Abstract
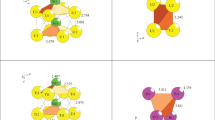
Using computer methods (the ToposPro software package), a combinatorial topological analysis and modeling of the self-assembly of U8Ni10Al36-mC54 (a = 15.5470 Å, b = 4.0610 Å, c = 16.4580 Å, β = 120.00°, V = 899.89 Å3, C m), U20Ni26-mC46 (a = 7.660 Å, b = 13.080 Å, c = 7.649 Å, β = 108.88°, V = 725.26 Å3, C2/m), and U8Co8-cI16 (a = 6.343 Å, V = 255.20 Å3, I 213) are carried out. For the U8Ni10Al36-mC54 crystal structure, 960 variants of the cluster representation of the 3D atomic grid with the number of structural units 5, 6, and 7 are established. Six crystallographically independent structural units in the form of a pyramid K5 = 0@Al(U2Al2), pyramid K6A = 0@U(NiAl4), and pyramid K6B = 0@U(NiAl4), as well as rings K3A = 0@NiAl2, K3B = 0@NiAl2, and K3C = 0@Al3, are determined. For the U20Ni26-mC46 crystal structure, the structural units K5 = Ni(Ni2U2) and icosahedra K13= Ni@Ni6U6 are defined. For the crystal structure U2Co2-cI16, the structural units—tetrahedra K4 = U2Co2—are defined. The symmetry and topological code of the processes of self-assembly of 3D structures from clusters-precursors are reconstructed in the following form: primary chain → layer → framework.









Similar content being viewed by others
REFERENCES
Inorganic Crystal Structure Database (ICSD), Karlsruhe: Fachinformationszentrum, USA: Natl. Inst. Stand. Technol.
Villars, P. and Cenzual, K., Pearson’s Crystal Data-Crystal Structure Database for Inorganic Compounds (PCDIC), Materials Park, OH: ASM Int.
Perricone, A. and Noel, H., Crystal structure refinements and magnetic behavior of U6Ni, UNi5, UNi2 and the substitution derivative UNi1.7Si0.3, Chem. Met. Alloys, 2008, vol. 1, pp. 54–57.
Perricone, A. and Noel, H., Crystal structure and magnetic properties of the binary uranium–nickel alloy UNi4, Intermetallics, 2002, vol. 10, pp. 519–521.
Perricone, A. and Noel, H., Characterization of the new uranium–nickel alloy and U10Ni13, J. Nucl. Mater., 2001, vol. 299, pp. 260–263.
Perricone, A., Potel, M., and Noel, H., Crystal structure and magnetic properties of the binary uranium–nickel alloy U11 and Ni16, J. Alloys Compd., 2002, vol. 340, pp. 39–42.
Grin, Y.N., Rogl, P., Akselrud, L.G., and Pertlik, F., X-ray studies in the systems ZrNi5–xOx and UNi5–xAlx, Z. Kristallogr., 1989, vol. 188, pp. 271–277.
Dwight, A.E., The unit-cell constants of some PuNi3-type compounds, Acta Crystallogr., Sect. B, 1968, vol. 24, pp. 1395–1396.
Dommann, A., Brandle, H., Hulliger, F., and Fischer, P., Crystal structure and magnetic order of and UCo5, J. Less-Common Met., 1990, vol. 158, pp. 287–294.
Baenziger, N.C., Rundle, R.E., Snow, A.I., and Wilson, A.S., Compounds of uranium with the transition metals of the first long period, Acta Crystallogr., 1950, vol. 3, pp. 34–40.
Kimball, C.W., Vaishnava, P.P., and Dwight, A.E., Phonon anomalies and local atomic displacements in the exchange-enhanced superconductor U6Fe, Phys. Rev. B, 1985, vol. 32, pp. 4419–4425.
Lebech, B., Wulff, M., Lander, G.H., Rebizant, J., Spirlet, J.C., and Delapalme, A., Neutron diffraction studies of the crystalline and magnetic properties of UFe2, J. Phys.: Condens. Matter, 1989, vol. 1, pp. 10229–10248.
Lawson, A.C., Jr., Smith, J.L., Willis, J.O., O’Rourke, J.A., Faber, J., and Hitterman, R.L., Orthorhombic structure of UMn2 at low and temperatures, J. Less-Common Met., 1985, vol. 107, pp. 243–248.
Richter, C.G., Jeitschko, W., Kuennen, B., and Gerdes, M.H., The ternary titanium transition metal bismuthides Ti4TBi2 with T = Cr, Mn, Fe, Co and Ni, J. Solid State Chem., 1997, vol. 133, pp. 400–406.
Bauer, E.D., Sidorov, V.A., Bobev, S., Mixson, D.J., Thompson, J.D., Sarrao, J.L., and Hundley, M.F., High-pressure investigation of the heavy-fermion antiferromagnet U3Ni5Al19, Phys. Rev. B, 2005, vol. 71, p. 014419.
Shevchenko, V.Ya., Ilyushin, G.D., and Blatov, V.A., Cluster self-organization of intermetallic systems: New two-layer nanocluster-precursor K44 = 0@8(U2Ni6) @ 36(U12Ni24) in the crystal structure U66Ni96-hR162, Glass Phys. Chem., 2021, vol. 47, pp. 525–532.
Ilyushin, G.D., Theory of cluster self-organization of crystal-forming systems. Geometrical-topological modeling of nanocluster precursors with a hierarchical structure, Struct. Chem., 2012, vol. 20, no. 6, pp. 975–1043.
Shevchenko, V.Ya., Medrish, I.V., Ilyushin, G.D., and Blatov, V.A., From clusters to crystals: Scale chemistry of intermetallics, Struct. Chem., 2019, vol. 30, pp. 2015–2027.
Ilyushin, G.D., Intermetallic compounds KnMm (M = Ag, Au, As, Sb, Bi, Ge, Sn, Pb): Geometrical and topological analysis, cluster precursors, and self-assembly of crystal structures, Crystallogr. Rep., 2020, vol. 65, no. 7, pp. 1095–1105.
Ilyushin, G.D., Intermetallic compounds NakMn (M = K, Cs, Ba, Ag, Pt, Au, Zn, Bi, Sb): Geometrical and topological analysis, cluster precursors, and self-assembly of crystal structures, Crystallogr. Rep., 2020, vol. 65, no. 4, pp. 539–545.
Blatov, V.A., Shevchenko, A.P., and Proserpio, D.M., Applied topological analysis of crystal structures with the program package ToposPro, Cryst. Growth Des., 2014, vol. 14, no. 7, pp. 3576–3585.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Shevchenko, V.Y., Ilyushin, G.D. Cluster Self-Organization of Intermetallic Systems: K3, K4, K5, K6, and K13 Clusters-Precursors for the Self-Assembly of U8Ni10Al36-mC54, U20Ni26-mC46, and U8Co8-cI16 Crystal Structures. Glass Phys Chem 49, 327–335 (2023). https://doi.org/10.1134/S1087659623600321
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1087659623600321