Abstract
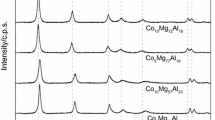
The CoAl hydroxides with Co/Al ratios of 2 and 4 were synthesized using traditional coprecipitation and a mechanochemical method. The structural properties of the samples at all stages of catalyst preparation, the transformations occurred on the reduction of cobalt from corresponding oxides, the textural characteristics of calcined and reduced systems, and the size, morphology, and composition of particles formed after high-temperature treatments were studied in detail. It was established that the synthesis procedure of CoAl hydroxides has a significant effect on the phase composition and properties of the resulting systems. The layered double hydroxide phase was formed only when the coprecipitation method was used. The mechanochemical approach made it possible to obtain materials with a higher specific surface area. According to TEM data, the samples synthesized by coprecipitation (after oxidative and reductive treatments) had a core–shell structure, where the core included metallic Co atoms and the shell mainly consisted of CoAl spinel. The samples prepared by the mechanochemical method had highly dispersed Co nanoparticles on their surface. The catalysts based on CoAl systems prepared by the mechanochemical method were more active in the hydrogenation of furfural: its conversion reached 97% on a sample with the ratio Co/Al = 4. In this case, the selectivity for furfuryl alcohol formation on the studied catalysts was almost 100% regardless of the synthesis procedure and Co/Al ratio.




Similar content being viewed by others
REFERENCES
Ekpeni, L.E.N., Benyounis, K.Y., Nkem-Ekpeni, F., Stokes, J., and Olabi, A.G., Energy Procedia, 2014, vol. 61, p. 1740.
Bozell, J.J. and Petersen, G.R., Green Chem., 2010, vol. 12, no. 4, p. 539.
Yan, K., Wu, G., Lafleur, T., and Jarvis, C., Renewable Sustainable Energy Rev., 2014, vol. 38, p. 663.
Mishra, D.K., Kumar, S., and Shukla, R.S., in Biomass, Biofuels, Biochemicals, Amsterdam: Elsevier, 2020, p. 323.
Bremner, J.G.M. and Keeys, R.K.F., J. Chem. Soc., 1947, p. 1068.
Fulajtárova, K., Soták, T., Hronec, M., Vávra, I., Dobročka, E., and Omastová, M., Appl. Catal., A, 2015, vol. 502, p. 78.
Mironenko, R.M., Belskaya, O.B., Talsi, V.P., and Likholobov, V.A., J. Catal., 2020, vol. 389, p. 721.
Taylor, M.J., Durndell, L.J., Isaacs, M.A., Parlett, C.M.A., Wilson, K., Lee, A.F., and Kyriakou, G., Appl. Catal. B: Environ., 2016, vol. 180, p. 580.
Bhogeswararao, S. and Srinivas, D., J. Catal., 2015, vol. 327, p. 65.
Audemar, M., Ciotonea, C., De Oliveira Vigier, K., Royer, S., Ungureanu, A., Dragoi, B., Dumitriu, E., and Jerome, F., ChemSusChem, 2015, vol. 8, no. 11, p. 1885.
Jiang, P., Li, X., Gao, W., Wang, X., Tang, Y., Lan, K., Wang, B., and Li, R., Catal. Commun., 2018, vol. 111, p. 6.
Gong, W., Chen, C., Zhang, H., Wang, G., and Zhao, H., Catal. Sci. Technol., 2018, vol. 8, no. 21, p. 5506.
Chen, X., Li, H., Luo, H., and Qiao, M., Appl. Catal. A: Gen., 2002, vol. 233, p. 13.
Srivastava, S., Mohanty, P., Parikh, J.K., Dalai, A.K., Amritphale, S.S., and Khare, A.K., Chin. J. Catal., 2015, vol. 36, no. 7, p. 933.
Mironenko, R.M., Likholobov, V.A., and Belskaya, O.B., Russ. Chem. Rev., 2022, vol. 91, no. 1, p. RCR5017.
Mascolo, G. and Mascolo, M.C., Micropor. Mesopor. Mater., 2015, vol. 214, p. 246.
Sulmonetti, T.P., Pang, S.H., Claure, M.T., Lee, S., Cullen, D.A., Agrawal, P.K., and Jones, C.W., Appl. Catal. A: Gen., 2016, vol. 517, p. 187.
Bertolini, G.R., Jiménez-Gómez, C.P., Cecilia, J.A., and Maireles-Torres, P., Catalysts, 2020, vol. 10, no. 5, p. 486.
Wu, J., Gao, G., Li, J., Sun, P., Long, X., and Li, F., Appl. Catal. B: Environ., 2017, vol. 203, p. 227.
Wang, T., Hu, A., Wang, H., and Xia, Y., J. Chin. Chem. Soc., 2019, vol. 66, no. 12, p. 1610.
Shao, Y., Wang, J., Sun, K., Gao, G., Li, C., Zhang, L., Zhang, S., Xu, L., Hu, G., and Hu, X., Renewable Energy, 2021, vol. 170, p. 1114.
Rudolf, C., Dragoi, B., Ungureanu, A., Chirieac, A., Royer, S., Nastro, A., and Dumitriu, E., Catal. Sci. Technol., 2014, vol. 4, no. 1, p. 179.
Biabani-Ravandi, A., Rezaei, M., and Fattah, Z., Proc. Saf. Environ. Prot., 2013, vol. 91, no. 6, p. 489.
Stepanova, L.N., Belskaya, O.B., Vasilevich, A.V., Leont’eva, N.N., Baklanova, O.N., and Likholobov, V.A, Kinet. Catal., 2018, vol. 59, no. 4, p. 521.
Lee, S.-B., Ko, E.-H., Park, J.Y., and Oh, J.-M., Nanomaterials, 2021, vol. 11, no. 5, p. 1153.
Bukhtiyarova, M.V., J. Solid State Chem., 2018, vol. 269, p. 494.
Tongamp, W., Zhang, Q., and Saito, F., Powder Technol., 2008, vol. 185, no. 1, p. 43.
Khusnutdinov, V.P. and Isupov, V.P., Inorg. Mater., 2008, vol. 44, no. 3, p. 263.
Stepanova, L.N., Kobzar, E.O., Leont’eva, N.N., Gulyaeva, T.I., Vasilevich, A.V., Babenko, A.V., Serkova, A.N., Salanov, A.N., and Belskaya, O.B., J. Alloys Compd., 2021, vol. 890, p. 161902.
Wang, B., Qu, J., Li, X., He, X., and Zhang, Q., J. Am. Ceram. Soc., 2016, vol. 99, no. 9, p. 2882.
Zhang, X. and Li, S., Appl. Surf. Sci., 2013, vol. 274, p. 158.
Zhu, J., Zeng, B., Mo, L., Jin, F., Deng, M., and Zhang, Q., Appl. Clay Sci., 2021, vol. 206, p. 106070.
Ay, A.N., Zümreoglu-Karan, B., and Mafra, L., Z. Anorg. Allg. Chem., 2009, vol. 635, no. 9, p. 1470.
Teodorescu, F., Slabu, A.I., Pavel, O.D., and Zăvoianu, R., Catal. Commun., 2019, vol. 133, p. 105829.
Kobzar, E.O., Stepanova, L.N., Leont’eva, N.N., and Belskaya, O.B., AIP Conf. Proc., 2020, vol. 2310, p. 030010.
Ferencz, Z., Kukovecz, Á., Kónya, Z., Sipos, P., and Pálinkó, I., Appl. Clay Sci., 2015, vol. 112, p. 94.
Ferencz, Z., Szabados, M., Adok-Sipiczki, M., Kukovecz, Á., Kónya, Z., Sipos, P., and Pálinkó, I., J. Mater. Sci., 2014, vol. 49, no. 24, p. 8478.
Qu, J., He, X., Chen, M., Huang, P., Zhang, Q., and Liu, X., J. Solid State Chem., 2017, vol. 250, p. 1.
Qu, J., He, X., Li, X., Ai, Z., Li, Y., Zhang, Q., and Liu, X., RSC Adv., 2017, vol. 7, no. 50, p. 31466.
Ferencz, Z., Szabados, M., Varga, G., Csendes, Z., Kukovecz, Á., Kónya, Z., Carlson, S., Sipos, P., and Pálinkó, I., J. Solid State Chem., 2016, vol. 233, p. 236.
Qu, J., He, X., Chen, M., Hu, H., Zhang, Q., and Liu, X., Mater. Chem. Phys., 2017, vol. 191, p. 173.
Qu, J., He, X., Wang, B., Zhong, L., Wan, L., Li, X., Song, S., and Zhang, Q., Appl. Clay Sci, 2016, vol. 120, p. 24.
Stepanova, L.N., Belskaya, O.B., Gulyaeva, T.I., Leont’eva, N.N., Salanov, A.N., and Likholobov, V., Catal. Today, 2019, vol. 357, p. 638.
Stepanova, L.N., Belskaya, O.B., Baklanova, O.N., Vasilevich, A.V., and Likholobov, V.A., Procedia Eng., 2016, vol. 152, p. 672.
Stepanova, L.N., Mironenko, R.M., Kobzar, E.O., Leont’eva, N.N., Gulyaeva, T.I., and Vasilevich, A.V., Salanov, and Lavrenov, A.V., ACS Sustain. Chem. Eng., 2022, vol. 3, no. 4, p. 400.
Wang, Y., Miao, Y., Li, S., Gao, L., and Xiao, G., Mol. Catal., 2017, vol. 436, p. 128.
Chen, X., Li, H., Luo, H., and Qiao, M., Appl. Catal., A, 2002, vol. 233, no. 1, p. 13.
Arnoldy, P. and Moulijn, J.A., J. Catal., 1985, vol. 93, no. 1, p. 38.
Ribet, S., Tichit, D., Coq, B., Ducourant, B., and Morato, F., J. Solid State Chem., 1999, vol. 142, no. 2, p. 382.
ACKNOWLEDGMENTS
The authors are grateful to I.V. Muromtsev for the study of samples by XRD analysis and to O.B. Bel’skaya for valuable comments on the material. The experiments were performed using the equipment of the Center for Collective Use “National Center for the Study of Catalysts” and the Omsk Center for Collective Use of the Siberian Branch of the Russian Academy of Sciences.
Funding
This work was financially supported by the Ministry of Science and Higher Education of the Russian Federation and performed within the framework of a state contract of the Boreskov Institute of Catalysis, Siberian Branch, Russian Academy of Sciences (project no. AAAA-A21-121011890074-4).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflicts of interest.
Additional information
Translated by V. Makhlyarchuk
Abbreviations and notation: FA, furfuryl alcohol; LDH, layered double hydroxides; XRD, X-ray diffraction analysis; BET, Brunauer–Emmett–Teller method; SBET, BET specific surface area; Vads, adsorption (total) pore volume; D, average pore diameter; TPR, temperature-programmed reduction; TEM, transmission electron microscopy; X, conversion of furfural; S, selectivity for product formation; PSDC, pore size distribution curve; FFT, fast Fourier transform technique.
Rights and permissions
About this article
Cite this article
Kobzar, E.O., Stepanova, L.N., Leont’eva, N.N. et al. Effect of the Composition and Synthesis Procedure of Catalysts Based on CoAl Hydroxides on Their Properties in Furfural Hydrogenation. Kinet Catal 64, 473–483 (2023). https://doi.org/10.1134/S0023158423040043
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0023158423040043