Abstract
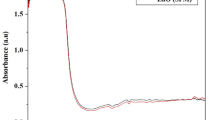
Tryptic hydrolysates of protein fractions obtained by the Osborne method from chickpea (Cicer arietinum L.) seeds interacted with zinc ions and the results of chelation were monitored by the Energy Dispersive X-Ray (EDX) technique. The glutelin hydrolysate (GluHyd) reacted with zinc ions and depicted a relatively higher zinc content. For this reason, the zinc complex of the glutelin hydrolysate (GluHyd-Zn) was studied deeper, and 11 peptides were identified in its more zinc-containing second fraction obtained after gel filtration. The peptide HKERVQLHIIPTAVGK showed a relatively higher chelating capacity (57.86 ± 2.14%). According to the result of the ICP-OS analysis, 1 mg peptide could chelate 381.61 ± 133.39 µg zinc, and the molar ratio of peptide-zinc was about 1:4. Spectral methods proved that side chain and C-termini carboxyl groups of the peptide mostly were involved in chelation and N atoms of amino side chains, imidazole group of histidine, and N-termini at some extents were occupied by the metal ions. Modeling of zinc-peptide interaction was done using Molecular Operating Environment (MOE) software. The results of the docking correlate with the experimental data.
ACE2 inhibitory effect of HKERVQLHIIPTAVGK-Zn complex (IC50 = 1.5 mg/mL) was better than that of HKERVQLHIIPTAVGK (IC50 = 2.2 mg/mL).







Similar content being viewed by others
Abbreviations
- AlbHyd:
-
Albumin hydrolysate
- GloHyd:
-
Globulin hydrolysate
- GluHyd:
-
Glutelin hydrolysate
- ProHyd:
-
Prolamin hydrolysate
- AlbHyd-Zn:
-
Albumin hydrolysate-zinc complex
- GloHyd-Zn:
-
Globulin hydrolysate-zinc complex
- GluHyd-Zn:
-
Glutelin hydrolysate-zinc complex
- ProHyd-Zn:
-
Prolamin hydrolysate-zinc complex
- MOE:
-
Molecular Operating Environment
- CPH:
-
Chickpea protein hydrolysate
- CPH-Zn:
-
Chickpea protein hydrolysate-Zn complex
- CPFs:
-
Chickpea protein fractions
- EDX:
-
Energy Dispersive X-Ray
- PAR:
-
4-(2-pyridylazo) resorcinol
References
Grüngreiff K, Reinhold D, Wedemeyer H (2016) The role of zinc in liver cirrhosis. Ann Hepatol 15:7–16. https://doi.org/10.5604/16652681.1184191
Wessels I, Rolles B, Rink L (2020) The potential impact of Zinc Supplementation on COVID-19 pathogenesis. Front Immunol 11:1712–1712. https://doi.org/10.3389/fimmu.2020.01712
Pradeep H, Najma U, Aparna HS (2021) Milk peptides as novel multi-targeted therapeutic candidates for SARS-CoV2. Protein J 40:310–327. https://doi.org/10.1007/s10930-021-09983-8
Mostafa-Hedeab G (2020) ACE2 as Drug Target of COVID-19 Virus Treatment, simplified updated review. Rep Biochem Mol Biol 9:97–105. https://doi.org/10.29252/rbmb.9.1.97
Liu LP, Zhang XL, Li J (2021) New perspectives on angiotensin-converting enzyme 2 and its related diseases. World J Diabetes 12:839–854. https://doi.org/10.4239/wjd.v12.i6.839
Prasad AS (2013) Discovery of human zinc deficiency: its impact on human health and disease. Adv Nutr 4:176–190. https://doi.org/10.3945/an.112.003210
Bhowmik D, Bhattacharjee C, Kumar S (2010) A potential medicinal importance of zinc in human health and chronic disease. Int J Pharm Biomed Sci 1:5–11
Jothimani D, Kailasam E, Danielraj S, Nallathambi B, Ramachandran H, Sekar P, Manoharan S, Ramani V, Narasimhan G, Kaliamoorthy I, Rela M (2020) COVID-19: poor outcomes in patients with zinc deficiency. Int J Infect Dis 100:343–349. https://doi.org/10.1016/j.ijid.2020.09.014
Plum LM, Rink L, Haase H (2010) The essential toxin: impact of zinc on human health. Int J Environ Res Public Health 7:1342–1365. https://doi.org/10.3390/ijerph7041342
Liao W, Lai T, Chen L, Fu J, Sreenivasan ST, Yu Z, Ren J (2016) Synthesis and characterization of a Walnut peptides-zinc complex and its antiproliferative activity against human breast carcinoma cells through the induction of apoptosis. J Agric Food Chem 64:1509–1519. https://doi.org/10.1021/acs.jafc.5b04924
Fang Z, Xu L, Lin Y, Cai X, Wang S (2018) The preservative potential of Octopus scraps peptides – zinc chelate against Staphylococcus aureus: its fabrication, antibacterial activity and action mode. Food Control 98. https://doi.org/10.1016/j.foodcont.2018.11.015
Miquel E, Farré R (2007) Effects and future trends of casein phosphopeptides on zinc bioavailability. Trends Food Sci Technol 18:139–143. https://doi.org/10.1016/j.tifs.2006.11.004
Sauer AK, Pfaender S, Hagmeyer S, Tarana L, Mattes AK, Briel F, Küry S, Boeckers TM, Grabrucker AM (2017) Characterization of zinc amino acid complexes for zinc delivery in vitro using Caco-2 cells and enterocytes from hiPSC. Biometals 30:643–661. https://doi.org/10.1007/s10534-017-0033-y
Lönnerdal B (2000) Dietary factors influencing zinc absorption. J Nutr 130:1378s–1383s. https://doi.org/10.1093/jn/130.5.1378S
Wang C, Li B, Ao J (2012) Separation and identification of zinc-chelating peptides from sesame protein hydrolysate using IMAC-Zn2² and LC-MS/MS. Food Chem 134:1231–1238. https://doi.org/10.1016/j.foodchem.2012.02.204
Zhu K-X, Wang X-P, Guo X-N (2015) Isolation and characterization of zinc-chelating peptides from wheat germ protein hydrolysates. JFF 12:23–32. https://doi.org/10.1016/j.jff.2014.10.030
Xie N, Huang J, Li B, Cheng J, Wang Z, Yin J, Yan X (2015) Affinity purification and characterisation of zinc chelating peptides from rapeseed protein hydrolysates: possible contribution of characteristic amino acid residues. Food Chem 173:210–217. https://doi.org/10.1016/j.foodchem.2014.10.030
Fu T, Zhang S, Sheng Y, Feng Y, Jiang Y, Zhang Y, Yu M, Wang C (2020) Isolation and characterization of zinc-binding peptides from mung bean protein hydrolysates. Eur Food Res Technol 246. https://doi.org/10.1007/s00217-019-03397-8
Zhu S, Zheng Y, He S, Su D, Nag A, Zeng Q, Yuan Y (2021) Novel Zn-Binding peptide isolated from soy protein hydrolysates: purification, structure, and digestion. J Agric Food Chem 69:483–490. https://doi.org/10.1021/acs.jafc.0c05792
Chen D, Liu Z, Huang W, Zhao Y, Dong S, Zeng M (2013) Purification and characterisation of a zinc-binding peptide from oyster protein hydrolysate. JFF 5:689–697. https://doi.org/10.1016/j.jff.2013.01.012
Chen Q, Guo L, Du F, Chen T, Hou H, Li B (2017) The chelating peptide (GPAGPHGPPG) derived from Alaska pollock skin enhances calcium, zinc and iron transport in Caco-2 cells. Int J Food Sci Technol 52:1283–1290. https://doi.org/10.1111/ijfs.13396
Liu X, Wang Z, Zhang J, Song L, Li D, Wu Z, Zhu B, Nakamura Y, Shahidi F, Yu C, Zhou D (2019) Isolation and identification of zinc-chelating peptides from sea cucumber (Stichopus japonicus) protein hydrolysate. J Sci Food Agric 99:6400–6407. https://doi.org/10.1002/jsfa.9919
Lupaescu AV, Mocanu CS, Drochioiu G, Ciobanu CI (2021) Zinc binding to NAP-Type neuroprotective peptides: nuclear magnetic resonance studies and molecular modeling. Pharmaceuticals (Basel) 14. https://doi.org/10.3390/ph14101011
Al-Snafi A (2016) The medical importance of Cicer arietinum -A review. IOSR J Pharm 6:29–40
Jukanti AK, Gaur PM, Gowda CLL, Chibbar RN (2012) Nutritional quality and health benefits of chickpea (Cicer arietinum L.): a review. BJN 108:S11–S26. https://doi.org/10.1017/S0007114512000797
Ahangaran M (2022) Bioactive peptides and antinutrients in chickpea: description and properties (a review). Trudy po prikladnoj botanike, genetike i selekcii v. 183:pp. 214-223-2022 v.2183 no.2021 https://doi.org/10.30901/2227-8834-2022-1-214-223
Real Hernandez LM (2019) Gonzalez de Mejia EJCrifs, safety f. CRFSFS 18:1913–1946
Megías C, Pedroche J, Yust MM, Girón-Calle J, Alaiz M, Millan F, Vioque J (2007) Affinity purification of copper chelating peptides from chickpea protein hydrolysates. J Agric Food Chem 55:3949–3954. https://doi.org/10.1021/jf063401s
Mukhamedov N, Mirzaakhmedov SY, Gao YH, Waili A, Ziyavitdinov ZF, Bozorov SS, Aisa HA, Yili A (2022) Zinc-binding peptides from protein of Cicer arietinum. Chem Nat Compd 58:86–89. https://doi.org/10.1007/s10600-022-03602-3
Agboola S, Ng D, Mills D (2005) Characterisation and functional properties of australian rice protein isolates. J Cereal Sci 41:283–290. https://doi.org/10.1016/j.jcs.2004.10.007
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. JBC 193:265–275
Wubulikasimu A, Omar A, Gao Y, Mukhamedov N, Arken A, Wali A, Mirzaakhmedov SY, Yili A (2021) Antioxidant hydrolysate of Sericin from Bombyx mori Cocoons. Chem Nat Compd 57:346–349. https://doi.org/10.1007/s10600-021-03348-4
Mukhamedov N, Wubulikasimu A, Rustamova N, Nuerxiati R, Mirzaakhmedov S, Ishimov U, Ziyavitdinov J, Yili A, Aisa H (2021) Synthesis and characterization of Novel chickpea protein hydrolysate-vanadium complexes having cell Inhibitory Effects on Lung Cancer A549 cells lines. Protein J 40. https://doi.org/10.1007/s10930-021-09979-4
Udechukwu MC, Downey B, Udenigwe CC (2018) Influence of structural and surface properties of whey-derived peptides on zinc-chelating capacity, and in vitro gastric stability and bioaccessibility of the zinc-peptide complexes. Food Chem 240:1227–1232. https://doi.org/10.1016/j.foodchem.2017.08.063
Soto-Madrid D, Pérez N, Gutiérrez Cutiño M, Matiacevich S, Zúñiga R (2022) Structural and physicochemical characterization of extracted proteins fractions from Chickpea (Cicer arietinum L.) as a potential food ingredient to Replace Ovalbumin in Foams and Emulsions. Polymers 1:110 https://doi.org/10.3390/polym15010110
Tavano OL, Neves VA (2008) Isolation, solubility and in vitro hydrolysis of chickpea vicilin-like protein. Food Sci Technol 41:1244–1251. https://doi.org/10.1016/j.lwt.2007.08.003
Wang R, He S, Xuan Y, Cheng C (2020) Preparation and characterization of whey protein hydrolysate-Zn complexes. J Food Meas Charact 14:254–261. https://doi.org/10.1007/s11694-019-00287-1
Dunbar RC (2015) Spectroscopy of metal-ion complexes with peptide-related ligands. Top Curr Chem 364:183–223. https://doi.org/10.1007/128_2014_578
Udechukwu MC, Downey B, Udenigwe CC (2018) Influence of structural and surface properties of whey-derived peptides on zinc-chelating capacity, and in vitro gastric stability and bioaccessibility of the zinc-peptide complexes. Food Chem 240:1227–1232. https://doi.org/10.1016/j.foodchem.2017.08.063
Fang Z, Xu L, Lin Y, Cai X, Wang S (2019) The preservative potential of Octopus scraps peptides – zinc chelate against Staphylococcus aureus: its fabrication, antibacterial activity and action mode. Food Control 98:24–33. https://doi.org/10.1016/j.foodcont.2018.11.015
Fu T, Zhang S, Sheng Y, Feng Y, Jiang Y, Zhang Y, Yu M, Wang C (2020) Isolation and characterization of zinc-binding peptides from mung bean protein hydrolysates. Eur Food Res Technol 246:113–124. https://doi.org/10.1007/s00217-019-03397-8
Wang Q, Xiong YL (2018) Zinc-binding behavior of hemp protein hydrolysates: Soluble versus insoluble zinc-peptide complexes. J Funct Foods 49:105–112. https://doi.org/10.1016/j.jff.2018.08.019
Fan W, Wang Z, Mu Z, Du M, Jiang L, Ei-Seedi H, Wang C (2020) Characterizations of a Food Decapeptide chelating with zn(II). eFood 1 https://doi.org/10.2991/efood.k.200727.001
Ke X, Hu X, Li L, Yang X, Chen S, Wu Y, Xue C (2021) A novel zinc-binding peptide identified from tilapia (Oreochromis niloticus) skin collagen and transport pathway across Caco-2 monolayers. Food Biosci 42:101127. https://doi.org/10.1016/j.fbio.2021.101127
Wang C, Li B, Ao J (2012) Separation and identification of zinc-chelating peptides from sesame protein hydrolysate using IMAC-Zn2 + and LC–MS/MS. Food Chem 134:1231–1238. https://doi.org/10.1016/j.foodchem.2012.02.204
Sun X, Sarteshnizi RA, Boachie RT, Okagu OD, Abioye RO, Pfeilsticker Neves R, Ohanenye IC, Udenigwe CC (2020) Peptide-Mineral Complexes: understanding their chemical interactions, bioavailability, and potential application in Mitigating Micronutrient Deficiency. Foods 9 https://doi.org/10.3390/foods9101402
Lu W, Dong C (2022) Research progress of metal chelating peptides. Food and Health 4:17–22. https://doi.org/10.53388/FH20221101019
Mayor-Ibarguren A, Busca-Arenzana C, Robles-Marhuenda Á (2020) A hypothesis for the possible role of Zinc in the immunological pathways related to COVID-19 infection. 11 https://doi.org/10.3389/fimmu.2020.01736
Speth RC, Carrera EJ, Jean-Baptiste M, Joachim A, Linares AJTFJ (2014) Concentration-dependent effects of zinc on angiotensin-converting enzyme-2 activity (1067.4). FASEB J 28:1067. https://doi.org/10.1096/fasebj.28.1_supplement.1067.4
Acknowledgements
The work was supported by CAS-TWAS President’s Fellowship Program.
This work was Funded by the Xinjiang Uygur Autonomous Region Tianshan Yingcai-Leading Talents in Scientific and Technological Innovation (Grand No. 2022TSYCLJ0008), Scientific Report of Foreign Expert by the PIFI Fund of the Chinese Academy of Sciences for invited scientists, 2019VBA0013, Chinese Academy of Sciences President’s International Fellowship Initiative (Grant No. 2023VBB0005), and National Foreign Expert Program “Foreign Young Talent Program (Grant No. QN2022045004L). We also thank Soyib Bozorov, Oybek Tursunkulov, and Uchqun Ishimov for their technical support.
Author information
Authors and Affiliations
Contributions
Nurkhodja Mukhamedov, Akmal Asrorov, Sharafitdin Mirzaakhmedov, Haji Akber Aisa, and Abulimiti Yili wrote the main manuscript text. Ansor Yashinov and Ahmidin Wali conducted experiments of biological activity screening and ACE2 inhibitory activity test. Muzaffar Kayumov made molecular modelling of peptide-zinc interaction. All authors reviewed the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors declare no conflict of interest.
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mukhamedov, N., Asrorov, A., Yashinov, A. et al. Synthesis and Characterisation of Chickpea Peptides-Zinc Chelates Having ACE2 Inhibitory Activity. Protein J 42, 547–562 (2023). https://doi.org/10.1007/s10930-023-10133-5
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10930-023-10133-5