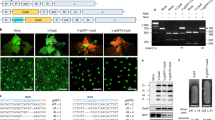
Abstract
CRISPR/Cas9 ribonucleoproteins enable DNA-free genome editing; thus, improving protoplast culture is essential for the efficient development of mutant plants. However, the use of protoplast cultures is limited because a universal method cannot be applied to diverse plants. Solanum lycopersicum ‘Heinz 1706,’ a model cultivar for tomato genome analysis, has not yet been studied for DNA-free genome editing. We optimized the protoplast culture method for the tomato model cultivar ‘Heinz 1706’ using combinations of plant growth regulators (PGRs) and basal media. Isolated protoplasts were cultured in R-Ini medium for cell division and micro-calli proliferation, and then the medium was changed to G-207.3 medium for callus formation. Among different concentrations of 1-naphthaleneacetic acid (NAA) and 6-benzylaminopurine (BAP), the combination of 0.05 mg/L NAA and 0.5 mg/L BAP in the R-Ini medium was observed to be highly efficient for micro-calli development. G-207.3 liquid medium was more efficient for mini-calli formation than the R-Ini liquid medium. For calli formation from the mini-calli, 0.1 mg/L 2,4-dichlorophenoxyacetic acid (2,4-D), 0.05 mg/L NAA, and 0.5 mg/L BAP were used in G-207.3 solid medium. In addition, five single guide RNAs (sgRNAs) were designed to target SlPelo using polyethylene glycol-mediated transfection, thereby developing a tool for tomato yellow leaf curl virus (TYLCV) resistance breeding. sgRNAs and Cas9 complexes were delivered into protoplasts using PEG-mediated transfection, and sgRNA2 resulted in a high mutagenesis efficiency. The results will be valuable for DNA-free genome editing in tomato for the development of new breeding materials.





Similar content being viewed by others
Data Availability
All data generated or analyzed during this study are included in this published article.
References
Abebe AM, Choi J, Kim Y, Oh C-S, Yeam I, Nou I-S, Lee JM (2020) Development of diagnostic molecular markers for marker-assisted breeding against bacterial wilt in tomato. Breed Sci 70:462–473. https://doi.org/10.1270/jsbbs.20027
Abebe AM, Oh C-S, Kim HT, Choi G, Seo E, Yeam I, Lee JM (2022) QTL-Seq analysis for identification of resistance loci to bacterial canker in Tomato. Front Plant Sci 12. https://doi.org/10.3389/fpls.2021.809959
Altpeter F, Springer NM, Bartley LE, Blechl AE, Brutnell TP, Citovsky V, Conrad LJ, Gelvin SB, Jackson DP, Kausch AP (2016) Advancing crop transformation in the era of genome editing. Plant Cell 28:1510–1520. https://doi.org/10.1105/tpc.16.00196
Anbinder I, Reuveni M, Azari R, Paran I, Nahon S, Shlomo H, Chen L, Lapidot M, Levin I (2009) Molecular dissection of Tomato leaf curl virus resistance in tomato line TY172 derived from Solanum peruvianum. Theor Appl Genet 119:519–530. https://doi.org/10.1007/s00122-009-1060-z
Andersson M, Turesson H, Olsson N, Fält AS, Ohlsson P, Gonzalez MN, Samuelsson M, Hofvander P (2018) Genome editing in potato via CRISPR-Cas9 ribonucleoprotein delivery. Physiol Plant 164:378–384. https://doi.org/10.1111/ppl.12731
Becker T, Franckenberg S, Wickles S, Shoemaker CJ, Anger AM, Armache J-P, Sieber H, Ungewickell C, Berninghausen O, Daberkow I, Karcher A, Thomm M, Hopfner K-P, Green R, Beckmann R (2012) Structural basis of highly conserved ribosome recycling in eukaryotes and archaea. Nature 482:501–506. https://doi.org/10.1038/nature10829
Bortesi L, Fischer R (2015) The CRISPR/Cas9 system for plant genome editing and beyond. Biotechnol Adv 33:41–52. https://doi.org/10.1016/j.biotechadv.2014.12.006
Cao Y, Zhou H, Zhou X, Li F (2020) Control of plant viruses by CRISPR/Cas system-mediated adaptive immunity. Front Microbiol 11:593700–593700. https://doi.org/10.3389/fmicb.2020.593700
Chinalia FA, Regali-Seleghin MH, Correa EM (2007) 2, 4-D toxicity: cause, effect and control. TAET 1:24–33
Cong L, Ran FA, Cox D, Lin S, Barretto R, Habib N, Hsu PD, Wu X, Jiang W, Marraffini LA, Zhang F (2013) Multiplex Genome Engineering using CRISPR/Cas Systems. Science 339:819–823. https://doi.org/10.1126/science.1231143
Gasiunas G, Barrangou R, Horvath P, Siksnys V (2012) Cas9–crRNA ribonucleoprotein complex mediates specific DNA cleavage for adaptive immunity in bacteria. PNAS 109:E2579–E86. https://doi.org/10.1073/pnas.1208507109
Gelvin SB (2017) Integration of Agrobacterium T-DNA into the plant genome. Annu Rev Genet 51:195–217. https://doi.org/10.1146/annurev-genet-120215-035320
Gronenborn B (2007) The tomato yellow leaf curl virus genome and function of its proteins. in, Tomato Yellow Leaf Curl Virus Disease: Management, Molecular Biology, Breeding for Resistance. https://doi.org/10.1007/978-1-4020-4769-5_5
Han J-S, Yooon M-K, jeong M (2008) Effects of a variety of treatments affecting chinese cabbage protoplast culture, and plant regeneration from protoplast-derived callus. J Plant Biotechnol 35:235–243. https://doi.org/10.5010/JPB.2008.35.3.235
Hanson PM, Bernacchi D, Green S, Tanksley SD, Muniyappa V, Padmaja AS, Chen H-m, Kuo G, Fang D, Chen J-t (2000) Mapping a wild Tomato Introgression Associated with Tomato Yellow Leaf Curl Virus Resistance in a cultivated Tomato Line. J Am Soc Hortic Sci 125:15–20. https://doi.org/10.21273/JASHS.125.1.15
Hossain M, Imanishi S, Egashira H (1995) An improvement of tomato protoplast culture for rapid plant regeneration. Plant Cell Tissue Organ Cult 42:141–146. https://doi.org/10.1007/BF00034230
Hutton SF, Scott JW, Schuster DJ (2012) Recessive resistance to Tomato yellow leaf curl virus from the Tomato Cultivar Tyking is located in the same region as Ty-5 on chromosome 4. HortScience 47:324–327. https://doi.org/10.21273/HORTSCI.47.3.324
Ji Y, Schuster DJ, Scott JW (2007) Ty-3, a begomovirus resistance locus near the Tomato yellow leaf curl virus resistance locus Ty-1 on chromosome 6 of tomato. Mol Breed 20:271–284. https://doi.org/10.1007/s11032-007-9089-7
Ji Y, Scott JW, Schuster DJ, Maxwell DP (2009) Molecular mapping of Ty-4, a New Tomato Yellow Leaf Curl Virus Resistance Locus on chromosome 3 of Tomato. J Am Soc Hortic Sci 134:281–288. https://doi.org/10.21273/JASHS.134.2.281
Jones HD (2015) Regulatory uncertainty over genome editing. Nat Plants 1:1–3. https://doi.org/10.1038/nplants.2014.11
Jung J, Kim HJ, Lee JM, Oh CS, Lee H-J, Yeam I (2015) Gene-based molecular marker system for multiple disease resistances in tomato against Tomato yellow leaf curl virus, late blight, and verticillium wilt. Euphytica 205:599–613. https://doi.org/10.1007/s10681-015-1442-z
Jupe F, Rivkin AC, Michael TP, Zander M, Motley ST, Sandoval JP, Slotkin RK, Chen H, Castanon R, Nery JR (2019) The complex architecture and epigenomic impact of plant T-DNA insertions. PLoS Genet 15:e1007819. https://doi.org/10.1371/journal.pgen.1007819
Kanchiswamy CN, Malnoy M, Velasco R, Kim J-S, Viola R (2015) Non-GMO genetically edited crop plants. Trends Biotechnol 33:489–491. https://doi.org/10.1016/j.tibtech.2015.04.002
Kassahun BM, Kang B-C, Su-Ji B, Nam YJ, Mundo GF, Kang G-H, Kim K, Han J-S (2021) Rapid delivery of Cas9 gene into the tomato cv.‘Heinz 1706’through an optimized Agrobacterium-mediated transformation procedure. Biocell 45:199. https://doi.org/10.32604/biocell.2021.012353
Kim D, Kang B-C, Kim J-S (2021) Identifying genome-wide off-target sites of CRISPR RNA–guided nucleases and deaminases with Digenome-seq. Nat Protoc 16:1170–1192. https://doi.org/10.1038/s41596-020-00453-6
Koiso N, Toda E, Ichikawa M, Kato N, Okamoto T (2017) Development of gene expression system in egg cells and zygotes isolated from rice and maize. Plant Direct 1:e00010. https://doi.org/10.1002/pld3.10
Lapidot M, Karniel U, Gelbart D, Fogel D, Evenor D, Kutsher Y, Makhbash Z, Nahon S, Shlomo H, Chen L (2015) A novel route controlling begomovirus resistance by the messenger RNA surveillance factor pelota. PLoS Genet 11:e1005538. https://doi.org/10.1371/journal.pgen.1005538
Latif M, Mumtaz N, Davey MR, Power JB (1993) Plant regeneration from protoplasts isolated from cell suspension cultures of the wild tomato, Lycopersicon chilense Dun. Plant Cell Tissue Organ Cult 32:311–317. https://doi.org/10.1007/BF00042294
Lee J, Oh C-S, Yeam I (2015) Molecular markers for selecting Diverse Disease Resistances in Tomato breeding programs. Plant Breed Biotechnol 3:308–322. https://doi.org/10.9787/PBB.2015.3.4.308
Lee JH, Chung DJ, Lee JM, Yeam I (2021) Development and application of gene-specific markers for Tomato Yellow Leaf Curl Virus Resistance in both field and Artificial Infections. Plants 10:9. https://doi.org/10.3390/plants10010009
Li X, Sandgrind S, Moss O, Guan R, Ivarson E, Wang ES, Kanagarajan S, Zhu L-H (2021) Efficient protoplast regeneration protocol and CRISPR/Cas9-Mediated editing of Glucosinolate Transporter (GTR) genes in rapeseed (Brassica napus L). Front Plant Sci 12. https://doi.org/10.3389/fpls.2021.680859
Liang Z, Chen K, Li T, Zhang Y, Wang Y, Zhao Q, Liu J, Zhang H, Liu C, Ran Y (2017) Efficient DNA-free genome editing of bread wheat using CRISPR/Cas9 ribonucleoprotein complexes. Nat Commun 8:1–5. https://doi.org/10.1038/ncomms14261
Liu C, Zhang L, Liu H, Cheng K (2017) Delivery strategies of the CRISPR-Cas9 gene-editing system for therapeutic applications. J Controlled Release 266:17–26. https://doi.org/10.1016/j.jconrel.2017.09.012
Liu Y, Andersson M, Granell A, Cardi T, Hofvander P, Nicolia A (2022) Establishment of a DNA-free genome editing and protoplast regeneration method in cultivated tomato (Solanum lycopersicum). https://doi.org/10.1007/s00299-022-02893-8. Plant Cell Rep
Malnoy M, Viola R, Jung M-H, Koo O-J, Kim S, Kim J-S, Velasco R, Nagamangala Kanchiswamy C (2016) DNA-free genetically edited grapevine and apple protoplast using CRISPR/Cas9 ribonucleoproteins. Front Plant Sci 7:1904. https://doi.org/10.3389/fpls.2016.01904
Manghwar H, Li B, Ding X, Hussain A, Lindsey K, Zhang X, Jin S (2020) CRISPR/Cas Systems in Genome Editing: methodologies and tools for sgRNA Design, off-target evaluation, and strategies to Mitigate Off-Target Effects. Adv Sci 7:1902312. https://doi.org/10.1002/advs.201902312
Menczel L, Wolfe K (1984) High frequency of fusion induced in freely suspended protoplast mixtures by polyethylene glycol and dimethylsulfoxide at high pH. Plant Cell Rep 3:196–198. https://doi.org/10.1007/BF00270199
Naim F, Shand K, Hayashi S, O’Brien M, McGree J, Johnson AA, Dugdale B, Waterhouse PM (2020) Are the current gRNA ranking prediction algorithms useful for genome editing in plants? PLoS ONE 15:e0227994. https://doi.org/10.1371/journal.pone.0227994
Nicolia A, Proux-Wéra E, Åhman I, Onkokesung N, Andersson M, Andreasson E, Zhu L-H (2015) Targeted gene mutation in tetraploid potato through transient TALEN expression in protoplasts. 204:17–24. https://doi.org/10.1016/j.jbiotec.2015.03.021
Nicolia A, Andersson M, Hofvander P, Festa G, Cardi T (2021a) Tomato protoplasts as cell target for ribonucleoprotein (RNP)-mediated multiplexed genome editing. Plant Cell Tissue Organ Cult 144:463–467. https://doi.org/10.1007/s11240-020-01954-8
Nicolia A, Fält A-S, Hofvander P, Andersson M (2021b) Protoplast-Based Method for Genome Editing in Tetraploid Potato. in Pasquale Tripodi (ed.), Crop Breeding: Genetic Improvement Methods (Springer US: New York, NY). https://doi.org/10.1007/978-1-0716-1201-9_12
Niedz RP, Rutter SM, Handley LW, Sink KC (1985) Plant regeneration from leaf protoplasts of six tomato cultivars. Plant Sci 39:199–204. https://doi.org/10.1016/0168-9452(85)90175-X
Park J, Bae S, Kim J-S (2015) Cas-Designer: a web-based tool for choice of CRISPR-Cas9 target sites. Bioinform 31:4014–4016. https://doi.org/10.1093/bioinformatics/btv537
Park J, Lim K, Kim J-S, Bae S (2016) Cas-analyzer: an online tool for assessing genome editing results using NGS data. Bioinform 33:286–288. https://doi.org/10.1093/bioinformatics/btw561
Peres LEP, Morgante PG, Vecchi C, Kraus JE, van Sluys M-A (2001) Shoot regeneration capacity from roots and transgenic hairy roots of tomato cultivars and wild related species. Plant Cell Tissue Organ Cult 65:37–44. https://doi.org/10.1023/A:1010631731559
Pramanik D, Shelake RM, Park J, Kim MJ, Hwang I, Park Y, Kim J-Y (2021) CRISPR/Cas9-mediated generation of pathogen-resistant tomato against tomato yellow leaf curl virus and powdery mildew. Int J Mol Sci 22:1878. https://doi.org/10.3390/ijms22041878
Prasad A, Sharma N, Hari-Gowthem G, Muthamilarasan M, Prasad M (2020) Tomato Yellow Leaf Curl Virus: Impact, Challenges, and management. Trends Plant Sci 25:897–911. https://doi.org/10.1016/j.tplants.2020.03.015
Ramos RS, Kumar L, Shabani F, Picanço MC (2019) Risk of spread of tomato yellow leaf curl virus (TYLCV) in tomato crops under various climate change scenarios. Agric Syst 173:524–535. https://doi.org/10.1016/j.agsy.2019.03.020
Ran FA, Hsu PD, Wright J, Agarwala V, Scott DA, Zhang F (2013) Genome engineering using the CRISPR-Cas9 system. Nat Protocols 8:2281–2308. https://doi.org/10.1038/nprot.2013.143
Reed KM, BOR B (2021) Protoplast Regeneration and its use in new plant breeding Technologies. Front Genome Ed 3:734951–734951. https://doi.org/10.3389/fgeed.2021.734951
Ren Y, Tao X, Li D, Yang X, Zhou X (2022) ty-5 confers broad-spectrum resistance to Geminiviruses. Viruses 14:1804. https://doi.org/10.3390/v14081804
Schuster DJ, Mann RS, Toapanta M, Cordero R, Thompson S, Cyman S, Shurtleff A, Morris IIRF (2010) Monitoring neonicotinoid resistance in biotype B of Bemisia tabaci in Florida. Pest Manage Sci 66:186–195. https://doi.org/10.1002/ps.1853
Shahin E (1985) Totipotency of tomato protoplasts. Theor Appl Genet 69:235–240. https://doi.org/10.1007/BF00662431
Song H, Mugnier P, Das AK, Webb HM, Evans DR, Tuite MF, Hemmings BA, Barford D (2000) The Crystal structure of human eukaryotic release factor eRF1—Mechanism of Stop Codon Recognition and Peptidyl-tRNA hydrolysis. Cell 100:311–321. https://doi.org/10.1016/S0092-8674(00)80667-4
Svitashev S, Schwartz C, Lenderts B, Young JK, Mark Cigan A (2016) Genome editing in maize directed by CRISPR–Cas9 ribonucleoprotein complexes. Nat Commun 7:13274. https://doi.org/10.1038/ncomms13274
Tan M-L, Rietveld EM, Van Marrewijk GA, Kool AJ (1987) Regeneration of leaf mesophyll protoplasts of tomato cultivars (L. esculentum): factors important for efficient protoplast culture and plant regeneration. Plant Cell Rep 6:172–175. https://doi.org/10.1007/BF00268470
Thyme SB, Akhmetova L, Montague TG, Valen E, Schier AF (2016) Internal guide RNA interactions interfere with Cas9-mediated cleavage. Nat Commun 7:11750. https://doi.org/10.1038/ncomms11750
Toda E, Koiso N, Takebayashi A, Ichikawa M, Kiba T, Osakabe K, Osakabe Y, Sakakibara H, Kato N, Okamoto T (2019) An efficient DNA- and selectable-marker-free genome-editing system using zygotes in rice. Nat Plants 5:363–368. https://doi.org/10.1038/s41477-019-0386-z
Tomato Genome Consortium x (2012) The tomato genome sequence provides insights into fleshy fruit evolution. Nature 485:635. https://doi.org/10.1038/nature11119
Woo JW, Kim J, Kwon SI, Corvalán C, Cho SW, Kim H, Kim S-G, Kim S-T, Choe S, Kim J-S (2015) DNA-free genome editing in plants with preassembled CRISPR-Cas9 ribonucleoproteins. Nat Biotechnol 33:1162–1164. https://doi.org/10.1038/nbt.3389
Xu J, Kang B-C, Naing AH, Bae S-J, Kim J-S, Kim H, Kim CK (2020) CRISPR/Cas9-mediated editing of 1-aminocyclopropane-1-carboxylate oxidase1 enhances Petunia flower longevity. Plant Biotechnol J 18:287–297. https://doi.org/10.1111/pbi.13197
Yoo S-D, Cho Y-H, Sheen J (2007) Arabidopsis mesophyll protoplasts: a versatile cell system for transient gene expression analysis. Nat Protoc 2:1565–1572. https://doi.org/10.1038/nprot.2007.199
Yoon Y-J, Venkatesh J, Lee J-H, Kim J, Lee H-E, Kim D-S, Kang B-C (2020) Genome editing of eIF4E1 in Tomato confers resistance to Pepper Mottle Virus. Front Plant Sci 11. https://doi.org/10.3389/fpls.2020.01098
Zamir D, Ekstein-Michelson I, Zakay Y, Navot N, Zeidan M, Sarfatti M, Eshed Y, Harel E, Pleban T, van-Oss H, Kedar N, Rabinowitch HD, Czosnek H (1994) Mapping and introgression of a tomato yellow leaf curl virus tolerance gene, TY-1. Theor Appl Genet 88:141–146. https://doi.org/10.1007/BF00225889
Zhang X-H, Tee LY, Wang X-G, Huang Q-S, Yang S-H (2015) Off-target Effects in CRISPR/Cas9-mediated Genome Engineering. Mol Ther Nucleic Acids 4:e264. https://doi.org/10.1038/mtna.2015.37
Zhang Y, Liang Z, Zong Y, Wang Y, Liu J, Chen K, Qiu J-L, Gao C (2016) Efficient and transgene-free genome editing in wheat through transient expression of CRISPR/Cas9 DNA or RNA. Nat Commun 7:1–8. https://doi.org/10.1038/ncomms12617
Zhang S, Shen J, Li D, Cheng Y (2021) Strategies in the delivery of Cas9 ribonucleoprotein for CRISPR/Cas9 genome editing. Theranostics 11:614–648. https://doi.org/10.7150/thno.47007
Zheng MY, Konzak CF (1999) Effect of 2,4-dichlorophenoxyacetic acid on callus induction and plant regeneration in anther culture of wheat (Triticum aestivum L). Plant Cell Rep 19:69–73. https://doi.org/10.1007/s002990050712
Acknowledgements
This study was supported by the National Research Foundation of Korea (2021R1A2C2093789) of the Republic of Korea.
Author information
Authors and Affiliations
Contributions
GHK and BCK performed the experiments. JSH and JML designed this study. GHK and JML drafted the manuscript. GHK and JML revised and corrected the manuscript. All the authors have read and approved the final manuscript.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Communicated by Sung-Chur Sim.
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kang, G.H., Kang, BC., Han, JS. et al. DNA-free genome editing in tomato protoplasts using CRISPR/Cas9 ribonucleoprotein delivery. Hortic. Environ. Biotechnol. 65, 131–142 (2024). https://doi.org/10.1007/s13580-023-00549-4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13580-023-00549-4