Abstract
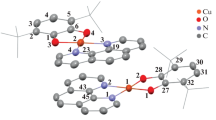
New heteroleptic zinc catecholate complexes based on 3,6-di-tert-butyl-o-benzoquinone and containing metal-coordinated N-donor ligands (2,2'-bipyridine and phenanthroline) were prepared by ligand exchange. According to X-ray diffraction data, both complexes were dimers with multiple intermolecular π–π interactions between the aromatic moieties of neighboring molecules (CCDC nos. 2222704 (I), 2222705 (II)). The electronic transmission spectra of crystalline samples of I and II and their solutions show broad absorption bands in the visible region with a maximum at about 500 nm.





Similar content being viewed by others
REFERENCES
Weinstein, J.A., Tierney, M.T., Davies, E.S., et al., Inorg. Chem., 2006, vol. 45, p. 4544.
Lu, X., Lee, S., Hong, Y., et al., J. Am. Chem. Soc., 2017, vol. 139, p. 13173.
Cai, K., Xie, J., and Zhao, D., J. Am. Chem. Soc., 2014, vol. 136, p. 28.
Pan, Z., Zhao, K., Wang, J., et al., ACS Nano, 2013, vol. 7, p. 5215.
Cui, B.-B., Zhong, Y.-W., and Yao, J., J. Am. Chem. Soc., 2015, vol. 137, p. 4058.
Cui, B.-B., Tang, J.-H., Yao, J., et al., Angew. Chem. Int. Ed., 2015, vol. 54, p. 9192.
Cameron, L.A., Ziller, J.W., and Heyduk, A.F., Chem. Sci., 2016, vol. 7, p. 1807.
Espa, D., Pilia, L., Marchio, L., et al., Dalton Trans., 2013, vol. 42, p. 12429.
Liu, Y., Zhang, Z., Chen, X., et al., Dyes Pigments, 2016, vol. 128, p. 179.
Wong, J.L., Higgins, R.F., Bhowmick, I., et al., Chem. Sci., 2016, vol. 7, p. 1594.
Kramer, W.W., Cameron, L.A., Zarkesh, R.A., et al., Inorg. Chem., 2014, vol. 53, p. 8825.
Ershova, I.V., Piskunov, A.V., and Cherkasov, V.K., Russ. Chem. Rev., 2020, vol. 89, p. 1157.
Pierpont, C.G., Coord. Chem. Rev., 2001, vols. 219–221, p. 415.
Abakumov, G.A., Piskunov, A.V., Cherkasov, V.K., et al., Russ. Chem. Rev., 2018, vol. 87, p. 393.
Rajput, A., Sharma, A.K., Barman, S.K., et al., Coord. Chem. Rev., 2020, vol. 414, p. 213240.
Pashanova, K.I., Poddel’sky, A.I., and Piskunov, A.V., Coord. Chem. Rev., 2022, vol. 459, p. 214399.
Kaim, W., Das, A., Fiedler, J., et al., Coord. Chem. Rev., 2020, vol. 404, p. 213114.
Dunn, T.J., Chiang, L., Ramogida, C.F., et al., Chem.-Eur. J., 2013, vol. 19, p. 9606.
Chiang, L., Kochem, A., Jarjayes, O., et al., Chem.-Eur. J., 2012, vol. 18, p. 14117.
Chiang, L., Herasymchuk, K., Thomas, F., et al., Inorg. Chem., 2015, vol. 54, p. 5970.
Storr, T., Wasinger, E.C., Pratt, R.C., et al., Angew. Chem. Int. Ed., 2007, vol. 46, p. 5198.
Kurahashi, T. and Fujii, H., J. Am. Chem. Soc., 2011, vol. 133, p. 8307.
Aono, S., Nakagaki, M., Kurahashi, T., et al., J. Chem. Theory Comput., 2014, vol. 10, p. 1062.
Kochem, A., Gellon, G., Leconte, N., et al., Chem.- Eur. J., 2013, vol. 19, p. 16707.
Clarke, R.M., Jeen, T., Rigo, S., et al., Chem. Sci., 2018, vol. 9, p. 1610.
Ward, M.D., J. Solid State Electrochem., 2005, vol. 9, p. 778.
Pashanova, K.I., Bitkina, V.O., Yakushev, I.A., et al., Molecules, 2021, vol. 26, p. 4622.
Pashanova, K.I., Ershova, I.V., Trofimova, O.Y., et al., Molecules, 2022, vol. 27.
Clarke, R.M., Hazin, K., Thompson, J.R., et al., Inorg. Chem., 2016, vol. 55, p. 762.
Lecarme, L., Chiang, L., Moutet, J., et al., Dalton Trans., 2016, vol. 45, p. 16325.
Yang, J., Kersi, D.K., Giles, L.J., et al., Inorg. Chem., 2014, vol. 5, p. 4791.
Rauth, G.K., Pal, S., Das, D., et al., Polyhedron, 2001, vol. 20, p. 363.
Heinze, K. and Reinhardt, S., Chem.-Eur. J., 2008, vol. 14, p. 9482.
Deibel, N., Schweinfurth, D., Fiedler, J., et al., Dalton Trans., 2011, vol. 40, p. 9925.
Scattergood, P.A., Jesus, P., Adams, H., et al., Dalton Trans., 2015, vol. 44, p. 11705.
Best, J., Sazanovich, I.V., Adams, H., et al., Inorg. Chem., 2010, vol. 49, p. 10041.
Roy, R., Chattopadhyay, P., and Sinha, C., Polyhedron, 1996, vol. 15, p. 3361.
Tahara, K., Kadowaki, T., Kikuchi, J., et al., Bull. Chem. Soc. Jpn., 2018, vol. 91, p. 1630.
Sobottka, S., Noßler, M., Ostericher, A.L., et al., Chem.-Eur. J., 2020, vol. 26, p. 1314.
Maleeva, A.V., Ershova, I.V., Trofimova, O.Y., et al., Mendeleev Commun., 2022, vol. 32, p. 83.
Ershova, I.V., Maleeva, A.V., Aysin, I.A., et al., Russ. Chem. Bull., 2023, vol. 72, p. 193.
Maleeva, A.V., Trofimova, O.Y., Ershova, I.V., et al., Russ. Chem. Bull., 2022, vol. 71, p. 1441.
Perrin, D.D., Armarego, W.L.F., and Perrin, D.R., Purification of Laboratory Chemicals, Oxford: Pergamon, 1980, p. 544.
Garnov, V.A., Nevodchikov, V.I., Abakumova, L.G., et al., Bull. Acad. Sci. USSR, 1987, vol. 36, p. 1728.
Piskunov, A.V., Maleeva, A.V., Abakumov, G.A., et al., Russ. J. Coord. Chem., 2011, vol. 37, p. 243.
APEX3, SAINT and SADABS, Madison: Bruker AXS Inc., 2016.
Krause, L., Herbst-Irmer, R., Sheldrick, G.M., et al., J. Appl. Crystallogr., 2015, vol. 48, p. 3.
Sheldrick, G.M., Acta Crystallogr., Sect. A: Found. Adv., 2015, vol. 71, p. 3.
Sheldrick, G.M., Acta Crystallogr., Sect. C: Struct. Chem., 2015, vol. 71, p. 3.
Dolomanov, O.V., Bourhis, L.J., Gildea, R.J., et al., J. Appl. Crystallogr., 2009, vol. 42, p. 339.
Frisch, M.J., Trucks, G.W., Schlegel, H.B., et al., Gaussian 09, Wallingford (CT): Revision D.01. Inc., 2013.
Yanai, T., Tew, D.P., and Handy, N.C., Chem. Phys. Lett., 2004, vol. 393, p. 51.
Loos, P., Comin, M., Blase, X., and Jacquemin, D., J. Chem. Theory Comput., 2021, p. 3666.
Butler, I.S., Gilson, D.F.R., Jean-Claude, B.J., et al., Inorg. Chim. Acta, 2014, vol. 423, p. 132.
Piskunov, A.V., Lado, A.V., Abakumov, G.A., et al., Russ. Chem. Bull., 2007, vol. 56, p. 97.
Piskunov, A.V., Maleeva, A.V., Mescheryakova, I.N., et al., Eur. J. Inorg. Chem., 2012, vol. 2012, p. 4318.
Piskunov, A.V., Lado, A.V., Fukin, G.K., et al., Heteroatom. Chem., 2006, vol. 17, p. 481.
Abakumov, G.A., Cherkasov, V.K., Piskunov, A.V., et al., Russ. Chem. Bull., 2006, vol. 55, p. 1146.
Piskunov, A.V., Maleeva, A.V., Bogomyakov, A.S., et al., Polyhedron, 2015, vol. 102, p. 715.
Chegerev, M.G., Piskunov, A.V., Maleeva, A.V., et al., Eur. J. Inorg. Chem., 2016, vol. 2016, p. 3813.
Brown, S.N., Inorg. Chem., 2012, vol. 51, p. 1251.
Wang, Q.-H., Long, D.-L., Hu, H.-M., et al., J. Coord. Chem., 2000, vol. 49, p. 201.
ACKNOWLEDGMENTS
This study was performed using the equipment of the Center for Collective Use, Analytical Center of the Razuvaev Institute of Organometallic Chemistry, Russian Academy of Sciences, supported by the grant “Provision of the Development of the Material and Technical Infrastructure of the Centers for the Collective Use of Research Equipment” (unique identifier RF–2296.61321X0017, contract no. 075-15-2021-670). X-ray diffraction study of complexes I and II was carried out using the equipment of the Center for Collective Use of Physical Investigation Methods of the Kurnakov Institute of General and Inorganic Chemistry, Russian Academy of Sciences.
Funding
This study was supported by the Russian Science Foundation (grant no. 22-13-00351).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by Z. Svitanko
Rights and permissions
About this article
Cite this article
Maleeva, A.V., Trofimova, O.Y., Yakushev, I.A. et al. Heteroleptic Zinc Catecholate Complexes with N-Donor Ligands. Russ J Coord Chem 49, 420–428 (2023). https://doi.org/10.1134/S1070328423600134
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1070328423600134