Abstract
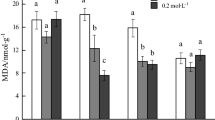
Among non-essential heavy metals, cadmium (Cd) pollution in soil continuously increasing by anthropogenic activity such as agriculture fertilizer and industrial sources worldwide and have several harmful impacts on plants and humans. Andrographis paniculata is a highly medicinal plant having diverse range of SMs like andrographolide (AG). The present study was carried out to investigate the effect of Cd on growth, activity of antioxidative enzymes and andrographolide production of A. paniculata. Cd was applied as cadmium chloride (CdCl2) at 5, 10 and 15 μg g−1 of soil. All the studied treatments of Cd were found to be toxic for A. paniculata. The root-shoot length, root-shoot fresh weight, root-shoot dry weight decreased with increasing concentrations of Cd. Total phenolics and flavonoids content significantly increased with increasing Cd concentration in soil. The activity of catalase (CAT), peroxidase (POX), ascorbate peroxidase (APX), glutathione reductase (GR), guaiacol peroxidase (GPX) and polyphenol oxidase (PPO) increased with increasing concentration of Cd compared to control plants. The maximum activity of CAT, POX, APX, GR, GPX and PPO was observed at 15 µg Cd g−1. The AG content in the leaves of plants increased with Cd application. Maximum AG content was observed at 10 μg g−1 applied dose of Cd. Out of the concentrations studied 5 and 10 μg g−1 were moderately toxic to A. paniculata and produced maximum amount of AG.










Similar content being viewed by others
Data availability
The data supporting this study’s findings are available from the corresponding author upon reasonable request.
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 105:121–126. https://doi.org/10.1016/S0076-6879(84)05016-3
Ahmad P, Abd Allah EF, Hashem A, Sarwat M, Gucel S (2016) Exogenous application of selenium mitigates cadmium toxicity in Brassica juncea L. (Czern & Cross) by up-regulating antioxidative system and secondary metabolites. J Plant Growth Regul 35:936–950. https://doi.org/10.1007/s00344-016-9592-3
Anjum SA, Tanveer M, Hussain S, Bao M, Wang L, Khan I, Ullah E, Tung SA, Samad RA, Shahzad B (2015) Cadmium toxicity in Maize (Zea mays L.): consequences on antioxidative systems, reactive oxygen species and cadmium accumulation. Environ Sci Pollut Res 22:17022–17030. https://doi.org/10.1007/s11356-015-4882-z
Antony A, Nagella P (2021) Heavy metal stress influence the andrographolide content, phytochemicals and antioxidant activity of Andrographis paniculata. Plant Sci Today 8(2):324–330. https://doi.org/10.14719/pst.2021.8.2.1034
Apel K, Hirt H (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55:373–399. https://doi.org/10.1146/annurev.arplant.55.031903.141701
Atanassova M, Georgieva S, Ivancheva K (2011) Total phenolic and total flavonoid contents, antioxidant capacity and biological contaminants in medicinal herbs. J Chem Technol Metall 46(1):81–88
Bajhol SH (2014) Abiotic stress mediated consequences on antioxidant defence system and secondary metabolites in Andrographis paniculata wall. ex nees. Dissertation, Shoolini University of Biotechnology and Management sciences.
Bhattacharya S, Puri S, Jamwal A, Sharma S (2012) Studies on seed germination and seedling growth in Kalmegh (Andrographis paniculata Wall. ex Nees) under abiotic stress conditions. Int J Environ Sci Technol 1(3):197–204
Das D, Bandyopadhyay M (2020) Novel approaches towards over-production of andrographolide in in vitro seedling cultures of Andrographis paniculata. S Afr J Bot 128:77–86. https://doi.org/10.1016/j.sajb.2019.10.015
Das P, Khare P, Singh RP, Yadav V, Tripathi P, Kumar A, Pandey V, Gaur P, Singh A, Das R, Hiremath C (2021) Arsenic-induced differential expression of oxidative stress and secondary metabolite content in two genotypes of Andrographis paniculata. J Hazard Mater 406:124302. https://doi.org/10.1016/j.jhazmat.2020.124302
Dawande AA, Sahay S (2021) Copper sulphate elicitation of optimized suspension culture of Andrographis paniculata Nees yields unprecedented level of andrographolide. J Microbiol Biotechnol Food Sci 2021:688–694. https://doi.org/10.15414/jmbfs.2020.9.4.688-694
Ekmekçi Y, Tanyolac D, Ayhan B (2008) Effects of cadmium on antioxidant enzyme and photosynthetic activities in leaves of two maize cultivars. J Plant Physiol 165(6):600–611. https://doi.org/10.1016/j.jplph.2007.01.017
Emanuil N, Akram MS, Ali S, El-Esawi MA, Iqbal M, Alyemeni MN (2020) Peptone-induced physio-biochemical modulations reduce cadmium toxicity and accumulation in spinach (Spinacia oleracea L.). Plants 9(12):1806. https://doi.org/10.3390/plants9121806
Gong M, Li YJ, Chen SZ (1998) Abscisic acid-induced thermotolerance in maize seedlings is mediated by calcium and associated with antioxidant systems. J Plant Physiol 153(3–4):488–496
Haider FU, Liqun C, Coulter JA, Cheema SA, Wu J, Zhang R, Wenjun M, Farooq M (2021) Cadmium toxicity in plants: Impacts and remediation strategies. Ecotoxicol Environ Saf 211:111887. https://doi.org/10.1016/j.ecoenv.2020.111887
Hassan MJ, Raza MA, Rehman SU, Ansar M, Gitari H, Khan I, Wajid M, Ahmed M, Shah GA, Peng Y, Li Z (2020) Effect of cadmium toxicity on growth, oxidative damage, antioxidant Défense system and cadmium accumulation in two sorghum cultivars. Plants 9(11):1575. https://doi.org/10.3390/2Fplants9111575
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts: I. kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125(1):189–198. https://doi.org/10.1016/0003-9861(68)90654-1
Irfan M, Ahmad A, Hayat S (2014) Effect of cadmium on the growth and antioxidant enzymes in two varieties of Brassica juncea. Saudi J Biol Sci 21(2):125–131. https://doi.org/10.1016/2Fj.sjbs.2013.08.001
Jamwal K, Bhattacharya S, Puri S (2018) Plant growth regulator mediated consequences of secondary metabolites in medicinal plants. J Appl Res Med Aromat Plants 9:26–38. https://doi.org/10.1016/j.jarmap.2017.12.003
Jan R, Asaf S, Numan M, Kim KM (2021) Plant secondary metabolite biosynthesis and transcriptional regulation in response to biotic and abiotic stress conditions. Agron 11(5):968. https://doi.org/10.3390/agronomy11050968
Jibril SA, Hassan SA, Ishak CF, Megat Wahab PE (2017) Cadmium toxicity affects phytochemicals and nutrient elements composition of lettuce (Lactuca sativa L.). Adv Agric 2017:1–7. https://doi.org/10.1155/2017/1236830
Joselin J, Jeeva S (2014) Andrographis paniculata: a review of its traditional uses. Phytochemistry and Pharmacology Med Aromat Plants 3(4):1000169. https://doi.org/10.4172/2167-0412.1000169
Joseph SM (2014) Scientific aspects of the therapeutic use of Andrographis paniculata (Kalmegh): a review. Int J Pharm Sci Rev Res 27:10–16
Khan MA, Khan S, Khan A, Alam M (2017) Soil contamination with cadmium, consequences and remediation using organic amendments. Sci Total Environ 601:1591–1605. https://doi.org/10.1016/j.scitotenv.2017.06.030
Kobyletska M, Kavulych Y, Romanyuk N, Korchynska O, Terek O (2022) Exogenous salicylic acid modifies cell wall lignification, total phenolic content, PAL-activity in wheat (Triticum aestivum L.) and buckwheat (Fagopyrum esculentum Moench) plants under cadmium chloride impact. Biointerface Res Appl Chem 13:117
Kumar A, Aery NC (2011) Effect of tungsten on growth, biochemical constituents, molybdenum and tungsten contents in wheat. Plant Soil Environ 57(11):519–525. https://doi.org/10.17221/345/2011-pse
Kumar S, Singh B, Bajpai V (2021) Andrographis paniculata (Burm. F.) Nees: Traditional uses, phytochemistry, pharmacological properties and quality control/quality assurance. J Ethnopharmacol 275:114054. https://doi.org/10.1016/j.jep.2021.114054
Kupper H, Kupper F, Spiller M (1996) Environmental relevance of heavy metal-substituted chlorophylls using the example of water plants. J Exp Bot 47(2):259–266. https://doi.org/10.1093/jxb/47.2.259
Latif J, Akhtar J, Ahmad I, Mahmood-ur-Rehman M, Shah GM, Zaman Q, Javaid T, Farooqi ZU, Shakar M, Saleem A, Rizwan M (2020) Unraveling the effects of cadmium on growth, physiology and associated health risks of leafy vegetables. Rev Bras Bot 43:799–811
Li X, Lan X, Feng X, Luan X, Cao X, Cui Z (2021) Biosorption capacity of Mucor circinelloides bioaugmented with Solanum nigrum L. for the cleanup of lead, cadmium and arsenic. Ecotoxicol Environ Saf 212:112014. https://doi.org/10.1016/j.ecoenv.2021.112014
Mehta S, Sharma AK, Singh RK (2021) Therapeutic journey of Andrographis paniculata (Burm. F.) Nees from natural to synthetic and nanoformulations. Mini Rev Med Chem 21(12):1556–1577. https://doi.org/10.2174/1389557521666210315162354
Mourya SK, Mohil P, Kumar A (2022) Abiotic stress-induced changes in antioxidative system and secondary metabolites production in Andrographis paniculata. J Stress Physiol Biochem 18(3):69–83
Nair DS, Manjula S (2020) Induction of root endosymbiosis as a highly sustainable and efficient strategy for overproduction of the medicinally important diterpenoid lactone-andrographolide in Andrographis paniculata (Burm. F.) Wall ex Nees. Ind Crops Prod 156:112835
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22(5):867–880
Nazar R, Iqbal N, Masood A, Khan MI, Syeed S, Khan NA (2012) Cadmium toxicity in plants and role of mineral nutrients in its alleviation. Am J Plant Sci 3:1476–1489. https://doi.org/10.4236/ajps.2012.310178
Okhuarobo A, Falodun JE, Erharuyi O, Imieje V, Falodun A, Langer P (2014) Harnessing the medicinal properties of Andrographis paniculata for diseases and beyond: a review of its phytochemistry and pharmacology. Asian Pac J Trop Dis 4(3):213–222. https://doi.org/10.1016/S2222-1808(14)60509-0
Pirselova B, Kuna R, Lukac P, Havrlentova M (2016) Effect of cadmium on growth, photosynthetic pigments, iron and cadmium accumulation of Faba Bean (cv. Astar). Agriculture 62(2):72–79. https://doi.org/10.1515/agri-2016-0008
Schaedle M, Bassham JA (1977) Chloroplast glutathione reductase. Plant Physiol 59(5):1011–1012. https://doi.org/10.1104/2Fpp.59.5.1011
Sharma SN, Jha Z, Sinha RK, Geda AK (2015) Jasmonate-induced biosynthesis of andrographolide in Andrographis paniculata. Physiol Plant 153(2):221–229. https://doi.org/10.1111/ppl.12252
Simoes AD, Moreira SI, Mosquim PR, Soares ND, Puschmann R (2015) The effects of storage temperature on the quality and phenolic metabolism of whole and minimally processed kale leaves. Acta Sci Agron 37(1):101–107. https://doi.org/10.4025/actasciagron.v37i1.18123
Siripong P, Kongkathip B, Preechanukool K, Picha P, Tunsuwan K, Taylor WC (1992) Cytotoxic diterpenoid constituents from Andrographis paniculata Nees leaves. J Sci Soc Thail 18(4):187–194. https://doi.org/10.2306/scienceasia1513-1874.1992.18.187
Tran TA, Popova LP (2013) Functions and toxicity of cadmium in plants: recent advances and future prospects. Turk J Bot 37(1):1–3
Uddin ABM, Khalid R, Alaama M, Abdualkader AM, Kasmuri A, Abbas SA (2016) Comparative study of three digestion methods for elemental analysis in traditional medicine products using atomic absorption spectrometry. J Anal Sci Technol 7(1):1–7
Waterhouse AL (2002) Determination of total phenolics. Curr Protocol Food Anal Chem 6(1):1–1
Yao W, An T, Xu Z, Zhang L, Gao H, Sun W, Liao B, Jiang C, Liu Z, Duan L, Ji A (2020) Genomic-wide identification and expression analysis of AP2/ERF transcription factors related to andrographolide biosynthesis in Andrographis paniculata. Ind Crops Prod 157:112878. https://doi.org/10.1016/j.indcrop.2020.112878
Younis U, Malik SA, Rizwan M, Qayyum MF, Ok YS, Shah MH, Rehman RA, Ahmad N (2016) Biochar enhances the cadmium tolerance in spinach (Spinacia oleracea) through modification of Cd uptake and physiological and biochemical attributes. Environ Sci Pollut Res 23:21385–21394. https://doi.org/10.1007/s11356-016-7344-3
Zaheer M, Giri CC (2015) Multiple shoot induction and jasmonic versus salicylic acid driven elicitation for enhanced andrographolide production in Andrographis paniculata. Plant Cell Tissue Organ Cult 122:553–563
Zhang XZ (1992) The measurement and mechanism of lipid peroxidation and SOD, POD and CAT activities in biological system. Research methodology of crop physiology. Agriculture press, Beijing, 208–211.
Zhao HJ, Tan JF (2005) Role of calcium ion in protection against heat and high irradiance stress-induced oxidative damage to photosynthesis of wheat leaves. Photosynthetica 43:473–476
Acknowledgements
The authors are grateful to MHRD, New Delhi for the financial help as RUSA 2.0. SKM is thankful to CSIR New Delhi for providing financial support in the form of SRF. Authors are thankful to Soil testing laboratory, RARI, Durgapura, Jaipur for soil analysis and Materials Research Centre (MRC), MNIT, Jaipur for providing AAS facility for Cd analysis.
Funding
This research received no specific grant from any funding agency.
Author information
Authors and Affiliations
Contributions
AK conceived the idea and designed the research plan. SKM executes the experiment and writing work, data analysis and prepares all figures. RV helped in the estimation of andrographolide content and interprets the results. AK and PM helped in results interpretation, and reviewing the manuscript for publication.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflicts of interest.
Additional information
Communicated by Marko Rohlfs.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mourya, S.K., Mohil, P., Vijayvergia, R. et al. Cadmium induced changes in antioxidant activity, oxidative damage and andrographolide production in Andrographis paniculata. Chemoecology 33, 125–134 (2023). https://doi.org/10.1007/s00049-023-00390-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00049-023-00390-2