Abstract
Purpose of Review
As litter decomposition is a fundamental process in forest ecosystems, representing the link between aboveground and belowground biogeochemical processes, we developed an effect size summarisation of the implications of forest management on litter decomposition rate, by applying a multi-level meta-analysis and multivariate mixed-effects meta-analytic linear models. Our aim was to review the findings of the current literature and to understand how forest management, silvicultural treatment, and forest operations could affect litter decomposition rate. Furthermore, we investigated the effects of environmental variables that included stand type, climatic conditions, and the percentage of biomass removal on litter decomposition rate.
Recent Findings
We found a statistically significant reduction in litter decomposition rate associated with clear-cutting, and no statistically significant differences for the overall effects of retention forestry and logging activities (disturbed forest soil in the form of skid trails or strip roads). Concerning the sub-group analysis and multivariate meta-regression, there were no significant effects for stand type (broadleaf, coniferous, or mixed) or climatic conditions (mean annual temperature and annual precipitation). The percentage of biomass removal showed a significant positive correlation with the effect size, thus indicating that higher biomass removal in the framework of retention forestry leads to an increase in litter decomposition rate. Also, the mesh size of the litterbag showed a positive correlation with the effect size, suggesting that there is a probability of a factor of stronger disturbance to large-body decomposer organisms such as microarthropods.
Summary
Litter decomposition rate is related, among other factors, to soil microclimatic conditions and soil biota. Therefore, this process can be strongly influenced by active forest management, meant as a silvicultural treatment carried out by applying a given harvesting system. In the context of retention forestry, increased light availability seems to be the driving force in shaping increased litter decomposition rates with increasing biomass removal. On the other hand, when a clear-cut is applied, the stronger modification to the edaphic community leads to decreased litter decomposition rates. It is worth noting that the modification may also be related to soil manipulation to favour the establishment of artificial regeneration. The findings, however, showed very large variability, thus suggesting the need for further research on such a complex topic. Multidisciplinary studies that analyse the microclimate and the edaphic biological communities along with the litter decomposition are particularly recommended.
Similar content being viewed by others
Introduction
Wood has been a crucial renewable resource for mankind for millennia [1, 2], and it also plays a key role in the framework of climate change mitigation, as it is a long-term carbon sink [3,4,5,6]. Forests provide not only wood as a renewable raw material but also present several other fundamental ecosystem services, for instance, hydrological protection [7,8,9] and biodiversity conservation [10, 11]. Sustainable forest management is required to ensure the supply of wood while maintaining other ecosystem services [12,13,14]. Indeed, the active forest management needed to retrieve wood-based products from forest stands implies the implementation of a given silvicultural treatment through various forest operation options [15•, 16].
Sustainable forest management (SFM) involves carrying out forest operations in a proper manner [17•], and for that reason it is also necessary to apply the criteria of sustainability to forest logging. This depends on putting into practice logging activities that are cost-effective, but at the same time ensure safe working conditions for the operators and do not trigger excessive disturbances to the forest ecosystem [17•, 18]. One of the major environmental aspects to be taken into consideration when dealing with the sustainability of forest operations is the disturbance that can occur to forest soil [19, 20•], as well as to the soil biodiversity, which is important for many ecosystem functions of forests [21]. For example, the reduction of diversity of soil biota is linked to the reduction of decomposition rates, which is crucial for soil formation and fertility [22].
The alteration of soil physicochemical parameters is a consequence of the traffic on forest soil of the machinery applied for forest operations, and it is indeed a practically unavoidable disturbance related to the implementation of logging activities [23, 24]. Machinery-induced soil compaction has been extensively investigated and reported in the scientific literature, highlighting its negative implications on hydrological conditions, i.e. increased erosion and runoff [25,26,27], as well as biological conditions, i.e. decreased biodiversity in soil edaphic fauna [28] and harsher growing conditions for natural regeneration [18] of the forest ecosystem. On the other hand, the possible disturbances related to forest operations and the related silvicultural treatments on the biological processes that occur in the forest soil, e.g. litter decomposition, have yet to be fully understood [29••].
Litter decomposition is a fundamental driver of carbon and nutrient cycling in forest ecosystems, and links aboveground and belowground biogeochemical processes [30, 31••]. Litter decomposition is a complex process, in which climatic and edaphic factors jointly shape the rate of litter decay [32]. The alterations of a forest stand after a silvicultural intervention and the related forest operations can also alter litter decomposition rates [33]. For instance, increased light availability and consequential changes in understorey microclimate—soil temperature and soil moisture regimes as a consequence of the alteration of the canopy cover—can foster shifts in the detritus turnover, as well as in nutrient mineralisation rates [34]. Considering the importance of the process of litter decomposition for forest ecosystems, the influence of active forest management on this process has been studied extensively during both the twentieth and twenty-first centuries. However, contrasting results have been obtained with a given silvicultural treatment that can foster either an increase or decrease in the decomposition rate [34].
Considering this lack of a clear trend, a literature review is needed on this topic to further progress the state of the art and to highlight future research directions. With the intent to properly summarise the current knowledge and develop a quantitative synthesis of the effects of active forest management on litter decomposition rate, we performed a global meta-analysis focused on the studies that have investigated the effects of forest operations and silvicultural treatments on litter decomposition rates in forest soils.
Meta-analysis is a statistical technique usually applied for literature reviews, which is used to summarise the results of several studies on the same topic [35,36,37,38]. The major advantage of applying meta-analysis consists of the fact that the results obtained from a group of studies, carried out with a similar experimental design, are statistically analysed to provide generalised answers that may not have been evident when studies were analysed singularly [39, 40].
We aimed to assess and quantify the effects of forest operations and silvicultural treatments on litter decomposition rate, and to identify and understand the environmental and silvicultural variables that could affect the magnitude of the alterations. These are called moderators in the framework of meta-analysis.
Materials and Methods
Literature Search, Inclusion Criteria, and Database Building
We conducted a systematic search of the literature within the databases Google Scholar, Scopus, and Web of Science using as keywords: litter decomposition rate, litter decay, clear-cut, clear-cutting, shelterwood system, selective logging, retention forestry, thinning, coppice, coppicing, harvesting system, harvesting method, skid trail, strip road, forest operations, and linking these keywords with the Boolean operators AND or OR (Fig. 1). No restriction was applied to the database, which means that we collected information from studies concerning all forest types and located in all zones of the globe.
To retrieve further literature sources, we used the snowball system, which involved scanning the reference list of recent papers (published in 2021 and 2022) to identify additional references. However, no additional literature references were identified using this method. The literature search identified 158 potentially suitable papers. We removed duplicates and then we eliminated studies for which the abstract and title did not indicate suitable content. Finally, we checked the 82 remaining studies using the following inclusion criteria: (i) the paper must be in the English language; (ii) the paper must be focused on evaluating the effects of silvicultural treatment and/or forest operations on litter decomposition rate; (iii) the decomposition experiment must be undertaken on forest soil (excluding, for instance, decomposition experiments in streams); (iv) the experimental design of the paper must provide a control treatment, this treatment can be represented by an unharvested stand when investigating the effects of silvicultural treatments and by the soil not affected by machinery passage when investigating the direct effects of forest operations. Through applying these inclusion criteria we filtered the papers to 44 [29••, 32,33,34, 41,42,43,44,45,46,47,48,49,50,51, 52••, 53,54,55,56,57,58,59,60,61,62,63,64,65,66,67,68,69,70,71,72,73,74,75,76, 77••, 78, 79••] papers that generated 237 paired comparisons. These studies were included in the database for the meta-analysis (Fig. 1; Table S1).
As a measure of the litter decomposition rate we choose the k constant (year−1). The k constant is the fundamental parameter in the single negative exponential decay model defined by Olson [80], which assumes that litter decomposition follows single-exponential dynamics (Eq. 1):
where Xt is the amount of litter at time t, X0 is the initial amount, and k is the decomposition rate constant. Litterbag studies are generally set up installing a certain number of litterbags in the forest floor, with the well-known mass of plant material installed inside (\({X}_{0})\). After variable collection times (ranging from a few weeks to several years) it is possible to retrieve the values of \({X}_{t}\), and by fitting Eq. (1) to the data, it is possible to estimate the value of k constant.
Throughout the review of the literature we recorded values of k from the studies in a database. When values of k were not reported numerically in the main text or tables but only in a graphical form, we used the WebplotDigitizer software to retrieve the numerical information. When a study did not report the k constant directly, but the percentage of mass lost or remaining in the litterbag, we calculated the value of the k constant using Eq. (1). For 21 of 237 paired comparisons in the database we did not obtain data about the dispersion measure of the k constant. Therefore, we investigated the relationship between the standard deviation and the average values presented in our database, and developed a predictive model for the missing standard deviation values. Specifically, we assessed missing standard deviation data using power models (Eq. 2):
where SD is the standard deviation, M is the mean, and a and b are model parameters. We fit models using the stats::nls() function available in R software. Models RMSE ranged from 0.002 to 0.033 (Table S2; Fig. S1 in the Appendix).
At this stage, the overall database was subdivided into three sub-databases: one for the effects of retention forestry (32 studies and 137 paired comparisons), one for the effects of clear-cutting (19 studies and 88 paired comparisons), and one for the effects of logging activities (3 studies and 12 paired comparisons). We grouped all cases of those silvicultural treatments which do not remove all the biomass in the stand as retention forestry [81]. For the calculation of the effect size, we considered the k constant in the harvested stand as the experimental treatment and the values in a nearby unharvested stand as the control. The same approach was applied to the sub-database dealing with the studies which investigated the effects of clear-cutting on litter decomposition rate. For the effects of logging activities, we considered k reported for soil affected by the passage of machinery applied in forest operations (i.e. skid trails) as experimental treatment values, and k reported for the soil not affected by machinery passage in the harvested stand as control values.
Meta-analysis Implementation
At first, we conducted a multivariate meta-analysis for each of the sub-databases (retention forestry, clear-cut, and logging operations). Then we developed multivariate mixed-effects meta-analytic linear models to investigate the effects of different categorial and continuous moderators on the magnitude of alteration of litter decomposition rate (Table 1).
To estimate the effect size we used Hedges’ \(g\), which represents an unbiased standardised mean difference [82], which following Borenstein et al. [83] (Eq. 3) is determined from
where \(\overline{{X }_{t}}\) and \(\overline{{X }_{c}}\) represent, respectively, the average values of the k constant in the experimental treatment and in the control, \({\sigma }_{\mathrm{pooled}}\) is the estimate of the pooled standard deviation [84], and \(J\) represents a correction factor for a small sample size [85]. The positive or negative sign of Hedges’ g value indicates, respectively, an increase or a decrease in litter decomposition rate as a consequence of the experimental treatment. Values of Hedges’ g of 0.5, 1.0, and 2.0 represent a “small”, “medium”, and “large” effect, respectively, while values higher than 10.0 represent a “very large” effect [86].
One of the most important requirements of meta-analysis is the independence of effect sizes [87]. In our database the major part of the studies were done with more than one comparison between experimental treatment and control. For instance, a study could investigate the effects of various thinning intensities by comparing the litter decomposition rates with a single control area. The effect sizes extracted from the same studies cannot be considered independent [87], due to spatial and temporal autocorrelation, as well as dependence within author groups, connected with methodological approaches. For that reason more complex meta-analytic models are needed to account for data nesting within the database [88], and therefore we applied multivariate mixed-effects meta-analytical linear models implemented in the metafor::rma.mv() function [89]. In these models we used the study as a random effect. Firstly, we checked differences in effect sizes among clear-cuts, retention forestry, and soil affected by logging using the model with one moderator. Then, for clear-cuts and retention forestry separately, we assessed the effects of all moderators (Table 1). We checked the fitness of models using Akaike’s information criterion (AIC), selecting models with the lowest AIC. We visualised the results of meta-analyses using orchard plots for categorical moderators and bubble plots for continuous moderators, implemented in the orchard package [90]. All statistical analyses were performed using the software R version 4.2.2. [91].
Results
Spatial Distribution of the Studies
Studies dealing with retention forestry are widespread around the world and they cover practically all the biomes, although a higher presence of such studies is evident in the Northern Hemisphere with particular attention given to the topic in countries such as China (Fig. 2). The distribution of studies dealing with clear-cutting is even more concentrated in the Northern Hemisphere with the topic investigated mostly in North America. Concerning the studies dealing with the effects of logging operations, only three papers are present in our database for this meta-analysis, one located in Canada, one in Turkey, and one in Southeast Asia. The spatial distribution shows there was low representation of tropical zones in Africa and a low representation within Asia and South America. The boreal forests of the Southern Hemisphere were represented by only two studies.
Differences Among Clear-cutting, Retention Forestry, and Forest Soil Affected by Logging Activities
The average standardised mean difference was − 0.575 ± 0.262, with an interquartile range of − 1.430 to 0.743. Meta-analysis revealed that the overall effect size was 0.310 (95%CI: − 0.953, 0.333), with study-related random effect SD = 2.134 and heterogeneity measure Q (df = 236) of 18,355.02 and AIC of 6607. Subgroup analysis (AIC = 6497.3) revealed a low but statistically significant effect size for clear-cut (g = − 0.925 ± 0.463) and non-significant effect sizes for retention forestry (g = 0.038 ± 0.370) and soil affected by logging (g = − 0.246 ± 0.229). The heterogeneity measure for residuals QE (df = 234) was of 17,953.9 (Fig. 3, Table S2).
Orchard plot of standardised mean differences (Hedges’ g) of studies assessing forest operations and effects of silvicultural treatments on decomposition rate (bubbles), with effect sizes (black dots) and 95% confidence intervals (lines) estimated using multivariate meta-analysis (Table S2). k denotes the number of effect sizes per estimate with the number of related studies in brackets
Drivers of Clear-cut Effects
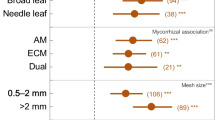
A multivariate meta-analysis of clear-cut effect sizes revealed that all hypothesised factors were included in the final model (AIC = 3077.8, AIC0 = 3934.1). In the final model the study-related random effect SD was 2.716 and the heterogeneity measure QE was 6439.8 (Table S3). Analysis revealed statistically and ecologically insignificant effects of stand type while statistically significant but biologically irrelevant effects of the substrate (Fig. 4). Effect sizes for mean annual temperature were non-significant, both statistically and biologically. Effect sizes for evaluation time were statistically insignificant, but the slope of partial regression revealed lower effect sizes for short evaluation times and higher effect sizes for longer periods. Similarly, we found a weak (statistically insignificant) positive effect of mean annual precipitation. For mesh size there was a statistically significant decreasing trend of effect sizes with increasing mesh size; however, it was driven by two outlying observations.
Effects of moderators driving effect size of clear-cutting on decomposition rate. Orchard plot (A) of standardised mean differences (Hedges’ g) shows differences between stand types and substrata in effect sizes (bubbles), with effect sizes (black dots) and 95% confidence intervals (lines) estimated using multivariate meta-analysis (Table S3). Bubble plots (B) show the predicted response of effect size to continuous moderators, with 95% confidence intervals
Drivers of Retention Forestry Effects
The multivariate meta-analysis of retention forestry effect sizes revealed that all hypothesised factors were included in the final model (AIC = 1448.0, AIC0 = 1638.9). In the final model study-related random effect SD = 3.459 and heterogeneity measure QE (df = 92) = 6534.6 (Table S4). Analysis revealed statistically and ecologically significant effects of stand type and statistically significant but ecologically insignificant effects of the substrate (Fig. 5). The predicted effect size was positive for mixed stands, negative for broadleaved, and close to 0 for coniferous stands. This was not a significant effect of mean annual temperature while the predicted effect size decreased with increasing mean annual precipitation. For evaluation time effect size was statistically and ecologically insignificant. For mesh size there was a statistically significant relationship with increasing effect sizes with increasing mesh size, however, with a low effect size. The proportion of stand removal statistically significantly affected the effect sizes. However, there was only a slight increase in effect sizes with an increased proportion of removal, and numerous cases revealed opposite trends, especially those with higher proportions of stand removal.
Effects of moderator driving effect sizes of retention forestry on decomposition rate. Orchard plot (A) of standardised mean differences (Hedges’ g) shows differences between stand types and substrata in effect sizes (bubbles), with effect sizes (black dots) and 95% confidence intervals (lines) estimated using multivariate meta-analysis (Table S4). Bubble plots (B) show the predicted response of effect size to continuous moderators, with 95% confidence intervals
Effects of Forest Operations (Decomposition in Forest Soil Affected by Logging Activities)
We found only three studies assessing the effects of soil disturbance as a consequence of logging operations on decomposition rates. Among these, we identified 12 observations that allowed for the calculation of overall effect size; however, due to geographical and ecological dispersion they did not allow us to identify drivers through a separate analysis. As previously reported, the overall average effect size indicates a decrease in litter decomposition rate in the soil affected by machinery passage. However, the difference in comparison to the soil not directly affected by logging activities was not significant. This was probably due to the presence of an outlier that reported an effect size that showed a statistically significant increase.
Discussion
Effects of Silvicultural Treatments on Litter Decomposition Rate
Notwithstanding the substantial variability observed in the various statistical analyses, the meta-analytic approach indicated trends and allowed the identification of needs for future investigation.
Firstly, our results highlighted the limited influence of environmental moderators, such as temperature and rainfall, on the effect size variation. Climatic conditions, in particular, temperature and precipitation, are known to be among the main drivers of the litter decomposition rate [92,93,94]. However, our results showed that climatic factors did not have a marked influence when investigating the effects of silvicultural treatments. In a few words, decomposition rates in forest ecosystems characterised by high temperature and high rainfall are obviously faster than under other conditions with lower temperature and lower rainfall, but the same silvicultural treatment applied in the two conditions leads to a similar magnitude of change in comparison to the litter decomposition rate in the unharvested control stand [32].
The stand type and the type of substrate used for the decomposition experiment did not show a clear influence on the effect size. It is clear that various litter types and stand types experience different litter decomposition rates [95,96,97], but on the other hand, the obtained results suggest that a given silvicultural treatment applied to a different stand influences the decomposition process with the same magnitude, leading to a similar percentage increase or decrease in the k constant.
The main finding of the present meta-analysis is the influence of the percentage of biomass removal. Surprisingly, the obtained results showed opposite trends when comparing retention forestry and clear-cutting. In particular, remaining in the framework of retention forestry, higher biomass removal revealed a slightly but significantly increased effect size with a positive slope of the regression equation, i.e. the higher the biomass removal the higher the increase in the litter decomposition rate (Fig. 5B). On the other hand, as shown in Fig. 3, clear-cut, which is actually 100% of biomass removal, leads to a statistically significant decrease in the litter decomposition rate in comparison to the unharvested control area, meaning that by removing all the biomass in the stand the decomposition process is slowed down significantly. It was expected that with increasing light availability on the forest floor, and subsequent increase in soil temperature, the litter decomposition rate accelerates [64, 98]. However, this was confirmed only within the framework of retention forestry. From the results it could be speculated that increasing biomass removal not only triggers higher decomposition rates due to increased light availability and increased soil temperature but also that, when applying clear-cutting, the modifications to the forest ecosystem go beyond the simple changes in light availability and soil temperature, thus modifying the soil processes to a greater extent and leading to a reduction of litter decomposition rate [41]. Recent studies have emphasised how retention forestry conserves higher biodiversity than clear-cutting, not only regarding plants [99], but mostly concerning ectomycorrhizal fungi [100] and soil macrofauna communities [101, 102], which are among the main drivers of litter decomposition.
The particularly strong disturbance to the overall soil environment related to clear-cutting can therefore lead to a change in the species community composition, thus slowing down the decomposition process [79••]. Furthermore, there are also some cases in which clear-cutting is associated with soil preparation for subsequent artificial regeneration [42], thus further increasing the disturbance to the forest soil, which can potentially lead to even stronger effects on biological processes such as litter decomposition. In our database there were not enough data to add the soil preparation as a moderator in the sub-group meta-analysis dealing with clear-cutting. However, it is possible to associate high effect size with clear-cut interventions followed by site preparation [42, 79••], as a consequence of organic matter removal which negatively affects microbial communities [42, 103]. Our results suggest that when the intervention is carried out in the framework of retention forestry, the main effects of the silvicultural treatment on the litter decomposition rate are associated with a change in light availability at the forest floor level; the bigger the canopy opening, the larger the amount of light reaching the soil and the faster the decomposition process. On the other hand, when the intervention is a clear-cut, light availability is no longer the main driver of the alteration to the decomposition process, but probably the stronger disturbance to the soil edaphic communities associated with clear-cutting, mostly when followed by site preparation activities, leads to a decrease in the litter decomposition rate.
Finally, it is also possible to comment on the effect of the mesh size on the magnitude of change related to silvicultural treatments. Although a statistically significant influence of mesh size was shown for clear-cutting, this result is clearly influenced by two outliers (Fig. 4B). On the other hand, the findings highlighted a positive correlation between the dimension of the mesh and the effect size concerning retention forestry. This means that the effect of retention forestry on litter decomposition rate is stronger when the litter-bag applied for the experiment has a large mesh. It would be interesting to clarify if this effect is somehow related to a stronger effect of canopy alteration on large-size soil fauna [104, 105] or to the fact that large mesh can favour the action of abrasive forces (for example, wind, precipitation, water flow), thus triggering material loss from the litterbag [106].
Effects of Forest Operations on Litter Decomposition Rate
The limited number of studies did not allow for the acquisition of enough information about the effects of forest operations and related soil disturbance on the litter decomposition rate. This was rather surprising, considering the intensive research effort to investigate soil disturbances in the field of applied forestry research [107,108,109]. Apart from the direct effects of machinery traffic on soil compaction [27], the implications of forest operations have been investigated from different points of view, for instance, the effects on erosion and runoff [7] or on the morphological features of natural regeneration [110]. Several studies also highlighted the strong effects of soil compaction on the soil microarthropod community [111,112,113]. The effects of machinery traffic and subsequent soil compaction on litter decomposition rate have been much less investigated and this is a fundamental aspect of forest operations requiring a deeper recognition.
It is well known that litter decomposition is a complex process that is driven by three main factors: environmental conditions, chemical composition of the litter, and soil biota [114]. Soil disturbances by logging activities can alter at least two of these factors: the environmental conditions and the soil organisms [51]. On the one hand, compacted soil can lead to increased temperature and thus higher decomposition rates [51]. On the other hand, the disturbances related to organic matter removal and alteration of soil edaphic communities can lead to decreased decomposition rates [52••]. Taking the above into account, the influence of logging activities on the litter decomposition rate is properly driven by the balance between these two aspects.
It is possible to hypothesise that the number of machine passes could be a strong influential factor considering its direct effects on soil compaction and organic matter removal, which could alter the microclimatic conditions and the microbial activity on the skid trails or strip roads [115, 116]. Enez et al. (2015) also mentioned the importance of the magnitude of soil disturbance in slowing down litter decomposition rates, with a strong decrease observed in scalped soil after bunching-extraction operation by forestry-fitted farm tractor [52••]. On the other hand, other studies reported that compacted soil after the passage of crawler tractors or excavators did not reveal strong changes in the litter decomposition rate when compared to the undisturbed soil [51, 60]. However, it is evident that the topic of alteration of litter decomposition rate by soil disturbance as a consequence of ground-based forest operations should be further investigated in future studies. Indeed, given that machinery traffic on the forest soil is the major driver of disturbance to the soil edaphic communities [16], and that such disturbance can usually affect up to 30–35% of the overall area of the cutting block [113, 117], it is fundamental to understand at a deeper level the influence of logging activities on the litter decomposition rate. Such a large proportion of the affected area cannot be neglected if we want to understand the complex process of litter decomposition and how it is related to active forest management.
Research Gaps and Directions for Future Studies
We need to gain a better understanding of the relationship between the driving factors for the decomposition rate and how they interact with each other, in order to draw solid ecosystem-specific conclusions for the future practices of sustainable forest management under climate change. This is particularly true for soil disturbances after forest operations. Due to the low number of studies, it was not possible to define a clear trend for the influence related to the disturbance to forest soil as a consequence of ground-based forest operations. It is therefore strongly recommended to implement further research on this topic, by investigating how the establishment of temporary skid trails or strip road patterns can affect the decomposition rate. Furthermore, such studies should go deeper into the frameworks of sustainable forest operations and reduced-impact logging, investigating and comparing the effects on the litter decomposition rate of logging activities performed by different machines, different levels of mechanisation, different approaches to timber extraction (for instance, skidding vs forwarding vs cable yarding), as well as the effectiveness of mitigation strategies and best management practices. These important topics have been practically neglected by researchers, even though knowledge of these topics is fundamental to shape sustainable management of forest ecosystems. Moreover, more studies are required from underrepresented regions and biomes to account for a wider spectrum of case-specific outcomes.
The major finding of this meta-analysis is the great variability in the observed effect sizes. The influence of silvicultural activities on litter decomposition rate deserves further attention. The intrinsic complexity of the process of litter decomposition suggests that a multidisciplinary approach to the topic is needed, by carrying out experiments that evaluate not only the decomposition process but also to monitor in parallel the microclimate and the various biological communities in the soil. Only in this way, recommendations for forest management can be formulated.
We argue that future studies covering the interactions of soil organisms, the diversity of microbial communities within forest ecosystems, and their management across a range of local to global scales are needed to further develop sustainable forest operation measures and recommendations for the protection of soil biodiversity [118]. Finally, we stress the importance of investigating forest ecosystem-specific responses of litter decomposition not only under changing climatic conditions but also for changing forest management trends [119, 120]. Specifically, future forest operations and silvicultural treatments for the adaptation to climate change should consider the effects of active forest management and logging activities on the functional diversity in forest soils.
Conclusion
In this review we undertook the first global meta-analysis on the role of silvicultural treatments and forest operations on litter decomposition rates. Our findings demonstrated that the influence of active forest management on the litter decomposition rate is the result of the interaction between the alteration of light and soil temperature conditions, related to canopy opening, and the disturbance which occurs to soil edaphic organisms. The relative equilibrium between these two driving effects shapes the direction (increased or decreased decomposition rate) of the change in the decomposition process triggered by active forest management, as well as its magnitude. For retention forestry the alteration to light conditions and related soil temperature seems to be predominant, leading to a positive correlation between the percentage of biomass removal and litter decomposition rate. In the case of clear-cutting, the litter decomposition rate tends to decrease, probably as a consequence of a stronger alteration to soil organism communities, and also related to further soil manipulation to facilitate artificial regeneration of the stand. In conclusion, we know remarkably little about the interactions of forest management activities, the soil biodiversity, and the changing climatic conditions. We strongly suggest studying the response of the litter decomposition rate to specific forest operations, approaches, and applications, while taking into account the possible effects of climate change. Within the context of local active forest management adaptation measures to combat the challenges of climate change, the effect on litter decomposition should be investigated in future research, as well as the development of best management practices to protect soil biodiversity following the paradigm of sustainable forest operations.
Data Availability
Data are available from the corresponding author upon reasonable request.
References
Papers of particular interest, published recently, have been highlighted as: • Of importance •• Of major importance
Lennox S, Backwell L, D’Errico F, Wadley L. A vegetation record based on charcoal analysis from Border Cave, KwaZulu-Natal, South Africa, ∼227 000 to ∼44 000 years ago. Quat Sci Rev. 2022;293:107676. https://doi.org/10.1016/j.quascirev.2022.107676.
Bahtiar ET, Denih A, Priadi T, Putra GR, Koswara A, Nugroho N, Hermawan D. Comparing the building code sawn lumber’s wet service factors (cm) with four commercial wood species laboratory tests. Forests. 2022;13:2094. https://doi.org/10.3390/f13122094.
Evans PD, Matsunaga H, Preston AF, Kewish CM. Wood protection for carbon sequestration — a review of existing approaches and future directions. Curr For Reports. 2022;8:181–98. https://doi.org/10.1007/s40725-022-00166-x.
Jagodziński AM, Dyderski MK, Horodecki P. Differences in biomass production and carbon sequestration between highland and lowland stands of Picea abies (L.) H. Karst. and Fagus sylvatica L. For Ecol Manag. 2020;474:118329. https://doi.org/10.1016/j.foreco.2020.118329.
Forrester DI, Tachauer IHH, Annighoefer P, et al. Generalized biomass and leaf area allometric equations for European tree species incorporating stand structure, tree age and climate. For Ecol Manag. 2017;396:160–75. https://doi.org/10.1016/j.foreco.2017.04.011.
Ochoa-Hueso R, Delgado-Baquerizo M, An King PT, Benham M, Arca V, Power SA. Ecosystem type and resource quality are more important than global change drivers in regulating early stages of litter decomposition. Soil Biol Biochem. 2019;129:144–52. https://doi.org/10.1016/j.soilbio.2018.11.009.
Picchio R, Jourgholami M, Zenner EK. Effects of forest harvesting on water and sediment yields: a review toward better mitigation and rehabilitation strategies. Curr For Reports. 2021;7:214–9. https://doi.org/10.1007/s40725-021-00146-7.
Sjöman H, Levinsson A, Emilsson T, Ibrahimova A, Alizade V, Douglas P, Wiström B. Evaluation of Alnus subcordata for urban environments through assessment of drought and flooding tolerance. Dendrobiology. 2021;85:39–50. https://doi.org/10.12657/denbio.085.005.
Manić M, Đorđević M, Đokić M, Dragović R, Kićović D, Đorđević D, Jović M, Smičiklas I, Dragović S. Remote sensing and nuclear techniques for soil erosion research in forest areas: case study of the Crveni Potok Catchment. Front Environ Sci. 2022;10:679. https://doi.org/10.3389/fenvs.2022.897248.
Courbaud B, Larrieu L, Kozak D, et al. Factors influencing the rate of formation of tree-related microhabitats and implications for biodiversity conservation and forest management. J Appl Ecol. 2022;59:492–503. https://doi.org/10.1111/1365-2664.14068.
Murdjoko A, Aristone Djitmau D, Ungirwalu A, et al. Pattern of tree diversity in lowland tropical forest in Nikiwar, West Papua, Indonesia. Dendrobiology. 2021;85:78–91. https://doi.org/10.12657/denbio.085.008.
Zhang Q, Zhang Q, Zhai Y, et al. Drivers of aboveground biomass shift with forest stratum in temperate forest of North China. Sci Total Environ. 2023;860:160548. https://doi.org/10.1016/j.scitotenv.2022.160548.
Robinson R, McCaw L, Wills A. Biodiversity monitoring informs forest management in south-west Western Australia: ten-year findings of Forestcheck. For Ecol Manag. 2023;529:120659. https://doi.org/10.1016/j.foreco.2022.120659.
Vančura K, Šimková M, Vacek Z, et al. Effects of environmental factors and management on dynamics of mixed calcareous forests under climate change in Central European lowlands. Dendrobiology. 2022;87:79–100. https://doi.org/10.12657/denbio.087.006.
• Latterini F, Venanzi R, Picchio R, Jagodziński AM. Short-term physicochemical and biological impacts on soil after forest logging in Mediterranean broadleaf forests: 15 years of field studies summarized by a data synthesis under the meta-analytic framework. Forestry. 2023. https://doi.org/10.1093/forestry/cpac060. Recent paper applying a meta-analytic approach to investigate the effects of forest operations on several features of the soil.
Venanzi R, Picchio R, Grigolato S, Latterini F. Soil and forest regeneration after different extraction methods in coppice forests. For Ecol Manag. 2019;454:117666. https://doi.org/10.1016/j.foreco.2019.117666.
• Marchi E, Chung W, Visser R, Abbas D, Nordfjell T, Mederski PS, McEwan A, Brink M, Laschi A. Sustainable forest operations (SFO): a new paradigm in a changing world and climate. Sci Total Environ. 2018;634:1385–97. https://doi.org/10.1016/j.scitotenv.2018.04.084. Review paper defining the fundamental paradigm of Sustainable Forest Operations.
Latterini F, Mederski PS, Jaeger D, Venanzi R, Tavankar F, Picchio R. The influence of various silvicultural treatments and forest operations on tree species biodiversity. Curr For Reports. 2023;9:59–71. https://doi.org/10.1007/s40725-023-00179-0.
Hoffmann S, Schönauer M, Heppelmann J, et al. Trafficability prediction using depth-to-water maps: the status of application in Northern and Central European forestry. Curr For Reports. 2022;8:55–71. https://doi.org/10.1007/s40725-021-00153-8.
• Labelle ER, Hansson L, Högbom L, Jourgholami M, Laschi A. Strategies to mitigate the effects of soil physical disturbances caused by forest machinery: a comprehensive review. Curr For Reports. 2022;8:20–37. https://doi.org/10.1007/s40725-021-00155-6. Recent review of the application of best management practices to decrease the negative impacts of forest operations on the soil.
Bardgett RD, van der Putten WH. Belowground biodiversity and ecosystem functioning. Nature. 2014;515:505–11. https://doi.org/10.1038/nature13855.
Nielsen UN, Ayres E, Wall DH, Bardgett RD. Soil biodiversity and carbon cycling: a review and synthesis of studies examining diversity-function relationships. Eur J Soil Sci. 2011;62:105–16. https://doi.org/10.1111/j.1365-2389.2010.01314.x.
Picchio R, Mederski PS, Tavankar F. How and how much, do harvesting activities affect forest soil, regeneration and stands? Curr For Reports. 2020;6:115–28. https://doi.org/10.1007/s40725-020-00113-8.
Schweier J, Magagnotti N, Labelle ER, Athanassiadis D. Sustainability impact assessment of forest operations: a review. Curr For Reports. 2019;5:101–13. https://doi.org/10.1007/s40725-019-00091-6.
Karami S, Jourgholami M, Attarod P, Venanzi R, Latterini F, Stefanoni W, Picchio R. The medium-term effects of forest operations on a mixed broadleaf forest: changes in soil properties and loss of nutrients. L Degrad Dev. 2023. https://doi.org/10.1002/ldr.4660.
Jourgholami M, Labelle ER. Effects of plot length and soil texture on runoff and sediment yield occurring on machine-trafficked soils in a mixed deciduous forest. Ann For Sci. 2020;77:1–11. https://doi.org/10.1007/s13595-020-00938-0.
Nazari M, Eteghadipour M, Zarebanadkouki M, Ghorbani M, Dippold MA, Bilyera N, Zamanian K. Impacts of logging-associated compaction on forest soils: a meta-analysis. Front For Glob Chang. 2021;4:780074. https://doi.org/10.3389/ffgc.2021.780074.
Latterini F, Venanzi R, Tocci D, Picchio R. Depth-to-water maps to identify soil areas that are potentially sensitive to logging disturbance : initial evaluations in the Mediterranean forest context. Land. 2022;11:709. https://doi.org/10.3390/land11050709.
•• Pang Y, Tian J, Lv X, Wang R, Wang D, Zhang F. Contrasting dynamics and factor controls in leaf compared with different-diameter fine root litter decomposition in secondary forests in the Qinling Mountains after 5 years of whole-tree harvesting. Sci Total Environ. 2022;838:156194. https://doi.org/10.1016/j.scitotenv.2022.156194. Paper investigating the effects of thinning by whole-tree harvesting of the decomposition of different substrates. Authors did not reveal a significant effect of such silvicultural treatment on the decomposition rate.
Wang J, You Y, Tang Z, Liu S, Sun OJ. Variations in leaf litter decomposition across contrasting forest stands and controlling factors at local scale. J Plant Ecol. 2015;8:261–72. https://doi.org/10.1093/jpe/rtu019.
•• Prescott CE, Vesterdal L. Decomposition and transformations along the continuum from litter to soil organic matter in forest soils. For Ecol Manag. 2021;498:119522. https://doi.org/10.1016/j.foreco.2021.119522. Recent review summarising the most novel findings on the topic of litter decomposition in forest stands.
Bravo-Oviedo A, Ruiz-Peinado R, Onrubia R, del Río M. Thinning alters the early-decomposition rate and nutrient immobilization-release pattern of foliar litter in Mediterranean oak-pine mixed stands. For Ecol Manag. 2017;391:309–20. https://doi.org/10.1016/j.foreco.2017.02.032.
Kunhamu TK, Kumar BM, Viswanath S. Does thinning affect litterfall, litter decomposition, and associated nutrient release in Acacia mangium stands of Kerala in peninsular India? Can J For Res. 2009;39:792–801. https://doi.org/10.1139/X09-008.
Blanco JA, Imbert JB, Castillo FJ. Thinning affects Pinus sylvestris needle decomposition rates and chemistry differently depending on site conditions. Biogeochemistry. 2011;106:397–414. https://doi.org/10.1007/s10533-010-9518-2.
Gong C, Tan Q, Liu G, Xu M. Impacts of tree mixtures on understory plant diversity in China. For Ecol Manag. 2021;498:119545. https://doi.org/10.1016/j.foreco.2021.119545.
Gatica G, Elena Fernández M, Paula Juliarena M, Gyenge J. Does forest management affect the magnitude and direction of the afforestation effect on soil methane fluxes? A meta-analysis. For Ecol Manag. 2022;507:120009. https://doi.org/10.1016/j.foreco.2022.120009.
Basile M, Krištín A, Mikusiński G, Thorn S, Żmihorski M, Pasinelli G, Brockerhoff EG. Salvage logging strongly affects woodpecker abundance and reproduction: a meta-analysis. Curr For Reports. 2022;9:1–14. https://doi.org/10.1007/s40725-022-00175-w.
Louis LT, Kizha AR, Daigneault A, Han H-S, Weiskittel A. Factors affecting operational cost and productivity of ground-based timber harvesting machines: a meta-analysis. Curr For Reports. 2022;8:38–54. https://doi.org/10.1007/s40725-021-00156-5.
Hedges LV, Gurevitch J, Curtis PS. The meta-analysis of response ratios in experimental ecology. Ecology. 1999;80:1150–6. https://doi.org/10.1890/0012-9658.
Lajeunesse MJ. On the meta-analysis of response ratios for studies with correlated and multi-group designs. Ecology. 2011;92:2049–55. https://doi.org/10.1890/11-0423.1.
Zheng Y, Guan F, Fan S, Yan X, Huang L. Dynamics of leaf-litter biomass, nutrient resorption efficiency and decomposition in a Moso bamboo forest after strip clearcutting. Front Plant Sci. 2022;12:3297. https://doi.org/10.3389/fpls.2021.799424.
Duchesne LC, Wetzel S. Effect of clear-cutting, prescribed burning and scarification on litter decomposition in an Eastern Ontario jack pine (Pinus banksiana) ecosystem. Int J Wildl Fire. 2000;9:195–201. https://doi.org/10.1071/WF00016.
Bird GA, Chatarpaul L. Effect of forest harvest on decomposition and colonization of maple leaf litter by soil microarthropods. Can J Soil Sci. 1988;68:29–40. https://doi.org/10.4141/cjss88-003.
Prescott CE, Hope GD, Blevins LL. Effect of gap size on litter decomposition and soil nitrate concentrations in a high-elevation spruce-fir forest. Can J For Res. 2003;33:2210–20. https://doi.org/10.1139/x03-152.
Symonds J, Morris DM, Kwiaton MM. Effect of harvest intensity and soil moisture regime on the decomposition and release of nutrients from needle and twig litter in northwestern Ontario. Boreal Environ Res. 2013;18:401–13.
Jerabkova L, Prescott CE, Kishchuk BE. Effect of variable-retention harvesting on soil nitrogen availability in boreal mixedwood forests. Can J For Res. 2006;36:3029–38. https://doi.org/10.1139/x06-175.
Oro Castro N, Moretto A, Selzer LJ, Escobar J. Effects of alternative silvicultural systems on litter decomposition and nutrients dynamics in sub-Antarctic forests. Agrofor Syst. 2019;93:885–99. https://doi.org/10.1007/s10457-018-0183-0.
Prescott CE. Effects of clearcutting and alternative silvicultural systems on rates of decomposition and nitrogen mineralization in a coastal montane coniferous forest. For Ecol Manag. 1997;95:253–60. https://doi.org/10.1016/S0378-1127(97)00027-3.
Cortina J, Vallejo VR. Effects of clearfelling on forest floor accumulation and litter decomposition in a radiata pine plantation. For Ecol Manag. 1994;70:299–310. https://doi.org/10.1016/0378-1127(94)90095-7.
Mabuhay-Omar J, … SO-P, 2018 U (2018) Effects of forest management practices on microbial biomass, litter decomposition, microbial abundance, and the soil’s physical and chemical properties of replacement plantations after pine wilt disease. Palawan Sci 10:48–68
Kranabetter JM, Chapman BK. Effects of forest soil compaction and organic matter removal on leaf litter decomposition in central British Columbia. Can J Soil Sci. 1999;79:543–50. https://doi.org/10.4141/S98-081.
•• Enez K, Aricak B, Sariyildiz T. Effects of forest harvesting activities on litter decomposition rates of scots pine, trojan fir and sweet chestnut. Šumarski List. 2015;139:361–8. Paper investigating the effects of different magnitudes of soil disturbance related to logging activities on litter decomposition rates. Authors highlighted a correlation between the magnitude of soil disturbance and the alteration of litter decomposition rate.
Son Y, Lee WK, Lee SE, Ryu SR. Effects of thinning on soil nitrogen mineralization in a Japanese larch plantation. Commun Soil Sci Plant Anal. 1999;30:2539–50. https://doi.org/10.1080/00103629909370393.
Li X, Zhang D, Zhang Y, Zhang J, Song S, Zhou Y. Forest gap size can efficiently promote litter decomposition and nutrient release in south-western China. South For. 2019;81:185–94. https://doi.org/10.2989/20702620.2018.1542564.
Purahong W, Kapturska D, Pecyna MJ, Schulz E, Schloter M, Buscot F, Hofrichter M, Krug̈er D. Influence of different forest system management practices on leaf litter decomposition rates, nutrient dynamics and the activity of ligninolytic enzymes: A case study from Central European forests. PLoS One. 2014;9:e93700. https://doi.org/10.1371/journal.pone.0093700.
Kim C, Son Y, Lee WK, Jeong J, Noh NJ. Influences of forest tending works on carbon distribution and cycling in a Pinus densiflora S. et Z. stand in Korea. For Ecol Manag. 2009;257:1420–6. https://doi.org/10.1016/j.foreco.2008.12.015.
Chen X, Page-Dumroese D, Lv R, Wang W, Li G, Liu Y. Interaction of initial litter quality and thinning intensity on litter decomposition rate, nitrogen accumulation and release in a pine plantation. Silva Fenn. 2014;48:1211. https://doi.org/10.14214/sf.1211.
Zhang J, Zhang D, Jian Z, Zhou H, Zhao Y, Wei D. Litter decomposition and the degradation of recalcitrant components in Pinus massoniana plantations with various canopy densities. J For Res. 2019;30:1395–405. https://doi.org/10.1007/s11676-018-0715-5.
Gliksman D, Haenel S, Osem Y, Yakir D, Zangy E, Preisler Y, Grünzweig JM. Litter decomposition in Mediterranean pine forests is enhanced by reduced canopy cover. Plant Soil. 2018;422:317–29. https://doi.org/10.1007/s11104-017-3366-y.
Yoshida T, Hasegawa M, Ito MT, Kawaguchi T, Seino T, Chung AYC, Kitayama K. Litter decomposition on forest roads versus inside tropical rainforests in Sabah, Malaysia. J Trop For Sci. 2019;31:108–13. https://doi.org/10.26525/jtfs2019.31.1.108113.
Li R, Guan X, Han J, et al. Litter decomposition was retarded by understory removal but was unaffected by thinning in a Chinese fir [Cunninghamia lanceolata (Lamb.) Hook] plantation. Appl Soil Ecol. 2021;163:103968. https://doi.org/10.1016/j.apsoil.2021.103968.
Martínez-Falcón AP, Moreno CE, Pavón NP. Comunidades de fauna y descomposición de la hojarasca en un bosque de pino-encino con tala selectiva y un bosque sin manejo en México. Bosque. 2015;36:81–94. https://doi.org/10.4067/S0717-92002015000100009.
Moroni MT, Zhu X. Litter-fall and decomposition in harvested and un-harvested boreal forests. For Chron. 2012;88:613–21. https://doi.org/10.5558/tfc2012-114.
Lado-Monserrat L, Lidón A, Bautista I. Litterfall, litter decomposition and associated nutrient fluxes in Pinus halepensis: influence of tree removal intensity in a Mediterranean forest. Eur J For Res. 2016;135:203–14. https://doi.org/10.1007/s10342-015-0923-x.
Lin N, Bartsch N, Heinrichs S, Vor T. Long-term effects of canopy opening and liming on leaf litter production, and on leaf litter and fine-root decomposition in a European beech (Fagus sylvatica L.) forest. For Ecol Manag. 2015;338:183–90. https://doi.org/10.1016/j.foreco.2014.11.029.
Hagemann U, Moroni MT. Moss and lichen decomposition in old-growth and harvested high-boreal forests estimated using the litterbag and minicontainermethods. Soil Biol Biochem. 2015;87:10–24. https://doi.org/10.1016/j.soilbio.2015.04.002.
Will GM, Hodgkiss PD, Madgwick HAI. Nutrient losses from litterbags containing Pinus radiata litter: influences of thinning, clearfelling, and urea fertiliser. New Zeal J For Sci. 1983;13:291–304.
Heneghan L, Salmore A, Crossley DA. Recovery of decomposition and soil microarthropod communities in an Appalachian watershed two decades after a clearcut. For Ecol Manag. 2004;189:353–62. https://doi.org/10.1016/j.foreco.2003.09.002.
Hope GD, Prescott CE, Blevins LL. Responses of available soil nitrogen and litter decomposition to openings of different sizes in dry interior Douglas-fir forests in British Columbia. For Ecol Manag. 2003;186:33–46. https://doi.org/10.1016/S0378-1127(03)00241-X.
Matos PS, Barreto-Garcia PAB, Gama-Rodrigues EF, de Paula A, de Oliveira AM. Short-term effects of forest management on litter decomposition in Caatinga dry forest. Energy, Ecol Environ. 2022;7:130–41. https://doi.org/10.1007/s40974-021-00231-4.
González G, Lodge DJ, Richardson BA, Richardson MJ. A canopy trimming experiment in Puerto Rico: the response of litter decomposition and nutrient release to canopy opening and debris deposition in a subtropical wet forest. For Ecol Manag. 2014;332:32–46. https://doi.org/10.1016/j.foreco.2014.06.024.
Son Y, Jun YC, Lee YY, Kim RH, Yang SY. Soil carbon dioxide evolution, litter decomposition, and nitrogen availability four years after thinning in a Japanese larch plantation. Commun Soil Sci Plant Anal. 2004;35:1111–22. https://doi.org/10.1081/CSS-120030593.
Chen Y, Zhang Y, Cao J, Fu S, Hu S, Wu J, Zhao J, Liu Z. Stand age and species traits alter the effects of understory removal on litter decomposition and nutrient dynamics in subtropical Eucalyptus plantations. Glob Ecol Conserv. 2019;20:e00693. https://doi.org/10.1016/j.gecco.2019.e00693.
Bauhus J, Vor T, Bartsch N, Cowling A. The effects of gaps and liming on forest floor decomposition and soil C and N dynamics in a Fagus sylvatica forest. Can J For Res. 2004;34:509–18. https://doi.org/10.1139/x03-218.
Lee J, Morrison IK, Leblanc JD, Dumas MT, Cameron DA. Carbon sequestration in trees and regrowth vegetation as affected by clearcut and partial cut harvesting in a second-growth boreal mixedwood. For Ecol Manag. 2002;169:83–101. https://doi.org/10.1016/S0378-1127(02)00300-6.
Mahmoudi MR, Bachtobji-Bouachir B, Sebai H, Ben-Attia M, Ghanem-Boughanmi N. Change of the litter fall, decomposition, and nutrient release in cork oak forest after anthropogenic disturbances in North West of Tunisia. Environ Sci Pollut Res. 2021;28:38584–93. https://doi.org/10.1007/s11356-021-13294-x.
•• Strukelj M, Brais S, Mazerolle MJ, Paré D, Drapeau P. Decomposition patterns of foliar litter and deadwood in managed and unmanaged stands: a 13-year experiment in boreal mixedwoods. Ecosystems. 2018;21:68–84. https://doi.org/10.1007/s10021-017-0135-y. Long-term research on the effects of forest management on litter decomposition rate. Authors highlighted greater effects of clear-cut than of partial harvesting in the context of boreal forestry.
Fenton NJ, Bergeron Y, Paré D. Decomposition rates of bryophytes in managed boreal forests: influence of bryophyte species and forest harvesting. Plant Soil. 2010;336:499–508. https://doi.org/10.1007/s11104-010-0506-z.
•• Laigle I, Moretti M, Rousseau L, et al. Direct and indirect effects of forest anthropogenic disturbance on above and below ground communities and litter decomposition. Ecosystems. 2021;24:1716–37. https://doi.org/10.1007/s10021-021-00613-z. Study which demonstrated the relationship between changes in litter decomposition rate and disturbance to soil edaphic organisms after forest management activities.
Olson JS. Energy storage and the balance of producers and decomposers in ecological systems. Ecology. 1963;44:322–31. https://doi.org/10.2307/1932179.
Mason WL, Diaci J, Carvalho J, Valkonen S. Continuous cover forestry in Europe: usage and the knowledge gaps and challenges to wider adoption. Forestry. 2022;95:1–12. https://doi.org/10.1093/forestry/cpac008.
Hedges LV. Distribution theory for glass’s estimator of effect size and related estimators. J Educ Stat. 1981;6:107–28.
Borenstein M, Hedges LV, Higgins JPT, Rothstein HR. Introduction to meta-analysis. Wiley; 2021.
Rosnow RL, Rosenthal R. Computing contrasts, effect sizes, and counternulls on other people’s published data: general procedures for research consumers. Psychol Methods. 1996;1:331.
Hedges LV, Olkin I. Statistical methods for meta-analysis. Academic press; 2014.
Durlak JA. How to select, calculate, and interpret effect sizes. J Pediatr Psychol. 2009;34:917–28. https://doi.org/10.1093/jpepsy/jsp004.
Cheung MW-L. A guide to conducting a meta-analysis with non-independent effect sizes. Neuropsychol Rev. 2019;29:387–96. https://doi.org/10.1007/s11065-019-09415-6.
Cheung MW-L. Modeling dependent effect sizes with three-level meta-analyses: a structural equation modeling approach. Psychol Methods. 2014;19:211–29. https://doi.org/10.1037/a0032968.
Viechtbauer W (2010) Conducting meta-analyses in R with the metafor package. J Stat Softw. https://doi.org/10.18637/jss.v036.i03
Nakagawa S, Lagisz M, O’Dea RE, Rutkowska J, Yang Y, Noble DWA, Senior AM. The orchard plot: cultivating a forest plot for use in ecology, evolution, and beyond. Res Synth Methods. 2021;12:4–12. https://doi.org/10.1002/jrsm.1424.
R Core Team R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. Accessible at https://www.R-project.org/. Accessed 10 Jan 2023.
Desie E, Zuo J, Verheyen K, et al. Disentangling drivers of litter decomposition in a multi-continent network of tree diversity experiments. Sci Total Environ. 2023;857:159717. https://doi.org/10.1016/j.scitotenv.2022.159717.
Kamczyc J, Dyderski MK, Horodecki P, Jagodziński AM. Temperature and precipitation affect seasonal changes in mite communities (Acari: Mesostigmata) in decomposing litter of broadleaved and coniferous temperate tree species. Ann For Sci. 2022;79:12. https://doi.org/10.1186/s13595-022-01129-9.
García-Palacios P, Maestre FT, Kattge J, Wall DH. Climate and litter quality differently modulate the effects of soil fauna on litter decomposition across biomes. Ecol Lett. 2013;16:1045–53. https://doi.org/10.1111/ele.12137.
Urbanowski CK, Horodecki P, Kamczyc J, Skorupski M, Jagodziński AM. Predatory mite instars (Acari, Mesostigmata) and decomposing tree leaves in mixed and monoculture stands growing on a spoil heap and surrounding forests. Exp Appl Acarol. 2021;84:703–31. https://doi.org/10.1007/s10493-021-00646-y.
Horodecki P, Nowiński M, Jagodziński AM. Advantages of mixed tree stands in restoration of upper soil layers on postmining sites: a five-year leaf litter decomposition experiment. L Degrad Dev. 2019;30:3–13. https://doi.org/10.1002/ldr.3194.
Horodecki P, Jagodziński AM. Site type effect on litter decomposition rates: a three-year comparison of decomposition process between spoil heap and forest sites. Forests. 2019;10:353. https://doi.org/10.3390/f10040353.
Brown JH, Gillooly JF, Allen AP, Savage VM, West GB. Toward a metabolic theory of ecology. Ecology. 2004;85:1771–89. https://doi.org/10.1890/03-9000.
Fedrowitz K, Koricheva J, Baker SC, et al. Can retention forestry help conserve biodiversity? A meta-analysis. J Appl Ecol. 2014;51:1669–79. https://doi.org/10.1111/1365-2664.12289.
Rosenvald R, Lõhmus A. For what, when, and where is green-tree retention better than clear-cutting? A review of the biodiversity aspects. For Ecol Manag. 2008;255:1–15. https://doi.org/10.1016/j.foreco.2007.09.016.
Beese WJ, Deal J, Dunsworth BG, Mitchell SJ, Philpott TJ. Two decades of variable retention in British Columbia: a review of its implementation and effectiveness for biodiversity conservation. Ecol Process. 2019;8:33. https://doi.org/10.1186/s13717-019-0181-9.
Mathieu J, Rossi J-P, Mora P, Lavelle P, da Martins PFS, Rouland C, Grimaldi M. Recovery of soil macrofauna communities after forest clearance in Eastern Amazonia, Brazil. Conserv Biol. 2005;19:1598–605. https://doi.org/10.1111/j.1523-1739.2005.00200.x.
Li Q, Lee Allen H, Wollum AG. Microbial biomass and bacterial functional diversity in forest soils: effects of organic matter removal, compaction, and vegetation control. Soil Biol Biochem. 2004;36:571–9. https://doi.org/10.1016/j.soilbio.2003.12.001.
Bradford MA, Tordoff GM, Eggers T, Jones TH, Newington JE. Microbiota, fauna, and mesh size interactions in litter decomposition. Oikos. 2002;99:317–23. https://doi.org/10.1034/j.1600-0706.2002.990212.x.
Handa IT, Aerts R, Berendse F, et al. Consequences of biodiversity loss for litter decomposition across biomes. Nature. 2014;509:218–21. https://doi.org/10.1038/nature13247.
Lecerf A. Methods for estimating the effect of litterbag mesh size on decomposition. Ecol Modell. 2017;362:65–8. https://doi.org/10.1016/j.ecolmodel.2017.08.011.
Picchio R, Venanzi R, Tavankar F, Luchenti I, Iranparast Bodaghi A, Latterini F, Nikooy M, Di Marzio N, Naghdi R. Changes in soil parameters of forests after windstorms and timber extraction. Eur J For Res. 2019;138:875–88. https://doi.org/10.1007/s10342-019-01210-5.
Tavankar F, Picchio R, Nikooy M, Jourgholami M, Naghdi R, Latterini F, Venanzi R. Soil natural recovery process and Fagus orientalis Lipsky seedling growth after timber extraction by wheeled skidder. Land. 2021;10:113. https://doi.org/10.3390/land10020113.
Picchio R, Mercurio R, Venanzi R, Gratani L, Giallonardo T, Lo Monaco A, Frattaroli AR. Strip clear-cutting application and logging typologies for renaturalization of pine afforestation—a case study. Forests. 2018;9:366. https://doi.org/10.3390/f9060366.
Tavankar F, Nikooy M, Ezzati S, Jourgholami M, Latterini F, Venanzi R, Picchio R. Long-term assessment of soil physicochemical properties and seedlings establishment after skidding operations in mountainous mixed hardwoods. Eur J For Res. 2022;141:571–85. https://doi.org/10.1007/s10342-022-01461-9.
Venanzi R, Picchio R, Grigolato S, Spinelli R. Soil disturbance induced by silvicultural treatment in chestnut (Castanea sativa Mill.) coppice and post-disturbance recovery. Forests. 2020;11:1053. https://doi.org/10.3390/f11101053.
Venanzi R, Picchio R, Spinelli R, Grigolato S. Soil disturbance and recovery after coppicing a Mediterranean oak stand: the effects of silviculture and technology. Sustainability. 2020;12:4074. https://doi.org/10.3390/su12104074.
Venanzi R, Latterini F, Stefanoni W, Tocci D, Picchio R. Variations of soil physico-chemical and biological features after logging using two different ground-based extraction methods in a beech high forest—a case study. Land. 2022;11:388. https://doi.org/10.3390/land11030388.
Aerts R. Climate, leaf litter chemistry and leaf litter decomposition in terrestrial ecosystems: a triangular relationship. Oikos. 1997;79:439. https://doi.org/10.2307/3546886.
Barlow J, Gardner TA, Ferreira LV, Peres CA. Litter fall and decomposition in primary, secondary and plantation forests in the Brazilian Amazon. For Ecol Manag. 2007;247:91–7. https://doi.org/10.1016/j.foreco.2007.04.017.
Ilstedt U, Nordgren A, Malmer A. Soil chemical and microbial properties after disturbance by crawler tractors in a Malaysian forest plantation. For Ecol Manag. 2006;225:313–9. https://doi.org/10.1016/j.foreco.2006.01.008.
Olesinski J, Lavigne MB, Kershaw JA, Krasowski MJ. Fine-root dynamics change during stand development and in response to thinning in balsam fir (Abies balsamea L. Mill.) forests. For Ecol Manag. 2012;286:48–58. https://doi.org/10.1016/j.foreco.2012.08.047.
McGuire KL, Treseder KK. Microbial communities and their relevance for ecosystem models: decomposition as a case study. Soil Biol Biochem. 2010;42:529–35. https://doi.org/10.1016/j.soilbio.2009.11.016.
Andersson E, Keskitalo E, Lawrence A. Adaptation to climate change in forestry: a perspective on forest ownership and adaptation responses. Forests. 2017;8:493. https://doi.org/10.3390/f8120493.
Nunes LJR, Meireles CIR, Pinto Gomes CJ, Almeida Ribeiro NMC. Forest contribution to climate change mitigation: management oriented to carbon capture and storage. Climate. 2020;8:21. https://doi.org/10.3390/cli8020021.
Acknowledgements
MKD acknowledges support from the START scholarship from the Foundation for Polish Science (FNP). Furthermore, this study was carried out in cooperation with the Agritech National Research Center—WP 4.1—Task 4.1.4 and received funding from the European Union Next-GenerationEU (PIANO NAZIONALE DI RIPRESA E RESILIENZA (PNRR) – MISSIONE 4 COMPONENTE 2, INVESTIMENTO 1.4 – D.D. 1032 17/06/2022, CN00000022). This manuscript reflects only the authors’ views and opinions, neither the European Union nor the European Commission can be considered responsible for them. Authors would like to thank three anonymous reviewers and the Editor for the valuable comments and suggestions.
Funding
The study was financed by the project AIMSUSFOR “Extending assessment of the environmental impacts to the forest ecosystem due to forest management: a comprehensive approach to enhance sustainable forestry in the context of climate change,” funded under the PASIFIC Call 1 announced by the Polish Academy of Sciences. This project has received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No 847639 and from the Polish Ministry of Education and Science. The study was further financed by the statutory research of the Institute of Dendrology PAS.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Francesco Latterini, Marcin K. Dyderski, Paweł Horodecki, Rodolfo Picchio, Rachele Venanzi, Katharina Lapin, and Andrzej M. Jagodziński declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Latterini, F., Dyderski, M.K., Horodecki, P. et al. The Effects of Forest Operations and Silvicultural Treatments on Litter Decomposition Rate: a Meta-analysis. Curr. For. Rep. 9, 276–290 (2023). https://doi.org/10.1007/s40725-023-00190-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s40725-023-00190-5