvascular density and immunohistochemical expression of VEGF, VEGFR-1 and VEGFR-2 in benign prostatic hyperplasia, high-grade prostate intraepithelial neoplasia and prostate cancer. Cent Eur J Urol 69:63–71. https://doi.org/10.5173/ceju.2016.726
Guo Y, Kyprianou N (1999) Restoration of transforming growth factor β signaling pathway in human prostate cancer cells suppresses tumorigenicity via induction of caspase-1-mediated apoptosis1. Cancer Res 59:1366–1371
Gupta S, Adhami VM, Subbarayan M et al (2004) Suppression of prostate carcinogenesis by dietary supplementation of celecoxib in transgenic adenocarcinoma of the mouse prostate model. Cancer Res 64:3334–3343. https://doi.org/10.1158/0008-5472.CAN-03-2422
Hilberg F, Roth GJ, Krssak M et al (2008) BIBF 1120: triple angiokinase inhibitor with sustained receptor blockade and good antitumor efficacy. Cancer Res 68:4774–4782. https://doi.org/10.1158/0008-5472.CAN-07-6307
Hisataki T, Itoh N, Suzuki K et al (2004) Modulation of phenotype of human prostatic stromal cells by transforming growth factor-betas. Prostate 58:174–182. https://doi.org/10.1002/pros.10320
Hofmann TG, Stollberg N, Schmitz ML, Will H (2003) HIPK2 regulates transforming growth factor-β-induced c-Jun NH2-terminal kinase activation and apoptosis in human hepatoma cells. Cancer Res 63:8271–8277
Huss WJ, Barrios RJ, Greenberg NM (2003) SU5416 Selectively Impairs Angiogenesis to Induce Prostate Cancer-specific Apoptosis1. Mol Cancer Ther 2:611–616
Ito C, Akimoto T, Ioka T et al (2009) TGF-beta inhibits vascular sprouting through TGF-beta; type I receptor in the mouse embryonic aorta. Tohoku J Exp Med 218:63–71. https://doi.org/10.1620/tjem.218.63
Ittmann M (2018) Anatomy and histology of the human and murine prostate. Cold Spring Harb Perspect Med. https://doi.org/10.1101/cshperspect.a030346
Kaplan-LPJ C-M, Ittmann MM et al (2003) Pathobiology of autochthonous prostate cancer in a pre-clinical transgenic mouse model. Prostate 55:219–237. https://doi.org/10.1002/pros.10215
Kido LA, Hetzl AC, Cândido EM et al (2014) Antiangiogenic and finasteride therapies: Responses of the prostate microenvironment in elderly mice. Life Sci 106:58–70. https://doi.org/10.1016/j.lfs.2014.04.027
Kido LA, Montico F, Sauce R et al (2016) Anti-inflammatory therapies in TRAMP mice: delay in PCa progression. Endocr Relat Cancer 23:235–250. https://doi.org/10.1530/ERC-15-0540
Kim BH, Kim C, Chang HS et al (2011) Cyclooxygenase-2 Overexpression in Chronic Inflammation Associated with Benign Prostatic Hyperplasia: Is It Related to Apoptosis and Angiogenesis of Prostate Cancer? Korean J Urol 52:253–259
Kumari J, Wagener FADTG, Kouwer PHJ (2022) Novel synthetic polymer-based 3D contraction assay: a versatile preclinical research platform for fibrosis. ACS Appl Mater Interfaces 14:19212–19225. https://doi.org/10.1021/acsami.2c02549
le Guelte A, Dwyer J, Gavard J (2011) Jumping the barrier: VE-cadherin, VEGF and other angiogenic modifiers in cancer. Biol Cell 103:593–605. https://doi.org/10.1042/BC20110069
Lin X, Wen J, Liu R et al (2018) Nintedanib inhibits TGF-β-induced myofibroblast transdifferentiation in human Tenon’s fibroblasts. Mol vis 24:789–800
Lucia MS, Sporn MB, Roberts AB, et al (1998) The role of transforming growth factor-β1, -β2, and -β3 in androgen-responsive growth of NRP-152 rat prostatic epithelial cells. J Cell Physiol 175:184–192. https://doi.org/10.1002/(SICI)1097-4652(199805)175:2<184::AID-JCP8>3.0.CO;2-K
Majack RA, Majesky MW, Goodman LV (1990) Role of PDGF-A expression in the control of vascular smooth muscle cell growth by transforming growth factor-beta. J Cell Biol 111:239–247. https://doi.org/10.1083/jcb.111.1.239
Masferrer JL, Leahy KM, Koki AT et al (2000) Antiangiogenic and Antitumor Activities of Cyclooxygenase-2 Inhibitors. Cancer Res 60:1306–1311
Mateus PAM, Kido LA, Silva RS et al (2019) Association of anti-inflammatory and antiangiogenic therapies negatively influences prostate cancer progression in TRAMP mice. Prostate 79:515–535. https://doi.org/10.1002/pros.23758
Miki T, Nonomura N, Nozawa M et al (1998) Angiogenesis inhibitor TNP-470 inhibits growth and metastasis of a hormone-independent rat prostatic carcinoma cell line. J Urol 160:210–213. https://doi.org/10.1016/S0022-5347(01)63092-1
Montgomery DC (1991) Design and analysis of experiments, 3rd edn. John Wiley & Sons Inc, New York
Montico F, Kido LA, Hetzl AC, Cagnon VHA (2015a) Prostatic angiogenic responses in late life: Antiangiogenic therapy influences and relation with the glandular microenvironment in the transgenic adenocarcinoma of mouse prostate (TRAMP) model. Prostate 75:484–499. https://doi.org/10.1002/pros.22934
Montico F, Kido LA, San Martin R et al (2015b) Reactive stroma in the prostate during late life: the role of microvasculature and antiangiogenic therapy influences. Prostate 75:1643–1661. https://doi.org/10.1002/pros.23045
Motohiko S, Yoshinobu O, Katsuyuki T (1995) Transforming growth factor-β 1 proliferated vascular smooth muscle cells from spontaneously hypertensive rats. Am J Hypertens 8:160–166. https://doi.org/10.1016/0895-7061(94)00191-D
Mukherjee R, Edwards J, Underwood MA, Bartlett JMS (2005) The relationship between angiogenesis and cyclooxygenase-2 expression in prostate cancer. BJU Int 96:62–66. https://doi.org/10.1111/j.1464-410X.2005.05568.x
Nakai K, Tanaka T, Murai T et al (2005) Invasive human pancreatic carcinoma cells adhere to endothelial tri-cellular corners and increase endothelial permeability. Cancer Sci 96:766–773. https://doi.org/10.1111/j.1349-7006.2005.00102.x
Nogueira PE, da Silva RF, Kido LA et al (2018) Nintedanib treatment delays prostate dorsolateral lobe cancer progression in the TRAMP model: contribution to the epithelial-stromal interaction balance. Cell Biol Int 42:153–168. https://doi.org/10.1002/cbin.10881
Oliveira DSM, Dzinic S, Bonfil AI et al (2016) The mouse prostate: a basic anatomical and histological guideline. Bosn J Basic Med Sci 16:8–13. https://doi.org/10.17305/bjbms.2016.917
Patel MI, Subbaramaiah K, Du B et al (2005) Celecoxib inhibits prostate cancer growth: evidence of a cyclooxygenase-2-independent mechanism. Clin Cancer Res 11:1999–2007. https://doi.org/10.1158/1078-0432.CCR-04-1877
Peehl DM, Sellers RG (1997) Induction of Smooth Muscle Cell Phenotype in Cultured Human Prostatic Stromal Cells. Exp Cell Res 232:208–215. https://doi.org/10.1006/excr.1997.3525
Peng Y-C, Levine CM, Zahid S et al (2013) Sonic hedgehog signals to multiple prostate stromal stem cells that replenish distinct stromal subtypes during regeneration. Proc Natl Acad Sci 110:20611–20616. https://doi.org/10.1073/pnas.1315729110
Prins GS (1989) Differential regulation of androgen receptors in the separate rat prostate lobes: androgen independent expression in the lateral lobe. J Steroid Biochem 33:319–326. https://doi.org/10.1016/0022-4731(89)90319-1
Prins GS, Birch L (1993) Immunocytochemical analysis of androgen receptor along the ducts of the separate rat prostate lobes after androgen withdrawal and replacement. Endocrinology 132:169–178. https://doi.org/10.1210/endo.132.1.8419121
Roach KM, Castells E, Dixon K, et al (2021) Evaluation of Pirfenidone and Nintedanib in a Human Lung Model of Fibrogenesis. Front Pharmacol 12:
Romero-Calvo I, Ocón B, Martínez-Moya P et al (2010) Reversible Ponceau staining as a loading control alternative to actin in Western blots. Anal Biochem 401:318–320. https://doi.org/10.1016/j.ab.2010.02.036
Rønnow SR, Dabbagh RQ, Genovese F et al (2020) Prolonged Scar-in-a-Jar: an in vitro screening tool for anti-fibrotic therapies using biomarkers of extracellular matrix synthesis. Respir Res 21:108. https://doi.org/10.1186/s12931-020-01369-1
Rowley DR (2014) Reprogramming the tumor stroma: a new paradigm. Cancer Cell 26:451–452. https://doi.org/10.1016/j.ccell.2014.09.016
Roy-Burman P, Wu H, Powell WC et al (2004) Genetically defined mouse models that mimic natural aspects of human prostate cancer development. Endocrine-Relat Cancer Endocr Relat Cancer Endocr Relat Cancer 11:225–254. https://doi.org/10.1677/erc.0.0110225
Saji S, Hirose M, Toi M (2004) Novel sensitizing agents: Potential Contribution of COX-2 inhibitor for endocrine therapy of breast cancer. Breast Cancer 11:129–133. https://doi.org/10.1007/BF02968291
Salm SN, Koikawa Y, Ogilvie V, et al (2000) Transforming growth factor-β is an autocrine mitogen for a novel androgen-responsive murine prostatic smooth muscle cell line, PSMC1. J Cell Physiol 185:416–424. https://doi.org/10.1002/1097-4652(200012)185:3<416::AID-JCP12>3.0.CO;2-Z
Satchi-Fainaro R, Mamluk R, Wang L et al (2005) Inhibition of vessel permeability by TNP-470 and its polymer conjugate, caplostatin. Cancer Cell 7:251–261. https://doi.org/10.1016/j.ccr.2005.02.007
Sherman MH, Yu RT, Engle DD et al (2014) Vitamin D receptor-mediated stromal reprogramming suppresses pancreatitis and enhances pancreatic cancer therapy. Cell 159:80–93. https://doi.org/10.1016/j.cell.2014.08.007
Silva RS, Kido LA, Montico F et al (2018b) Steroidal hormone and morphological responses in the prostate anterior lobe in different cancer grades after Celecoxib and Goniothalamin treatments in TRAMP mice. Cell Biol Int 42:1006–1020. https://doi.org/10.1002/cbin.10967
Sinha R, Cooper TK, Rogers CJ et al (2014) Dietary methionine restriction inhibits prostatic intraepithelial neoplasia in TRAMP mice. Prostate 74:1663–1673. https://doi.org/10.1002/pros.22884
Stouffer GA, Owens GK (1994) TGF-beta promotes proliferation of cultured SMC via both PDGF-AA-dependent and PDGF-AA-independent mechanisms. J Clin Invest 93:2048–2055. https://doi.org/10.1172/JCI117199
Tang S-N, Zhang J, Wu W et al (2015) Chemopreventive effects of Korean angelica versus its major pyranocoumarins on two lineages of transgenic adenocarcinoma of mouse prostate carcinogenesis. Cancer Prev Res 8:835–844. https://doi.org/10.1158/1940-6207.CAPR-15-0051
Tang S-N, Zhang J, Jiang P et al (2016) Gene expression signatures associated with suppression of TRAMP prostate carcinogenesis by a kavalactone-rich Kava fraction. Mol Carcinog 55:2291–2303. https://doi.org/10.1002/mc.22469
Tkacz VL, Tohnya TM, Figg William II (2005) Cyclooxygenase-2 and angiogenesis in prostate cancer. Cancer Biol Ther 4:813–814. https://doi.org/10.4161/cbt.4.8.2089
Tuxhorn JA, Ayala GE, Rowley DR (2001) Reactive stroma in prostate cancer progression. J Urol 166:2472–2483. https://doi.org/10.1016/S0022-5347(05)65620-0
Tuxhorn JA, Ayala GE, Smith MJ et al (2002) Reactive stroma in human prostate cancer: induction of myofibroblast phenotype and extracellular matrix remodeling1. Clin Cancer Res 8:2912–2923
Viallard C, Larrivée B (2017) Tumor angiogenesis and vascular normalization: alternative therapeutic targets. Angiogenesis 20:409–426. https://doi.org/10.1007/s10456-017-9562-9
Wallez Y, Vilgrain I, Huber P (2006) Angiogenesis: the VE-Cadherin Switch. Trends Cardiovasc Med 16:55–59. https://doi.org/10.1016/j.tcm.2005.11.008
Wang W, Bergh A, Damber J-E (2005) Cyclooxygenase-2 expression correlates with local chronic inflammation and tumor neovascularization in human prostate cancer. Clin Cancer Res 11:3250–3256. https://doi.org/10.1158/1078-0432.CCR-04-2405
Wang L, Zhang J, Zhang Y et al (2011) Lobe-specific lineages of carcinogenesis in the transgenic adenocarcinoma of mouse prostate and their responses to chemopreventive selenium. Prostate 71:1429–1440. https://doi.org/10.1002/pros.21360
Wang Z, Dabrosin C, Yin X et al (2015) Broad targeting of angiogenesis for cancer prevention and therapy. Semin Cancer Biol 35:S224–S243. https://doi.org/10.1016/j.semcancer.2015.01.001
Wang Y, Chen J, Wu Z et al (2021) Mechanisms of enzalutamide resistance in castration-resistant prostate cancer and therapeutic strategies to overcome it. Br J Pharmacol 178:239–261. https://doi.org/10.1111/bph.15300
Weibel ER (1963) Principles and methods for the morphometric study of the lung and other organs. Lab Invest 12:131–155
Weidner N, Carroll PR, Flax J et al (1993) Tumor angiogenesis correlates with metastasis in invasive prostate carcinoma. Am J Pathol 143:401–409
Weißhardt P, Trarbach T, Dürig J et al (2012) Tumor vessel stabilization and remodeling by anti-angiogenic therapy with bevacizumab. Histochem Cell Biol 137:391–401. https://doi.org/10.1007/s00418-011-0898-8
Wollin L, Maillet I, Quesniaux V et al (2014) Antifibrotic and anti-inflammatory activity of the tyrosine kinase inhibitor nintedanib in experimental models of lung fibrosis. J Pharmacol Exp Ther 349:209. https://doi.org/10.1124/jpet.113.208223
Yamanaka T, Harimoto N, Yokobori T et al (2020) Nintedanib inhibits intrahepatic cholangiocarcinoma aggressiveness via suppression of cytokines extracted from activated cancer-associated fibroblasts. Br J Cancer 122:986–994. https://doi.org/10.1038/s41416-020-0744-7
Yamaoka M, Yamamoto T, Ikeyama S et al (1993) Angiogenesis inhibitor TNP-470 (AGM-1470) potently inhibits the tumor growth of hormone-independent human breast and prostate carcinoma cell lines. Cancer Res 53:5233–5236
Yamashita S (2004) Localization of estrogen and androgen receptors in male reproductive tissues of mice and rats. Anat Rec A Discov Mol Cell Evol Biol 279A:768–778. https://doi.org/10.1002/ar.a.20061
Yonenaga Y, Mori A, Onodera H et al (2005) Absence of smooth muscle actin-positive pericyte coverage of tumor vessels correlates with hematogenous metastasis and prognosis of colorectal cancer patients. Oncology 69:159–166. https://doi.org/10.1159/000087840
Yoshimura R, Sano H, Masuda C, et al (2000) Expression of cyclooxygenase-2 in prostate carcinoma. Cancer 89:589–596. https://doi.org/10.1002/1097-0142(20000801)89:3<589::AID-CNCR14>3.0.CO;2-C
Zar JH (1999) Biostatistical analysis, 4th edn. Prentice Hall Upper, New Jersey
Zhang J, Wang L, Zhang Y, et al (2011) Lobe-specific proteome changes in the dorsal-lateral and ventral prostate of TRAMP mice versus wild-type mice. Proteomics 11:2542–2549. https://doi.org/10.1002/pmic.201100008
Acknowledgements
We thank The São Paulo Research Foundation (FAPESP) for the financial support provided for this study (Grant: 2018/16299-9).
Funding
Fundação de Amparo à Pesquisa do Estado de São Paulo, 2018/16299-9.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest that could be perceived as prejudicing the impartiality of the research reported.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Montico, F., Lamas, C.d., Rossetto, I.M.U. et al. Lobe-specific responses of TRAMP mice dorsolateral prostate following celecoxib and nintedanib therapy. J Mol Histol 54, 379–403 (2023). https://doi.org/10.1007/s10735-023-10130-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-023-10130-z