Abstract
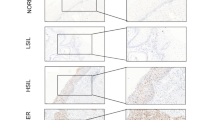
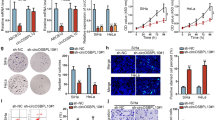
Cervical cancer (CC) is the second most common type of cancer in women, and presents a serious threat to public health. We aimed to investigate the regulatory impacts of CDGSH iron-sulfur domain-containing protein 2 (CISD2) in CC and to discuss its relationship with E2F transcription factor 7 (E2F7). With the employment of real-time reverse transcriptase-polymerase chain reaction (RT-qPCR) and western blot, the expression of CISD2 and E2F7 in SiHa cells before or after transfection was estimated. Cell counting kit-8 (CCK-8) assay, Terminal deoxynucleotidyl transferase (TdT) dUTP Nick-End Labeling (TUNEL) assay, wound healing and transwell were used to detect the proliferation, apoptosis, migration and invasion of SiHa cells. The activity of CISD2 was detected using luciferase report assay and chromatin immunoprecipitation (ChIP) assay was used to confirm the binding of E2F7 and CISD2 promoter. The contents of proliferation- and apoptosis-related proteins were detected using western blot. Results revealed that CISD2 expression was greatly enhanced in CC cell lines. CISD2 depletion inhibited the proliferation, migration and invasion of SiHa cells but promoted the cell apoptosis. It was also found that E2F7 was remarkably elevated in SiHa cells. According to JASPAR database, the binding sites of E2F7 and CISD2 were predicted and ChIP confirmed the binding of E2F7 and CISD2 promoter. Results obtained from luciferase report assay indicated that E2F7 overexpression increased the activity of CISD2 promoter region. Furthermore, further functional experiments demonstrated that the impacts of E2F7 interference on the proliferation, migration, invasion and apoptosis of SiHa cells were reversed by CISD2 overexpression. In summary, CISD2 silence could alleviate the malignant progression of CC and could be transcribed by E2F7.






Similar content being viewed by others
Data Availability
The experimental data will be available from the corresponding author on reasonable request.
References
Carvajal LA, Hamard PJ, Tonnessen C, Manfredi JJ (2012) E2F7, a novel target, is up-regulated by p53 and mediates DNA damage-dependent transcriptional repression. Genes Dev 26(14):1533–1545. https://doi.org/10.1101/gad.184911.111
Chaichian S, Shafabakhsh R, Mirhashemi SM, Moazzami B, Asemi Z (2020) Circular RNAs: a novel biomarker for cervical cancer. J Cell Physiol 235(2):718–724. https://doi.org/10.1002/jcp.29009
de Bruin A, Maiti B, Jakoi L, Timmers C, Buerki R, Leone G (2003) Identification and characterization of E2F7, a novel mammalian E2F family member capable of blocking cellular proliferation. J Biol Chem 278(43):42041–42049. https://doi.org/10.1074/jbc.M308105200
Gao F, Xu T, Wang X, Zhong S, Chen S, Zhang M et al (2017) CIP2A mediates fibronectin-induced bladder cancer cell proliferation by stabilizing beta-catenin. J Exp Clin Cancer Res 36(1):70. https://doi.org/10.1186/s13046-017-0539-8
Guo X, Liu L, Zhang Q, Yang W, Zhang Y (2020) E2F7 transcriptionally inhibits MicroRNA-199b expression to promote USP47, thereby enhancing Colon cancer tumor stem cell activity and promoting the occurrence of Colon cancer. Front Oncol 10:565449. https://doi.org/10.3389/fonc.2020.565449
Herrera FG, Chan P, Doll C, Milosevic M, Oza A, Syed A et al (2007) A prospective phase I-II trial of the cyclooxygenase-2 inhibitor celecoxib in patients with carcinoma of the cervix with biomarker assessment of the tumor microenvironment. Int J Radiat Oncol Biol Phys 67(1):97–103. https://doi.org/10.1016/j.ijrobp.2006.08.024
Hertlein L, Lenhard M, Kirschenhofer A, Kahlert S, Mayr D, Burges A et al (2011) Cetuximab monotherapy in advanced cervical cancer: a retrospective study with five patients. Arch Gynecol Obstet 283(1):109–113. https://doi.org/10.1007/s00404-010-1389-1
Holt SH, Darash-Yahana M, Sohn YS, Song L, Karmi O, Tamir S et al (2016) Activation of apoptosis in NAF-1-deficient human epithelial breast cancer cells. J Cell Sci 129(1):155–165. https://doi.org/10.1242/jcs.178293
Huang YL, Shen ZQ, Huang CH, Lin CH, Tsai TF (2021) Cisd2 slows down liver aging and attenuates age-related metabolic dysfunction in male mice. Aging Cell 20(12):e13523. https://doi.org/10.1111/acel.13523
Kaczanowski S (2016) Apoptosis: its origin, history, maintenance and the medical implications for cancer and aging. Phys Biol 13(3):031001. https://doi.org/10.1088/1478-3975/13/3/031001
Kim JY, Byun SJ, Kim YS, Nam JH (2017) Disease courses in patients with residual tumor following concurrent chemoradiotherapy for locally advanced cervical cancer. Gynecol Oncol 144(1):34–39. https://doi.org/10.1016/j.ygyno.2016.10.032
Li SM, Chen CH, Chen YW, Yen YC, Fang WT, Tsai FY et al (2017) Upregulation of CISD2 augments ROS homeostasis and contributes to tumorigenesis and poor prognosis of lung adenocarcinoma. Sci Rep 7(1):11893. https://doi.org/10.1038/s41598-017-12131-x
Liao HY, Liao B, Zhang HH (2021) CISD2 plays a role in age-related diseases and cancer. Biomed Pharmacother 138:111472. https://doi.org/10.1016/j.biopha.2021.111472
Liu L, Xia M, Wang J, Zhang W, Zhang Y, He M et al (2014) CISD2 expression is a novel marker correlating with pelvic lymph node metastasis and prognosis in patients with early-stage cervical cancer. Med Oncol 31(9):183. https://doi.org/10.1007/s12032-014-0183-5
Ma YS, Lv ZW, Yu F, Chang ZY, Cong XL, Zhong XM et al (2018) MicroRNA-302a/d inhibits the self-renewal capability and cell cycle entry of liver cancer stem cells by targeting the E2F7/AKT axis. J Exp Clin Cancer Res 37(1):252. https://doi.org/10.1186/s13046-018-0927-8
Nevins JR (2001) The Rb/E2F pathway and cancer. Hum Mol Genet 10(7):699–703. https://doi.org/10.1093/hmg/10.7.699
Pistritto G, Trisciuoglio D, Ceci C, Garufi A, D’Orazi G (2016) Apoptosis as anticancer mechanism: function and dysfunction of its modulators and targeted therapeutic strategies. Aging 8(4):603–619. https://doi.org/10.18632/aging.100934
Revathidevi S, Murugan AK, Nakaoka H, Inoue I, Munirajan AK (2021) APOBEC: a molecular driver in cervical cancer pathogenesis. Cancer Lett 496:104–116. https://doi.org/10.1016/j.canlet.2020.10.004
Saadi AA (2021) Development and Validation of Questionnaire to assess knowledge about Cervical Cancer among Women aged 20 to 65 years in Oman. Asian Pac J Cancer Prev 22(1):69–74. https://doi.org/10.31557/APJCP.2021.22.1.69
Saei Ghare Naz M, Kariman N, Ebadi A, Ozgoli G, Ghasemi V, Rashidi Fakari F (2018) Educational Interventions for Cervical Cancer Screening Behavior of Women: a systematic review. Asian Pac J Cancer Prev 19(4):875–884. https://doi.org/10.22034/APJCP.2018.19.4.875
Schiffman M, Solomon D (2013) Clinical practice. Cervical-cancer screening with human papillomavirus and cytologic cotesting. N Engl J Med 369(24):2324–2331. https://doi.org/10.1056/NEJMcp1210379
Shao F, Li Y, Hu W, Yu J, Wu H, Ying K et al (2020) Downregulation of CISD2 has Prognostic Value in Non-Small Cell Lung Cancer and inhibits the tumorigenesis by inducing mitochondrial dysfunction. Front Oncol 10:595524. https://doi.org/10.3389/fonc.2020.595524
Shen YY, Cui JY, Yuan J, Wang X (2018) MiR-451a suppressed cell migration and invasion in non-small cell lung cancer through targeting ATF2. Eur Rev Med Pharmacol Sci 22(17):5554–5561. https://doi.org/10.26355/eurrev_201809_15818
Suh DH, Kim JW, Aziz MF, Devi UK, Ngan HY, Nam JH et al (2010) Asian society of gynecologic oncology workshop 2010. J Gynecol Oncol 21(3):137–150. https://doi.org/10.3802/jgo.2010.21.3.137
Sun AG, Meng FG, Wang MG (2017a) CISD2 promotes the proliferation of glioma cells via suppressing beclin1mediated autophagy and is targeted by microRNA449a. Mol Med Rep 16(6):7939–7948. https://doi.org/10.3892/mmr.2017.7642
Sun Y, Jiang Y, Huang J, Chen H, Liao Y, Yang Z (2017b) CISD2 enhances the chemosensitivity of gastric cancer through the enhancement of 5-FU-induced apoptosis and the inhibition of autophagy by AKT/mTOR pathway. Cancer Med 6(10):2331–2346. https://doi.org/10.1002/cam4.1169
Tewari KS, Sill MW, Long HJ 3rd, Penson RT, Huang H, Ramondetta LM et al (2014) Improved survival with bevacizumab in advanced cervical cancer. N Engl J Med 370(8):734–743. https://doi.org/10.1056/NEJMoa1309748
Weijts BG, Bakker WJ, Cornelissen PW, Liang KH, Schaftenaar FH, Westendorp B et al (2012) E2F7 and E2F8 promote angiogenesis through transcriptional activation of VEGFA in cooperation with HIF1. EMBO J 31(19):3871–3884. https://doi.org/10.1038/emboj.2012.231
Xie Q, Tang T, Pang J, Xu J, Yang X, Wang L (2020) LSD1 promotes bladder Cancer progression by upregulating LEF1 and enhancing EMT. Front Oncol 10:1234. https://doi.org/10.3389/fonc.2020.01234
Yang L, Hong S, Wang Y, He Z, Liang S, Chen H (2016) A novel prognostic score model incorporating CDGSH iron sulfur domain2 (CISD2) predicts risk of disease progression in laryngeal squamous cell carcinoma. Oncotarget 7(16):22720–22732. https://doi.org/10.18632/oncotarget.8150
Zong S, Liu X, Zhou N, Yue Y (2019) E2F7, EREG, miR-451a and miR-106b-5p are associated with the cervical cancer development. Arch Gynecol Obstet 299(4):1089–1098. https://doi.org/10.1007/s00404-018-5007-y
Acknowledgements
Not applicable.
Funding
This work was supported by the Natural Science Research Project of Anhui Educational Committee (Project No. KJ2021A0754) and Bengbu Science and Technology Innovation Guidance Project in 2021 (Project No. 20210337).
Author information
Authors and Affiliations
Contributions
Lingling Wang, Yan Wang and Guoliu Ye designed the study and conducted the experiments. Lingling Wang, Caizhi Wang and Kang Yang analyzed the data. Yan Wang and Caizhi Wang drafted the manuscript and interpreted the data. Kang Yang and Guoliu Ye revised the manuscript for important intellectual content. All authors have read and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
Not applicable.
Patient consent for publication
Not applicable.
Competing interests
The authors declare no competing financial interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, L., Wang, Y., Wang, C. et al. CISD2 transcriptional activated by transcription factor E2F7 promotes the malignant progression of cervical cancer. J Mol Histol 54, 489–498 (2023). https://doi.org/10.1007/s10735-023-10145-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-023-10145-6