Abstract
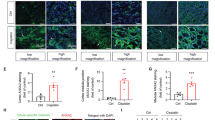
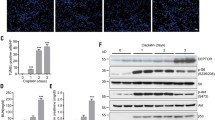
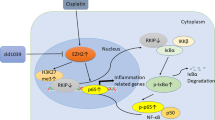
Wnt signaling is a principal pathway regulating the essential activities of cell proliferation. Here, we investigated the effect of Wnt/β-catenin signaling on in vivo drug-induced renal injury through the deletion of Dact2, a Wnt antagonist, and deciphered the underlying mechanism. Wild-type (WT) and Dact2 knockout (KO) mice were administered a single intraperitoneal injection of cisplatin to induce renal injury. The injury was alleviated in Dact2 KO mice, which showed lower levels of blood urea nitrogen and creatinine. RNA sequencing revealed 194 differentially expressed genes (DEGs) between WT and Dact2 KO mouse kidney before cisplatin treatment. Among them, higher levels of Igf1, one of the Wnt target genes responsible for “Positive regulation of cell proliferation” in KO mice, were confirmed along with the induction of Ki67 expression. In RNA-seq analysis comparing WT and Dact2 KO mice after cisplatin treatment, genes related to “Apoptosis” and “Activation of mitogen-activated protein kinase (MAPK) activity” were among the downregulated DEGs in KO mice. These results were corroborated in western blotting of proteins related to apoptosis and proapoptotic MAPK pathway; the expression of which was found to be lower in cisplatin-treated KO mice. Importantly, β-catenin was found to directly bind to and regulate the transcription of Igf1, leading to the alleviation of cisplatin-induced cytotoxicity by the Wnt agonist, CHIR-99021. In addition, Igf1 knockdown accelerated cisplatin-induced cytotoxicity, accompanied by the MAPK upregulation. Our findings suggest that Dact2 knockout could protect cisplatin-induced nephrotoxicity by inhibiting apoptosis, possibly through the regulation of the Igf1-MAPK axis associated with Wnt/β-catenin signaling.









Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request. All generated RNA-seq were deposited into the National Center for Biotechnology Information (NCBI) Sequence Read Archive (SRA) under project accession PRJNA984810.
Code availability
Not applicable.
Abbreviations
- AKI:
-
Acute kidney injury
- BUN:
-
Blood urea nitrogen
- ChIP:
-
Chromatin immunoprecipitation
- Dact2 :
-
Dishevelled-associated antagonist of beta-catenin 2
- DAVID:
-
Database for Annotation, Visualization and Integrated Discovery
- DEGs:
-
Differentially expressed genes
- FDR:
-
False discovery rate
- GO:
-
Gene Ontology
- IGF1:
-
Insulin-like growth factor 1
- KEGG:
-
Kyoto Encyclopedia of Genes and Genomes
- MAPK:
-
Mitogen-activated protein kinase
- siRNA:
-
Small interfering RNA
- TUNEL:
-
Terminal deoxynucleotidyl transferase dUTP nick end labeling
References
Bennett M, Macdonald K, Chan SW, Luzio JP, Simari R, Weissberg P. Cell surface trafficking of Fas: a rapid mechanism of p53-mediated apoptosis. Science. 1998;282(5387):290–3. https://doi.org/10.1126/science.282.5387.290.
Berger K, Moeller MJ. Mechanisms of epithelial repair and regeneration after acute kidney injury. Semin Nephrol. 2014;34(4):394–403. https://doi.org/10.1016/j.semnephrol.2014.06.006.
Brown A, Kumar S, Tchounwou PB. Cisplatin-based chemotherapy of human cancers. J Cancer Sci Ther. 2019;11(4)
Coelho S, Cabral G, Lopes JA, Jacinto A. Renal regeneration after acute kidney injury. Nephrology (Carlton). 2018;23(9):805–14. https://doi.org/10.1111/nep.13256.
Cosin-Roger J, Ortiz-Masia D, Calatayud S, Hernandez C, Esplugues JV, Barrachina MD. The activation of Wnt signaling by a STAT6-dependent macrophage phenotype promotes mucosal repair in murine IBD. Mucosal Immunol. 2016;9(4):986–98. https://doi.org/10.1038/mi.2015.123.
Culine S, Harter V, Gravis G, Flechon A, Chevreau C, Mahammedi H, et al. Chemotherapy for muscle-invasive bladder cancer: impact of cisplatin delivery on renal function and local control rate in the randomized phase III VESPER (GETUG-AFU V05) trial. Clin Genitourin Cancer. 2021;19(6):554–62. https://doi.org/10.1016/j.clgc.2021.08.005.
Cummings BS, Schnellmann RG. Cisplatin-induced renal cell apoptosis: caspase 3-dependent and -independent pathways. J Pharmacol Exp Ther. 2002;302(1):8–17. https://doi.org/10.1124/jpet.302.1.8.
Dai C, Stolz DB, Kiss LP, Monga SP, Holzman LB, Liu Y. Wnt/beta-catenin signaling promotes podocyte dysfunction and albuminuria. J Am Soc Nephrol. 2009;20(9):1997–2008. https://doi.org/10.1681/ASN.2009010019.
Ding H, Kopple JD, Cohen A, Hirschberg R. Recombinant human insulin-like growth factor-I accelerates recovery and reduces catabolism in rats with ischemic acute renal failure. J Clin Invest. 1993;91(5):2281–7. https://doi.org/10.1172/jci116456.
Fisher DA, Kivimäe S, Hoshino J, Suriben R, Martin PM, Baxter N, et al. Three Dact gene family members are expressed during embryonic development and in the adult brains of mice. Dev Dyn. 2006;235(9):2620–30. https://doi.org/10.1002/dvdy.20917.
Friedlaender M, Popovtzer MM, Weiss O, Nefesh I, Kopolovic J, Raz I. Insulin-like growth factor-1 (IGF-1) enhances recovery from HgCl2-induced acute renal failure: the effects on renal IGF-1, IGF-1 receptor, and IGF-binding protein-1 mRNA. J Am Soc Nephrol. 1995;5(10):1782–91. https://doi.org/10.1681/asn.V5101782.
Gautier JC, Riefke B, Walter J, Kurth P, Mylecraine L, Guilpin V, et al. Evaluation of novel biomarkers of nephrotoxicity in two strains of rat treated with cisplatin. Toxicol Pathol. 2010;38(6):943–56. https://doi.org/10.1177/0192623310379139.
Goes N, Urmson J, Vincent D, Ramassar V, Halloran PF. Effect of recombinant human insulin-like growth factor-1 on the inflammatory response to acute renal injury. J Am Soc Nephrol. 1996;7(5):710–20. https://doi.org/10.1681/asn.V75710.
Hao J, Li TG, Qi X, Zhao DF, Zhao GQ. WNT/beta-catenin pathway up-regulates Stat3 and converges on LIF to prevent differentiation of mouse embryonic stem cells. Dev Biol. 2006;290(1):81–91. https://doi.org/10.1016/j.ydbio.2005.11.011.
Hsu WL, Ma YL, Liu YC, Tai DJC, Lee EHY. Restoring Wnt6 signaling ameliorates behavioral deficits in MeCP2 T158A mouse model of Rett syndrome. Sci Rep. 2020;10(1):1074. https://doi.org/10.1038/s41598-020-57745-w.
Imberti B, Morigi M, Tomasoni S, Rota C, Corna D, Longaretti L, et al. Insulin-like growth factor-1 sustains stem cell mediated renal repair. J Am Soc Nephrol. 2007;18(11):2921–8. https://doi.org/10.1681/ASN.2006121318.
Kaldis P, Pagano M. Wnt signaling in mitosis. Dev Cell. 2009;17(6):749–50. https://doi.org/10.1016/j.devcel.2009.12.001.
Kawakami T, Ren S, Duffield JS. Wnt signalling in kidney diseases: dual roles in renal injury and repair. J Pathol. 2013;229(2):221–31. https://doi.org/10.1002/path.4121.
Kellum JA, Romagnani P, Ashuntantang G, Ronco C, Zarbock A, Anders HJ. Acute kidney injury. Nat Rev Dis Primers. 2021;7(1):52. https://doi.org/10.1038/s41572-021-00284-z.
Kuncewitch M, Yang WL, Corbo L, Khader A, Nicastro J, Coppa GF, et al. WNT agonist decreases tissue damage and improves renal function after ischemia-reperfusion. Shock. 2015;43(3):268–75. https://doi.org/10.1097/SHK.0000000000000293.
Latcha S, Jaimes EA, Patil S, Glezerman IG, Mehta S, Flombaum CD. Long-term renal outcomes after cisplatin treatment. Clin J Am Soc Nephrol. 2016;11(7):1173–9. https://doi.org/10.2215/CJN.08070715.
Lee WC, Hough MT, Liu W, Ekiert R, Lindström NO, Hohenstein P, et al. Dact2 is expressed in the developing ureteric bud/collecting duct system of the kidney and controls morphogenetic behavior of collecting duct cells. Am J Physiol Renal Physiol. 2010;299(4):F740–51. https://doi.org/10.1152/ajprenal.00148.2010.
Lin Q, Wang Y, Chen D, Sheng X, Liu J, Xiong H. Cisplatin regulates cell autophagy in endometrial cancer cells via the PI3K/AKT/mTOR signalling pathway. Oncol Lett. 2017;13(5):3567–71. https://doi.org/10.3892/ol.2017.5894.
Lin SL, Li B, Rao S, Yeo EJ, Hudson TE, Nowlin BT, et al. Macrophage Wnt7b is critical for kidney repair and regeneration. Proc Natl Acad Sci U S A. 2010;107(9):4194–9. https://doi.org/10.1073/pnas.0912228107.
Lin Z, Sun L, Xie S, Zhang S, Fan S, Li Q, et al. Chemotherapy-induced long non-coding RNA 1 promotes metastasis and chemo-resistance of TSCC via the Wnt/beta-catenin signaling pathway. Mol Ther. 2018;26(6):1494–508. https://doi.org/10.1016/j.ymthe.2018.04.002.
Longo KA, Kennell JA, Ochocinska MJ, Ross SE, Wright WS, MacDougald OA. Wnt signaling protects 3T3-L1 preadipocytes from apoptosis through induction of insulin-like growth factors. J Biol Chem. 2002;277(41):38239–44. https://doi.org/10.1074/jbc.M206402200.
Los M, Mozoluk M, Ferrari D, Stepczynska A, Stroh C, Renz A, et al. Activation and caspase-mediated inhibition of PARP: a molecular switch between fibroblast necrosis and apoptosis in death receptor signaling. Mol Biol Cell. 2002;13(3):978–88. https://doi.org/10.1091/mbc.01-05-0272.
Lu HF, Lai KC, Hsu SC, Lin HJ, Yang MD, Chen YL, et al. Curcumin induces apoptosis through FAS and FADD, in caspase-3-dependent and -independent pathways in the N18 mouse-rat hybrid retina ganglion cells. Oncol Rep. 2009;22(1):97–104. https://doi.org/10.3892/or_00000411.
Mansouri A, Ridgway LD, Korapati AL, Zhang Q, Tian L, Wang Y, et al. Sustained activation of JNK/p38 MAPK pathways in response to cisplatin leads to Fas ligand induction and cell death in ovarian carcinoma cells. J Biol Chem. 2003;278(21):19245–56. https://doi.org/10.1074/jbc.M208134200.
Muller M, Wilder S, Bannasch D, Israeli D, Lehlbach K, Li-Weber M, et al. p53 activates the CD95 (APO-1/Fas) gene in response to DNA damage by anticancer drugs. J Exp Med. 1998;188(11):2033–45. https://doi.org/10.1084/jem.188.11.2033.
Naughton CA. Drug-induced nephrotoxicity. Am Fam Physician. 2008;78(6):743–50.
Nusse R. Wnts and Hedgehogs: lipid-modified proteins and similarities in signaling mechanisms at the cell surface. Development. 2003;130(22):5297–305. https://doi.org/10.1242/dev.00821.
Pei Y, Brun SN, Markant SL, Lento W, Gibson P, Taketo MM, et al. WNT signaling increases proliferation and impairs differentiation of stem cells in the developing cerebellum. Development. 2012;139(10):1724–33. https://doi.org/10.1242/dev.050104.
Quesada A, Lee BY, Micevych PE. PI3 kinase/Akt activation mediates estrogen and IGF-1 nigral DA neuronal neuroprotection against a unilateral rat model of Parkinson’s disease. Dev Neurobiol. 2008;68(5):632–44. https://doi.org/10.1002/dneu.20609.
Rolland AL, Garnier AS, Meunier K, Drablier G, Briet M. Drug-induced acute kidney injury: a study from the French Medical Administrative and the French National Pharmacovigilance Databases using capture-recapture method. J Clin Med. 2021;10(2) https://doi.org/10.3390/jcm10020168.
Sebastian A, Hum NR, Murugesh DK, Hatsell S, Economides AN, Loots GG. Wnt co-receptors Lrp5 and Lrp6 differentially mediate Wnt3a signaling in osteoblasts. PLoS One. 2017;12(11):e0188264. https://doi.org/10.1371/journal.pone.0188264.
Shen DW, Pouliot LM, Hall MD, Gottesman MM. Cisplatin resistance: a cellular self-defense mechanism resulting from multiple epigenetic and genetic changes. Pharmacol Rev. 2012;64(3):706–21. https://doi.org/10.1124/pr.111.005637.
Subramaniam S, Shahani N, Strelau J, Laliberté C, Brandt R, Kaplan D, et al. Insulin-like growth factor 1 inhibits extracellular signal-regulated kinase to promote neuronal survival via the phosphatidylinositol 3-kinase/protein kinase A/c-Raf pathway. J Neurosci. 2005;25(11):2838–52. https://doi.org/10.1523/jneurosci.5060-04.2005.
Tanigawa S, Wang H, Yang Y, Sharma N, Tarasova N, Ajima R, et al. Wnt4 induces nephronic tubules in metanephric mesenchyme by a non-canonical mechanism. Dev Biol. 2011;352(1):58–69. https://doi.org/10.1016/j.ydbio.2011.01.012.
Terada Y, Tanaka H, Okado T, Shimamura H, Inoshita S, Kuwahara M, et al. Expression and function of the developmental gene Wnt-4 during experimental acute renal failure in rats. J Am Soc Nephrol. 2003;14(5):1223–33. https://doi.org/10.1097/01.asn.0000060577.94532.06.
Toh TB, Lim JJ, Hooi L, Rashid M, Chow EK. Targeting Jak/Stat pathway as a therapeutic strategy against SP/CD44+ tumorigenic cells in Akt/beta-catenin-driven hepatocellular carcinoma. J Hepatol. 2020;72(1):104–18. https://doi.org/10.1016/j.jhep.2019.08.035.
Wang D, Dai C, Li Y, Liu Y. Canonical Wnt/beta-catenin signaling mediates transforming growth factor-beta1-driven podocyte injury and proteinuria. Kidney Int. 2011;80(11):1159–69. https://doi.org/10.1038/ki.2011.255.
Wang S, Dong Y, Zhang Y, Wang X, Xu L, Yang S, et al. DACT2 is a functional tumor suppressor through inhibiting Wnt/beta-catenin pathway and associated with poor survival in colon cancer. Oncogene. 2015;34(20):2575–85. https://doi.org/10.1038/onc.2014.201.
Wang W, Wen D, Duan W, Yin J, Cui C, Wang Y, et al. Systemic administration of scAAV9-IGF1 extends survival in SOD1(G93A) ALS mice via inhibiting p38 MAPK and the JNK-mediated apoptosis pathway. Brain Res Bull. 2018;139:203–10. https://doi.org/10.1016/j.brainresbull.2018.02.015.
Wang X, Martindale JL, Holbrook NJ. Requirement for ERK activation in cisplatin-induced apoptosis. J Biol Chem. 2000;275(50):39435–43. https://doi.org/10.1074/jbc.M004583200.
Woelfle J, Chia DJ, Rotwein P. Mechanisms of growth hormone (GH) action. Identification of conserved Stat5 binding sites that mediate GH-induced insulin-like growth factor-I gene activation. J Biol Chem. 2003;278(51):51261–6. https://doi.org/10.1074/jbc.M309486200.
Wong DWL, Yiu WH, Chan KW, Li Y, Li B, Lok SWY, et al. Activated renal tubular Wnt/beta-catenin signaling triggers renal inflammation during overload proteinuria. Kidney Int. 2018;93(6):1367–83. https://doi.org/10.1016/j.kint.2017.12.017.
Yang HL, Tsai YC, Korivi M, Chang CT, Hseu YC. Lucidone promotes the cutaneous wound healing process via activation of the PI(3)K/AKT, Wnt/beta-catenin and NF-kappaB signaling pathways. Biochim Biophys Acta Mol Cell Res. 2017;1864(1):151–68. https://doi.org/10.1016/j.bbamcr.2016.10.021.
Yimit A, Adebali O, Sancar A, Jiang Y. Differential damage and repair of DNA-adducts induced by anti-cancer drug cisplatin across mouse organs. Nat Commun. 2019;10(1):309. https://doi.org/10.1038/s41467-019-08290-2.
Zhou D, Fu H, Xiao L, Mo H, Zhuo H, Tian X, et al. Fibroblast-specific beta-catenin signaling dictates the outcome of AKI. J Am Soc Nephrol. 2018;29(4):1257–71. https://doi.org/10.1681/ASN.2017080903.
Funding
This work was supported by the New Faculty Startup Fund from Seoul National University. This work was supported by the Basic Science Research Program (2021R1A2C1009563) through the National Research Foundation (NRF) of Korea, funded by the Ministry of Science and ICT (MSIT), Republic of Korea.
Author information
Authors and Affiliations
Contributions
Conceptualization of this research, conduct of experiments, acquisition and analysis of data, and drafting the manuscript: CK and WK. Material support, interpretation of data, and performance of statistical analysis: DHW and JK. Acquisition and analysis of data: DBH and NK. Interpretation of data: MK, YJ, and YIP. Conduct of experiments and interpretation of data: JWP. Conceptualization of this research and supervision and editing of final draft of the manuscript: JWY.
Corresponding author
Ethics declarations
Ethics approval
Ethical approval for the experimental protocols was obtained from the Institutional Animal Care and Use Committee of the Catholic University of Korea (2020-022).
Consent to participate
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kim, C., Kwak, W., Won, DH. et al. Loss of Dact2 alleviates cisplatin-induced nephrotoxicity through regulation of the Igfl-MAPK pathway axis. Cell Biol Toxicol 39, 3197–3217 (2023). https://doi.org/10.1007/s10565-023-09827-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-023-09827-4