Abstract
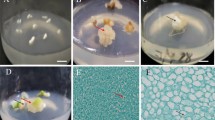
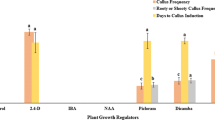
Teucrium (Teucrium polium L.) is one of the valuable medicinal plants, but its poor seed germination is one of the principal problems of its extensive cultivation. Somatic embryogenesis is crucial for medicinal plant genetic improvement, germplasm preservation, and in vitro mass propagation. Synthetic seed production is a suitable alternative for natural seed production in biotechnology and plant development. The objectives of the present study were to optimize somatic embryogenesis, produce synthetic seeds, and evaluate the genetic fidelity of the regenerated T. polium. Results suggested that MS medium supplemented with 1.0 mg L−1 kinetin (Kin) alone and without auxin demonstrated the maximum embryogenesis and regeneration rates in leaf and petiole explants. The simultaneous occurrence of several somatic embryonic developmental stages, such as the globular, heart-shaped, torpedo-shaped, and cotyledonary stages, on an embryogenic callus clearly suggested that T. polium embryogenesis accurately captures an asynchronous process. The petiole and leaf explants cultured on MS medium with 1.0 mg L−1 Kin showed the highest rates of plant regeneration with spontaneously root development. Compared to somatic embryos, synthetic seeds with meristem sources lacked callogenesis. At 25 °C for 4 wk and at 4 °C for 3 wk, the meristem source caused the highest percentage of synthetic seeds to germinate (96.66%), while − 18 °C had a negative impact on the germination of the synthetic seeds. The results suggested that there was no genetic variation identified in the ISSR analysis on in vitro–generated T. polium plants.








Similar content being viewed by others
Data Availability
Data are available from the corresponding author on request.
References
Ahmed AB, Mohajer S, Elnaiem EM, Taha RB (2012) In vitro regeneration, acclimatization, antimicrobial studies of selected ornamental plants. INTECH. https://doi.org/10.5772/50690
Alizadeh Z, Ahmadi DN, Mohammadi M, Karimizadeh RA, Memari HR, Bahmankar M (2016) Assessment of molecular variation in pea germplasm using RAPD markers. Electronic JBio 12:22–27
Al-Qudah TS, Shibli RA, Alali FQ (2011) In vitro propagation and secondary metabolites production in wild germander (Teucrium polium L.). In Vitro Cell Dev Biol - Plant 47:496–505. https://doi.org/10.1007/s11627-011-9352-9
Ammirato P, Steward FC (1971) Some effects of environment on the development of embryos from cultured free cells. Bot Gazett 132:149–158. https://doi.org/10.1086/336573
Bahmankar M, Mortazavian SMM, Tohidfar M, Sadat Noori SA, Izadi Darbandi A, Corrado G, Rao R (2017) Chemical compositions, somatic embryogenesis, and somaclonal variation in cumin. BioMed Res Intl 2017:7283806. https://doi.org/10.1155/2017/7283806
Bahmankar M, Mortazavian SMM, Tohidfar M, Sadat Noori SA, Izadi Darbandi A, Salehi M, Rao R (2018) Physio-biochemical characters, embryo regeneration and limonene synthase gene expression in cumin. Ind Crops Prod 121:195–205. https://doi.org/10.1016/j.indcrop.2018.05.006
Bakhtiar Z, Mirjalili MH, Sonboli A, MoridiFarimani M, Ayyari M (2014) In vitro propagation, genetic and phytochemical assessment of Thymus persicus — a medicinally important source of pentacyclic triterpenoids. Biologia 69:594–603. https://doi.org/10.2478/s11756-014-0346-z
Bhatia S, Bera T (2015) Classical and nonclassical techniques for secondary metabolite production in plant cell culture. In: S Bhatia, K Sharma, R Dahiya, T Bera (ed.) Modern Applications of Plant Biotechnology in Pharmaceutical Sciences. Elsevier, Netherlands. pp. 231– 291
Bukhari NA, Al-Otaibi RA, Ibhrahim MM (2015) Biodiversity characteristics of Teucrium polium species in Saudi Arabia. Saudi J Biol Sci 22:181–185. https://doi.org/10.1016/j.sjbs.2014.11.002
Chandrasekhara MR, Sri Rama Murthy K, Pullaiah T (2012) Synthetic seeds: a review in agriculture and forestry. Afr J Biotechnol 11:14254-14275. https://doi.org/10.5897/AJB12.770
Chauhan KR, Natarajan S, Webb M (2018) Dormancy breaking and germination of cat thyme Teucrium marum (Labiatae). African J Biotechnol 17:91–95. https://doi.org/10.5897/AJB2015.15012
Compton ME, Benton CM, Gray DJ, Songstad DD (1992) Plant recovery from maize somatic embryos subjected to controlled relative humidity dehydration. In Vitro Cell Dev Biol - Plant 28:197–201. https://doi.org/10.1007/BF02823317
Danso KE, Ford-Lloyd BV (2003) Encapsulation of nodal cuttings and shoot tips for storage and exchange of cassava germplasm. Plant Cell Rep 21:718–725. https://doi.org/10.1007/s00299-003-0594-9
Darvishzadeh R (2016) Detection of ISSR markers linked to seed oil biochemical characteristics in castor (Ricinus communis L.) through association analysis. Genetika 48:807–817. https://doi.org/10.2298/GENSR1603807D
Dave A, Batra A (1995) Role of protein metabolism constituents in somatic embryo formation in cumin. Indian J Plant Physiol 38:25–27
El Dawayati MM, EL Bar OHA, Zaid ZE, El Din AFMZ (2012) In vitro morpho-histological studies of newly developed embryos from abnormal malformed embryos of date palm cv Gundila under desiccation effect of polyethelyne glycol treatments. Ann Agri Sci 57:117–128. https://doi.org/10.1016/j.aoas.2012.08.005
Duncan DB (1955) Multiple range and multiple F tests. Biometrics 11:1. https://doi.org/10.2307/3001478
el Oualidi J, Verneau O, Puech S, Dubuisson J-Y (1999) Utility of rDNA ITS sequences in the systematics of Teucrium section Polium (Lamiaceae). Plant System Evol 215:49–70. https://doi.org/10.1007/BF00984647
Eshratifar M, Attar F, Mahdigholi K (2011) Micromorphological studies on nutlet and leaf indumentum of genus Teucrium L. (Lamiaceae) in Iran. Turkish J Bot 35:25–35. https://doi.org/10.3906/bot-0902-5
Esmaeili MA, Yazdanparast R (2004) Hypoglycaemic effect of Teucrium polium: studies with rat pancreatic islets. J Ethnopharmacol 95:27–30. https://doi.org/10.1016/j.jep.2004.06.023
Faisal M, Siddique I, Anis M (2006) An efficient plant regeneration system for Mucuna pruriens L. (DC.) using cotyledonary node explants. In Vitro Cell Dev Biol - Plant 42:59–64. https://doi.org/10.1079/IVP2005717
Fehér A (2003) Transition of somatic plant cells to an embryogenic state. Plant Cell Tiss Org Cult 74:201–228. https://doi.org/10.1023/A:1024033216561
Gaj MD (2004) Factors influencing somatic embryogenesis induction and plant regeneration with particular reference to Arabidopsis thaliana (L.) Heynh. Plant Grow Regul 43:27–47. https://doi.org/10.1023/B:GROW.0000038275.29262.fb
Ghasempour M, Iranbakhsh A, Ebadi M, OraghiArdebili Z (2019) Multi-walled carbon nanotubes improved growth, anatomy, physiology, secondary metabolism, and callus performance in Catharanthus roseus: an in vitro study. 3 Biotech 9:404. https://doi.org/10.1007/s13205-019-1934-y
GorginiShabankare H, Asgharipour MR, Fakheri B (2015) Morpho-chemical diversity among Iranian Teucrium polium L. (Lamiaceae) Populations in Fars Province. J Agric Sci Technol 17:705–716
Gray DJ, Conger BV, Songstad DD (1987) Desiccated quiescent somatic embryos of orchardgrass for use as synthetic seeds. In Vitro Cell Dev Biol 23:29–33. https://doi.org/10.1007/BF02623490
Grieb B, Schäfer F, Imani J, Mashayekhi F, Nezamabadi K, Arnholdt-Schmitt B, Neumann KH (1997) Changes in soluble proteins and phytohormone concentrations of cultured carrot petiole explants during induction of somatic embryogenesis (Daucus carota L.). Angewandte Bot 71:94–103
Guohua M (1998) Effects of cytokinins and auxins on cassava shoot organogenesis and somatic embryogenesis from somatic embryo explant. Plant Cell Tiss Org Cult 54:1–7. https://doi.org/10.1023/A:1006065120629
Hazarika BN (2006) Morpho-physiological disorders in in vitro culture of plants. Sci Hort 108:105–120. https://doi.org/10.1016/j.scienta.2006.01.038
Larkin PJ, Scowcroft WR (1981) Somaclonal variation — a novel source of variability from cell cultures for plant improvement. Theoret Appl Genet 60:197–214. https://doi.org/10.1007/BF02342540
Lim ZX, Pick A, Ling K, Hussein S (2009) Callus induction of Ocimum sanctum and estimation of its total flavonoids content. Asia J Agri Sci 1:55–61
Luna LG (1968) Manual of histologic staining methods of the Armed Forces Institute of Pathology, ed. 3. New York: McGraw-Hill
Mahin G, Ahmad M, Fathalah F, Kamal GB (2011) Comparison of callus induction and somatic embryogenesis of some Iranian cottons (Gossypium spp.) with Coker 312 and histology of somatic embryogenesis. African J Biotechnol 10:2915–2922. https://doi.org/10.5897/AJB10.2229
Martini AN, Papafotiou M, Majumder DAN (2016) Micropropagation of Teucrium capitatum L. Acta Hort 1113:183–188. https://doi.org/10.17660/ActaHortic.2016.1113.27
Moghanloo M, Iranbakhsh A, Ebadi M, Nejad ST, Oraghi AZ (2019a) Seed priming with cold plasma and supplementation of culture medium with silicon nanoparticle modified growth, physiology, and anatomy in Astragalus fridae as an endangered species. Acta Physiol Plant 41:54. https://doi.org/10.1007/s11738-019-2846-5
Moghanloo M, Iranbakhsh A, Ebadi M, Oraghi Ardebili Z (2019b) Differential physiology and expression of phenylalanine ammonia lyase (PAL) and universal stress protein (USP) in the endangered species Astragalus fridae following seed priming with cold plasma and manipulation of culture medium with silica nanoparticles. 3 Biotech 9:288. https://doi.org/10.1007/s13205-019-1822-5
Murashige T, Skoog F (1962) A revised medium for rapid growth and bio assays with tobacco tissue cultures. Physiol Plant 15:473–497. https://doi.org/10.1111/j.1399-3054.1962.tb08052.x
Murray MG, Thompson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucl Acid Res 8:4321–4326. https://doi.org/10.1093/nar/8.19.4321
Quiroz-Figueroa F, Fuentes-Cerda C, Rojas-Herrera R, Loyola-Vargas V (2002) Histological studies on the developmental stages and differentiation of two different somatic embryogenesis systems of Coffea arabica. Plant Cell Rep 20:1141–1149. https://doi.org/10.1007/s00299-002-0464-x
Rabba’a MM, Shibli RA, Shatnawi MA (2012) Cryopreservation of Teucrium polium L shoot-tips by vitrification and encapsulation-dehydration. Plant Cell Tiss Org Cult 110:371–382. https://doi.org/10.1007/s11240-012-0158-1
Rad FA, Jafari M, Khezrinejad N, Miandoab MP (2014) An efficient plant regeneration system via direct organogenesis with in vitro flavonoid accumulation and analysis of genetic fidelity among regenerants of Teucrium polium L. Hort Environ Biotechnol 55:568–577. https://doi.org/10.1007/s13580-014-0611-7
Rajaee Behbahani S, Iranbakhsh A, Ebadi M, Majd A,Oraghi Ardebili Z (2020) Red elemental selenium nanoparticles mediated substantial variations in growth, tissue differentiation, metabolism, gene transcription, epigenetic cytosine DNA methylation, and callogenesis in bitter melon (Momordica charantia); an in vitro experiment. PLOS One 15:e0235556. https://doi.org/10.1371/journal.pone.0235556
Ray S, Maiti S, Sa B (2008) Preliminary investigation on the development of diltiazem resin complex loaded carboxymethyl xanthan beads. AAPS PharmSciTech 9:295–301. https://doi.org/10.1208/s12249-007-9012-x
Rechinger KH, Wendelbo P (1982) Flora Iranica 150:749–781
Rihan H, Kareem F, El-Mahrouk M, Fuller M (2017) Artificial seeds (Principle, Aspects and Applications). Agronomy 7:71. https://doi.org/10.3390/agronomy7040071
Rout GR, Samantaray S, Das P (2000) In vitro manipulation and propagation of medicinal plants. Biotechnol Adv 18:91–120. https://doi.org/10.1016/S0734-9750(99)00026-9
Sabooni N, Shekafandeh A (2017) Somatic embryogenesis and plant regeneration of blackberry using the thin cell layer technique. Plant Cell Tiss Org Cult 130:313–321. https://doi.org/10.1007/s11240-017-1225-4
Sadeghi H, Jamalpoor S, Shirzadi MH (2014) Variability in essential oil of Teucrium polium L. of different latitudinal populations. Indust Crop Prod 54:130–134. https://doi.org/10.1016/j.indcrop.2014.01.015
Singh SK, Rai MK, Asthana P, Sahoo L (2010) Alginate-encapsulation of nodal segments for propagation, short-term conservation and germplasm exchange and distribution of Eclipta alba (L.). Acta Physiol Plant 32:607–610. https://doi.org/10.1007/s11738-009-0444-7
Soneji JR, Rao PS, Mhatre M (2002) In vitro regeneration from leaf explants of pineapple (Ananas comosus L, Merr). J Plant Biochem Biotechnol 11:117–119. https://doi.org/10.1007/BF03263147
Sotoodehnia-Korani S, Iranbakhsh A, Ebadi M, Majd A, OraghiArdebili Z (2020) Selenium nanoparticles induced variations in growth, morphology, anatomy, biochemistry, gene expression, and epigenetic DNA methylation in Capsicum annuum; an in vitro study. Environ Pollut 265:114727. https://doi.org/10.1016/j.envpol.2020.114727
Tawfik AA, Noga G (2001) Adventitious shoot proliferation from hypocotyl and internodal stem explants of cumin. Plant Cell Tiss Org Cult 66:141–147. https://doi.org/10.1023/A:1010622721314
Tawfik AA, Noga G (2002) Cumin regeneration from seedling derived embryogenic callus in response to amended kinetin. Plant Cell Tiss Org Cult 69:35–40. https://doi.org/10.1023/A:1015078409682
Tokuji Y, Kuriyama K (2003) Involvement of gibberellin and cytokinin in the formation of embryogenic cell clumps in carrot (Daucus carota). J Plant Physiol 160:133–141. https://doi.org/10.1078/0176-1617-00892
Venditti A, Frezza C, Trancanella E, MajdZadeh SM, Foddai S, Sciubba F, Delfini M, Serafini M, Bianco A (2017) A new natural neo -clerodane from Teucrium polium L. collected in northern Iran. Indust Crop Prod 97:632–638. https://doi.org/10.1016/j.indcrop.2017.01.010
West TP, Ravindra MB, Preece JE (2006) Encapsulation, cold storage, and growth of Hibiscus moscheutos nodal segments. Plant Cell Tiss Org Cult 87:223–231. https://doi.org/10.1007/s11240-006-9155-6
Wójcikowska B, Gaj MD (2017) Expression profiling of auxin response factor genes during somatic embryogenesis induction in Arabidopsis. Plant Cell Rep 36:843–858. https://doi.org/10.1007/s00299-017-2114-3
Zimmerman JL (1993) Somatic embryogenesis: a model for early development in higher plants. Plant Cell 1411–1423. https://doi.org/10.1105/tpc.5.10.1411
Acknowledgements
Dr. Moslem Bahmankar, Assistant Professor at the Seed and Plant Improvement Research Department, Safiabad Agricultural Research and Education and Natural Resources Center, AREEO, Dezful, Iran, is acknowledged by the authors for his valuable cooperation and detailed suggestions during the experiment.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Saadat, S., Majd, A., Naseri, L. et al. Optimization of somatic embryogenesis, synthetic seed production, and evaluation of genetic fidelity in Teucrium polium L.. In Vitro Cell.Dev.Biol.-Plant 59, 483–496 (2023). https://doi.org/10.1007/s11627-023-10360-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11627-023-10360-6