Abstract
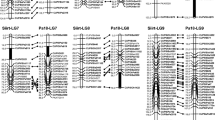
Pistachio (Pistacia vera L.) is one of the commercially important cultivated nut tree species in Turkey. Pistachio nuts have many beneficial properties to human health and are widely used in the food industry. Cultivar breeding programs in pistachio take a long time, huge labor, high cost, and large land areas due to the alternate bearing and the long juvenile period of pistachio. Molecular breeding approaches are widely used to shorten long-term breeding programs in pistachio. In this study, we aimed to (i) construct the first SNP-based high-density genetic linkage maps using an interspecific F1 population derived from a cross between P. vera cv. ‘Ohadi’ and P. atlantica cv. ‘Pa-18’ (‘Ohadi × Pa-18’), and (ii) perform QTL analysis in leaf, shoot, and inflorescence traits in pistachio. To obtain the SNP markers, the DArT-Seq™ genotyping-by-sequencing method was used. A total of 1407 SNPs were mapped in the ‘Ohadi’ female, whereas the ‘Pa-18’ male map consisted of a total of 2518 SNP markers. The total length of the ‘Ohadi’ map was 1122.2 cm with an average length of 74.8 cM, while the ‘Pa-18’ map was 1307.2 cM in length with an average length of 87.1 cM. The marker density was 1.2 and 1.9 markers per cM in ‘Ohadi’ and ‘Pa-18’ linkage maps, respectively. The ‘Ohadi’ map had an average of 93.8 markers per linkage group (LG), while the ‘Pa-18’ map had an average of 167.9 markers per linkage group. A total of four significant QTLs that are related to terminal leaflet base, leaf width, tree vigor, and inflorescence rachis length were detected in three linkage groups in the ‘Ohadi’ genetic linkage map. While the detected QTLs can be used for marker-assisted breeding and genetic studies, the generated SNP-based genetic linkage maps in this study will be used for QTL analysis of other important agronomic traits in pistachio.



Similar content being viewed by others
References
Ahmad R, Ferguson L, Southwick SM (2005) Molecular marker analyses of pistachio rootstocks by simple sequence repeats and sequence-related amplified polymorphisms. J Hortic Sci Biotech 80:382–386
Bourke PM, Gitonga VW, Voorrips RE et al (2018) Multi-environment QTL analysis of plant and flower morphological traits in tetraploid rose. Theor Appl Genet 131:2055–2069. https://doi.org/10.1007/s00122-018-3132-4
Brown PJ, Klein PE, Bortiri E et al (2006) Inheritance of inflorescence architecture in sorghum. Theor Appl Genet 113:931–942. https://doi.org/10.1007/s00122-006-0352-9
Chitwood DH, Headland LR, Kumar R et al (2012) The developmental trajectory of leaflet morphology in wild tomato species. Plant Physiol 158(3):1230–1240. https://doi.org/10.1104/pp.111.192518
Chitwood DH, Kumar R, Headland LR et al (2013) A quantitative genetic basis for leaf morphology in a set of precisely defined tomato introgression lines. Plant Cell 25(7):2465–2481. https://doi.org/10.1105/tpc.113.112391
Conner PJ, Brown SK, Weeden NF (1998) Molecular-marker analysis of quantitative traits for growth and development in juvenile apple trees. Theor Appl Genet 96:1027–1035. https://doi.org/10.1007/s001220050835
Cooper M, Gho C, Leafgren R et al (2014) Breeding drought-tolerant maize hybrids for the US corn-belt: discovery to product. J Exp Bot 65(21):6191–6204
Cronk QCB, Needham I, Rudall PJ (2015) Evolution of catkins: inflorescence morphology of selected Salicaceae in an evolutionary and developmental context. Front Plant Sci 6:1030. https://doi.org/10.3389/fpls.2015.01030
Doyle J, Doyle J (1987) A rapid isolation procedure for small amounts of leaf tissue. Phytochem Bull. https://doi.org/10.2307/4119796
Elshire RJ, Glaubitz JC, Sun Q et al (2011) A robust, simple genotyping-by-sequencing (GBS) approach for high diversity species. PloS One 6(5):e19379. https://doi.org/10.1371/journal.pone.0019379
Faostat (2022) FAOSTAT: Agriculture Data. Available online
Foster TM, Celton JM, Chagne D et al (2015) Two quantitative trait loci, Dw1 and Dw2, are primarily responsible for rootstock-induced dwarfing in apple. Hortic Res 2. https://doi.org/10.1038/hortres.2015.1
Grattapaglia D, Sederoff R (1994) Genetic linkage maps of Eucalyptus grandis and Eucalyptus urophylla using a pseudo-testcross: mapping strategy and RAPD markers. Genetics. https://doi.org/10.1093/genetics/137.4.1121
Han TH, Van Eck HJ, De Jeu MJ, Jacobsen E (2002) Mapping of quantitative trait loci involved in ornamental traits in Alstroemeria. HortScience 37(3):585–592. https://doi.org/10.21273/hortsci.37.3.585
Harlan JR, Wet JMJ (1972) A simplified classification of cultivated sorghum 1. Crop Sci 12(2):172–176. https://doi.org/10.2135/cropsci1972.0011183x001200020005x
Hermann K, Kuhlemeier C (2011) The genetic architecture of natural variation in flower morphology. Curr Opin Plant Biol 14(1):60–65
Hofer J, Turner L, Hellens R et al (1997) UNIFOLIATA regulates leaf and flower morphogenesis in pea. Curr Biol 7(8):581–587. https://doi.org/10.1016/S0960-9822(06)00257-0
Horiguchi G, Kim GT, Tsukaya H (2005) The transcription factor AtGRF5 and the transcription coactivator AN3 regulate cell proliferation in leaf primordia of Arabidopsis thaliana. Plant J 43(1):68–78. https://doi.org/10.1111/j.1365-313X.2005.02429.x
Hormaza JI, Dollo L, Polito VS (1994a) Determination of relatedness and geographical movements of Pistacia vera (Pistachio; Anacardiaceae) germplasm by RAPD analysis. Econ Bot 48:349–358. https://doi.org/10.1007/BF02862231
Hormaza JI, Dollo L, Polito VS (1994b) Identification of a RAPD marker linked to sex determination in Pistacia vera using bulked segregant analysis. Theor Appl Genet 89:9–13. https://doi.org/10.1007/BF00226975
Hormaza JI, Pinney K, Polito VS (1998) Genetic diversity of pistachio (Pistacia vera, Anacardiaceae) germplasm based on randomly amplified polymorphic DNA (RAPD) markers. Econ Bot 52:78–87. https://doi.org/10.1007/BF02861298
Huang X, Paulo MJ, Boer M et al (2011) Analysis of natural allelic variation in Arabidopsis using a multiparent recombinant inbred line population. Proc Natl Acad Sci U S A 108(11):4488–4493. https://doi.org/10.1073/pnas.1100465108
IPGRI (1997) Descriptors for Pistachio (Pistacia vera L.) International Plant Genetic Resources Institute, Rome, Italy
Kafkas S (2002) Developing of monoecious pistachio (P. vera L.) populations and the sex determination mechanism in Pistacia by crossbreeding. In: Acta Horticulturae, pp 285–289
Kafkas S (2006) Phylogenetic analysis of the genus Pistacia by AFLP markers. Plant Syst Evol 262:113–124. https://doi.org/10.1007/s00606-006-0460-7
Kafkas S (2019) Advances in Breeding of Pistachio, Advances in genetic and breeding of nuts. Burleigh Dodds Science Publishing. https://doi.org/10.19103/AS.2018.0042.17
Kafkas S, Çetiner S, Perl-Treves R (2001) Development of sex-associated RAPD markers in wild Pistacia species. J Hortic Sci Biotech 76(2):242–246. https://doi.org/10.1080/14620316.2001.11511358
Kafkas S, Kaska N, Wassimi AN, Padulosi S (2006a) Molecular characterisation of Afghan pistachio accessions by amplified fragment length polymorphisms (AFLPs). J Hortic Sci Biotech 81(5):864–868. https://doi.org/10.1080/14620316.2006.11512151
Kafkas S, Khodaeiaminjan M, Güney M, Kafkas E (2015) Identification of sex-linked SNP markers using RAD sequencing suggests ZW/ZZ sex determination in Pistacia vera L. BMC Genomics 16:98. https://doi.org/10.1186/s12864-015-1326-6
Kafkas S, Gözel H, Bozkurt H, Karci H, Topcu H, Paizila A, Kefayati S, Islam RMd, Zhaanbaev M, Kafkas E, Acar I, Uzun M (2017) Marker-assisted cultivar breeding in pistachio. VII International Symposium on Almonds and Pistachios, abstract book, p 67
Kafkas S, Ma X, Zhang X et al (2022) The pistachio genomes provide insights into nut tree domestication and ZW sex chromosome evolution. Plant Commun 4:100497
Kafkas S, Ozkan H, Ak BE et al (2006b) Detecting DNA polymorphism and genetic diversity in a wide pistachio germplasm: comparison of AFLP, ISSR, and RAPD markers. J Am Soc Hort Sci 131(4):522–529. https://doi.org/10.21273/jashs.131.4.522
Kafkas S, Perl-Treves R (2001) Morphological and molecular phylogeny of Pistacia species in Turkey. Theor Appl Genet 102:908–915. https://doi.org/10.1007/s001220000526
Kafkas S, Perl-Treves R, Kaska N (2000) Unusual Pistacia atlantica Desf. (Anacardiaceae) monoecious sex type in the Yunt Mountains of the Manisa province of Turkey. Isr J Plant Sci 48(4):277–280. https://doi.org/10.1092/UFCU-7LF6-T0A3-UXWY
Karimi HR, Kafkas S (2011) Genetic relationships among Pistacia species studied by SAMPL markers. Plant Syst Evol 297:207–212. https://doi.org/10.1007/s00606-011-0508-1
Karimi S, Rahemi M, Maftoun M et al (2009) Effects of long-term salinity on growth and performance of two pistachio (Pistacia L.) rootstocks. Aust J Basic Appl Sci 3:1630–1639
Khodaeiaminjan M, Kafkas S, Motalebipour EZ, Coban N (2018) In silico polymorphic novel SSR marker development and the first SSR-based genetic linkage map in pistachio. Tree Genet Genomes 14:45. https://doi.org/10.1007/s11295-018-1259-8
Kilian A, Wenzl P, Huttner E et al (2012) Diversity arrays technology: a generic genome profiling technology on open platforms BT - data production and analysis in population genomics: methods and protocols. In: Methods in Molecular Biology, pp 67–89
Kimber CT, Dahlberg JA, Kresovich S (2013) The gene pool of sorghum bicolor and its improvement. Genomics of the Saccharinae:23–41
Kosambi DD (2016) The estimation of map distances from recombination values. In: D.D. Kosambi, pp 125–130
Luo C, Shu B, Yao Q, Wu H, Xu W, Wang S (2016) Construction of a High-Density Genetic Map Based on Large-Scale Marker Development in Mango Using Specific-Locus Amplified Fragment Sequencing (SLAF-seq). Front Plant Sci. https://doi.org/10.3389/fpls.2016.01310
Motalebipour EZ, Gozel H, Khodaeiaminjan M, Kafkas S (2018) SSR-based genetic linkage map construction in pistachio using an interspecific F1 population and QTL analysis for leaf and shoot traits. Mol Breed 38:134. https://doi.org/10.1007/s11032-018-0893-z
Mzena GP, Kusolwa PM, Rwegasira GM, Yao NK (2018) Discovery of novel single nucleotide polymorphic (SNP) markers for genetic mapping of cashew (Anacardium occidentale L.). Int J Agric Environ Biores
Nguyen DT, Hayes JE, Harris J, Sutton T (2022) Fine mapping of a vigor QTL in chickpea (Cicer arietinum L.) Reveals a Potential Role for Ca4_TIFY4B in Regulating Leaf and Seed Size. Front Plant Sci 13:829566. https://doi.org/10.3389/fpls.2022.829566
Nicotra AB, Leigh A, Boyce CK et al (2011) The evolution and functional significance of leaf shape in the angiosperms. Funct Plant Biol 38(7):535–552
Parfitt DE, Badenes ML (1997) Phylogeny of the genus Pistacia as determined from analysis of the chloroplast genome. Proc Natl Acad Sci 94(15):7987–7992
Şahin-Çevik M, Moore GA (2012) Quantitative trait loci analysis of morphological traits in citrus. Plant Biotechnol Rep 6:47–57. https://doi.org/10.1007/s11816-011-0194-z
Sansaloni C, Petroli C, Jaccoud D et al (2011) Diversity Arrays Technology (DArT) and next-generation sequencing combined: genome-wide, high throughput, highly informative genotyping for molecular breeding of Eucalyptus. In: BMC proceedings. BioMed Central. https://doi.org/10.1186/1753-6561-5-s7-p54
Segura V, Denancé C, Durel CE, Costes E (2007) Wide range QTL analysis for complex architectural traits in a 1-year-old apple progeny. Genome 50(2):159–171. https://doi.org/10.1139/G07-002
Street NR, Sjödin A, Bylesjö M et al (2008) A cross-species transcriptomics approach to identify genes involved in leaf development. BMC Genomics 9:589. https://doi.org/10.1186/1471-2164-9-589
Turkeli Y, Kafkas S (2013) First genetic linkage map in pistachio constructed using an interspecific cross between Pistacia vera L. and monoecious Pistacia atlantica Desf. Sci Hortic 151:30–37. https://doi.org/10.1016/j.scienta.2012.11.024
Van Ooijen JW (2004) MapQTL® 5, software for the mapping of quantitative trait loci in experimental populations. Kyazma B V
Van Ooijen JW (2006) JoinMap ® 4 software for the calculation of genetic linkage maps in experimental populations. JoinMap
Virlet N, Costes E, Martinez S et al (2015) Multispectral airborne imagery in the field reveals genetic determinisms of morphological and transpiration traits of an apple tree hybrid population in response to water deficit. J Exp Bot 66(18):5453–5465. https://doi.org/10.1093/jxb/erv355
Voorrips RE (2002) MapChart: software for the graphical presentation of linkage maps and QTLs. J Hered 93(1):77–78
Witt Hmon KP, Shehzad T, Okuno K (2013) Variation in inflorescence architecture associated with yield components in a sorghum germplasm. Plant Genet Resour Characterisation Util 11(3):258–265. https://doi.org/10.1017/S1479262113000154
Zaloğlu S, Kafkas S, Doğan Y, Güney M (2015) Development and characterization of SSR markers from pistachio (Pistacia vera L.) and their transferability to eight Pistacia species. Sci Hortic (Amsterdam) 189:94–103. https://doi.org/10.1016/j.scienta.2015.04.006
Zhang D, Yuan Z (2014) Molecular control of grass inflorescence development. Annu Rev Plant Biol 65:553–578
Data archiving statement
All data generated or analyzed during this study are included in this published article and its supplementary information files. The name of the loci, their positions, segregations, X2, and significance (p) belonging to maps are shared in Supplementary file-1. The correlation of a total of 18 observations and measurements of ‘Ohadi × Pa-18’ F1 individuals are calculated and plotted in Supplementary file-2. Their mean, max., min., and standard deviations are given in Supplementary file-3. The linked loci associated with traits were blasted to pistachio genome and gene IDs, and their descriptions are shared in Supplementary file-4.
Funding
This research was supported by TÜBİTAK (project no.: TOVAG 100O113). The authors thank the University Çukurova, Scientific Research Projects Unit, and TÜBİTAK for financial support.
Author information
Authors and Affiliations
Contributions
MRI and SK conceived and designed the experiments. MRI and HT performed most of the experiments and analyzed the data. HTo and HK assisted with the experiments. MRI, HK, and SK wrote the manuscript. The authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable
Consent for publication
All authors have read and approved the manuscript for publication.
Competing interests
The authors declare no competing interests.
Additional information
Communicated by V. Decroocq
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Islam, M.R., Tevfik, H., Topçu, H. et al. Construction of a high-density genetic linkage map and QTL analysis using an interspecific F1 population in pistachio. Tree Genetics & Genomes 19, 37 (2023). https://doi.org/10.1007/s11295-023-01613-x
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11295-023-01613-x