Abstract
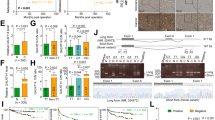
Radioresistance is the primary reason for radiotherapy failure in non-small cell lung cancer (NSCLC) patients. Glycosylation-related alterations are critically involved in tumor radioresistance. However, the relationship between glycosylation and NSCLC radioresistance is unclear. Here, we generated radioresistant NSCLC cell models by using fractionated irradiation. The aberrant glycosylation involved in NSCLC-related radioresistance was elucidated by transcriptomic, proteomic, and glycomic analyses. We conducted in vitro and in vivo investigations for determining the biological functions of glycosylation. Additionally, its downstream pathways and upstream regulators were inferred and verified. We demonstrated that mucin-type O-glycosylation and the O-glycosylating enzyme GALNT2 were highly expressed in radioresistant NSCLC cells. GALNT2 was found to be elevated in NSCLC tissues; this elevated level showed a remarkable association with response to radiotherapy treatment as well as overall survival. Functional experiments showed that GALNT2 knockdown improved NSCLC radiosensitivity via inducing apoptosis. By using a lectin pull-down system, we revealed that mucin-type O-glycans on IGF1R were modified by GALNT2 and that IGF1R could affect the expression of apoptosis-related genes. Moreover, GALNT2 knockdown-mediated in vitro radiosensitization was enhanced by IGF1R inhibition. According to a miRNA array analysis and a luciferase reporter assay, miR-30a-5p negatively modulated GALNT2. In summary, our findings established GALNT2 as a key contributor to the radioresistance of NSCLC. Therefore, targeting GALNT2 may be a promising therapeutic strategy for NSCLC.
Graphical Abstract









Similar content being viewed by others
Data Availability
All data generated or analyzed in this study are present in this published article and its supplementary information files.
Abbreviations
- GALNT2:
-
N-acetylgalactosaminyltransferase 2
- qRT-PCR:
-
real-time PCR
- VVA:
-
Vicia villosa lectin
- IHC:
-
immunohistochemistry
- NSCLC:
-
non-small cell lung cancer
- 3′-UTR:
-
3′-untranslated region
References
Alfaro-Arnedo E, López IP, Piñeiro-Hermida S, Canalejo M, Gotera C, Sola JJ, Roncero A, Peces-Barba G, Ruíz-Martínez C, Pichel JG. IGF1R acts as a cancer-promoting factor in the tumor microenvironment facilitating lung metastasis implantation and progression. Oncogene. 2022;10:1–15.
Beaman EM, Carter DRF, Brooks SA. GALNTs: master regulators of metastasis-associated epithelial-mesenchymal transition (EMT)? Glycobiology. 2022;32:556–79.
Cai H, Zou J, Wang W, Yang A. BMP2 induces hMSC osteogenesis and matrix remodeling. Mol Med Rep. 2021;23:125.
Cao L, Zhou Y, Li X, Lin S, Tan Z, Guan F. Integrating transcriptomics, proteomics, glycomics and glycoproteomics to characterize paclitaxel resistance in breast cancer cells. J Proteomics. 2021;243:104266.
Chen C, Tang J, Xu S, Zhang W, Jiang H. miR-30a-5p inhibits proliferation and migration of lung squamous cell carcinoma cells by targeting FOXD1. Biomed Res Int. 2020;2020:2547902.
De Bousser E, Meuris L, Callewaert N, Festjens N. Human T cell glycosylation and implications on immune therapy for cancer. Hum Vaccin Immunother. 2020;16:2374–88.
Dong X, Chen C, Deng X, Liu Y, Duan Q, Peng Z, Luo Z, Shen L. A novel mechanism for C1GALT1 in the regulation of gastric cancer progression. Cell Biosci. 2021;11:166.
Dong X, Luo Z, Wang Y, Meng L, Duan Q, Qiu L, Peng F, Shen L. Altered O-glycosylation is associated with inherent radioresistance and malignancy of human laryngeal carcinoma. Exp Cell Res. 2018;362:302–10.
Gabay G, Faigenboim A, Dahan Y, Izhaki Y, Itkin M, Malitsky S, Elkind Y, Flaishman MA. Transcriptome analysis and metabolic profiling reveal the key role of α-linolenic acid in dormancy regulation of European pear. J Exp Bot. 2019;70:1017–31.
Guan L, Fan P, Liu X, Zhou M, Wu Y, Liu R, Liu Y, Bai H. Maternal GALNT2 variations affect blood pressure, atherogenic index, and fetal growth, depending on bmi in gestational diabetes mellitus. Front Endocrinol (Lausanne). 2021;12:690229.
Guarino MP, Altomare A, Barera S, Locato V, Cocca S, Franchin C, Arrigoni G, Vannini C, Grossi S, Campomenosi P, Pasqualetti V, Bracale M, Alloni R, De Gara L, Cicala M. Effect of Inulin on proteome changes induced by pathogenic lipopolysaccharide in human colon. PLoS One. 2017;12:e0169481.
Guo Y, Sun W, Gong T, Chai Y, Wang J, Hui B, Li Y, Song L, Gao Y. miR-30a radiosensitizes non-small cell lung cancer by targeting ATF1 that is involved in the phosphorylation of ATM. Oncol Rep. 2017;37:1980–8.
Ho WL, Chou CH, Jeng YM, Lu MY, Yang YL, Jou ST, Lin DT, Chang HH, Lin KH, Hsu WM, Huang MC. GALNT2 suppresses malignant phenotypes through IGF-1 receptor and predicts favorable prognosis in neuroblastoma. Oncotarget. 2014;5:12247–59.
Huang Y, Yang X, Lu Y, Zhao Y, Meng R, Zhang S, Dong X, Xu S, Wu G. UBE2O targets Mxi1 for ubiquitination and degradation to promote lung cancer progression and radioresistance. Cell Death Differ. 2021;28:671–84.
Jie X, Fong WP, Zhou R, Zhao Y, Meng R, Zhang S, Dong X, Zhang T, Yang K, Wu G, Xu S. USP9X-mediated KDM4C deubiquitination promotes lung cancer radioresistance by epigenetically inducing TGF-β2 transcription. Cell Death Differ. 2021;28:2095–111.
Joshi HJ, Hansen L, Narimatsu Y, Freeze HH, Henrissat B, Bennett E, Wandall HH, Clausen H, Schjoldager KT. Glycosyltransferase genes that cause monogenic congenital disorders of glycosylation are distinct from glycosyltransferase genes associated with complex diseases. Glycobiology. 2018;28:284–94.
Kato K, Hansen L, Clausen H. Polypeptide N-acetylgalactosaminyltransferase-associated Phenotypes in Mammals. Molecules. 2021;26:5504.
Li X, Zhang Y, Zhang M, Wang Y. GALNT2 regulates ANGPTL3 cleavage in cells and in vivo of mice. Sci Rep. 2020;10:16168.
Marucci A, Antonucci A, De Bonis C, Mangiacotti D, Scarale MG, Trischitta V, Di Paola R. GALNT2 as a novel modulator of adipogenesis and adipocyte insulin signaling. Int J Obes (Lond). 2019;43:2448–57.
Mendelevich A, Vinogradova S, Gupta S, Mironov AA, Sunyaev SR, Gimelbrant AA. Replicate sequencing libraries are important for quantification of allelic imbalance. Nat Commun. 2021;12:3370.
Mereiter S, Balmaña M, Campos D, Gomes J, Reis CA. Glycosylation in the era of cancer-targeted therapy: where are we heading? Cancer Cell. 2019;36:6–16.
Pang J, Li P, He H, Xu S, Liu Z. Molecularly imprinted polymers outperform lectin counterparts and enable more precise cancer diagnosis. Chem Sci. 2022;13:4589–97.
Phan T, Nguyen VH, A'Lincourt Salazar M, Wong P, Diamond DJ, Yim JH, Melstrom LG. Inhibition of autophagy amplifies baicalein-induced apoptosis in human colorectal cancer. Mol Ther Oncolytics. 2020;19:1–7.
Rodrigues JG, Duarte HO, Reis CA, Gomes J. Aberrant protein glycosylation in cancer: implications in targeted therapy. Biochem Soc Trans. 2021;49:843–54.
Shen L, Xia M, Deng X, Ke Q, Zhang C, Peng F, Dong X, Luo Z. A lectin-based glycomic approach identifies FUT8 as a driver of radioresistance in oesophageal squamous cell carcinoma. Cell Oncol (Dordr). 2020;43:695–707.
Silsirivanit A. Glycosylation markers in cancer. Adv Clin Chem. 2019;89:189–213.
Simpson AD, Soo YWJ, Rieunier G, Aleksic T, Ansorge O, Jones C, Macaulay VM. Type 1 IGF receptor associates with adverse outcome and cellular radioresistance in paediatric high-grade glioma. Br J Cancer. 2020;122:624–9.
Sun X, He Z, Guo L, Wang C, Lin C, Ye L, Wang X, Li Y, Yang M, Liu S, Hua X, Wen W, Long Z, Zhang W, Li H, Jian Y, Zhu Z, Wu X, Lin H. ALG3 contributes to stemness and radioresistance through regulating glycosylation of TGF-β receptor II in breast cancer. J Exp Clin Cancer Res. 2021;40:149.
Sun Z, Xue H, Wei Y, Wang C, Yu R, Wang S, Xu J, Qian M, Meng Q, Li G. Mucin O-glycosylating enzyme GALNT2 facilitates the malignant character of glioma by activating the EGFR/PI3K/Akt/mTOR axis. Clin Sci (Lond). 2019;133:1167–84.
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global Cancer Statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 Countries. CA Cancer J Clin. 2021;71:209–49.
Wang W, Sun R, Zeng L, Chen Y, Zhang N, Cao S, Deng S, Meng X, Yang S. GALNT2 promotes cell proliferation, migration, and invasion by activating the Notch/Hes1-PTEN-PI3K/Akt signaling pathway in lung adenocarcinoma. Life Sci. 2021;276:119439.
Werner H, Meisel-Sharon S, Bruchim I. Oncogenic fusion proteins adopt the insulin-like growth factor signaling pathway. Mol Cancer. 2018;17:28.
Wu Q, Tian AL, Kroemer G, Kepp O. Autophagy induction by IGF1R inhibition with picropodophyllin and linsitinib. Autophagy. 2021;17:2046–7.
Yang J, Rao S, Cao R, Xiao S, Cui X, Ye L. miR-30a-5p suppresses lung squamous cell carcinoma via ATG5 - mediated autophagy. Aging (Albany NY). 2021;13:17462–72.
Yu Y, Wang Z, Zheng Q, Li J. GALNT2/14 overexpression correlate with prognosis and methylation: potential therapeutic targets for lung adenocarcinoma. Gene. 2021;790:145689.
Zhang H, Deng W, Lu C, He M, Yan H. SMRT sequencing of full-length transcriptome and gene expression analysis in two chemical types of Pogostemon cablin (Blanco) Benth. PeerJ. 2022;10:e12940.
Zhou X, Zeng B, Li Y, Wang H, Zhang X. LINC02532 contributes to radiosensitivity in clear cell renal cell carcinoma through the miR-654-5p/YY1 Axis. Molecules. 2021;26:7040.
Funding
The present work was aided by the Natural Science Foundation of Hubei Province (2022CFB491), the Advantages Discipline Group (Medicine) Project in Higher Education of Hubei Province (2021-2025) (2022XKQT2), and the National Undergraduate Training Program for Innovation and Entrepreneurship (S202210929015).
Author information
Authors and Affiliations
Contributions
Study concept and design: all authors. Material preparation and data collection and analysis: Xiaoxia Dong, Yahui Leng, Tian Tian, Qing Hu, Shuang Chen, and Yufeng Liu. First draft of the manuscript: Li Shen. All authors have commented on previous manuscript versions and have read and approved the final version.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The Ethics Committee of the Hubei University of Medicine (HBMU) approved the present research. Following the Declaration of Helsinki, all the included subjects provided their written informed consent. The Animal Ethics Committee of the HBMU approved all animal experiments.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Online Resource 1:
Table S1. Sequence information of mimics and shRNAs (DOC 28 kb)
Online Resource 2:
Table S2. Sequences of primer pairs used for qRT-PCR (DOC 29 kb)
Online Resource 3:
Table S3. DEGs identified by RNA-seq (XLS 3293 kb)
Online Resource 4:
Table S4. Differentially expressed proteins identified by iTRAQ (XLSX 114 kb)
Online Resource 5:
Table S5. Summary of mean fluorescence intensities (XLS 42 kb)
Online Resource 6:
Table S6. Differentially expressed miRNAs identified by miRNA array (XLSX 13 kb)
ESM 7
(DOC 24 kb)
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Dong, X., Leng, Y., Tian, T. et al. GALNT2, an O-glycosylating enzyme, is a critical regulator of radioresistance of non-small cell lung cancer: evidence from an integrated multi-omics analysis. Cell Biol Toxicol 39, 3159–3174 (2023). https://doi.org/10.1007/s10565-023-09825-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-023-09825-6