Abstract
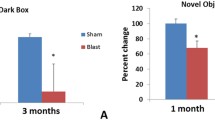
Traumatic brain injury (TBI), a neurovascular injury caused by external force, is a common diagnosis among veterans and those experiencing homelessness (HL). There is a significant overlap in the veteran and homeless population, possibly accounting for the two to seven times greater incidence of TBI among those experiencing HL than the general population. Despite these statistics, individuals experiencing HL are often underdiagnosed and ineffectively treated for TBI. We introduced a novel model of HL. Over 5 weeks, adult Sprague–Dawley rats were randomly assigned to one of the following conditions: TBI only, HL only, TBI + HL, or control (n = 9 per group). To emulate HL, animals (2 animals per cage) were exposed to soiled beddings for 5 weeks. Subsequently, animals were introduced to TBI by using the moderate controlled cortical impact model, then underwent 4 consecutive days of behavioral testing (beam walk (BW), elevated body swing test (EBST), forelimb akinesia (FA), paw grasp (PG), Rotorod, and elevated T-maze). Nissl staining was performed to determine the peri-impact cell survival and the integrity of corpus callosum area. Motor function was significantly impaired by TBI, regardless of housing (beam walk or BW 85.0%, forelimb akinesia or FA 104.7%, and paw grasp or PG 100% greater deficit compared to control). Deficits were worsened by HL in TBI rats (BW 93.3%, FA 40.5%, and PG 50% greater deficit). Two-way ANOVA revealed BW (F(4, 160) = 31.69, p < 0.0001), FA (F(4, 160) = 13.71, p < 0.0001), PG (F(4, 160) = 3.873, p = 0.005), Rotorod (F(4, 160), p = 1.116), and EBST (F(4, 160) = 6.929, p < 0.0001) showed significant differences between groups. The Rotorod and EBST tests showed TBI-induced functional deficits when analyzed by day, but these deficits were not exacerbated by HL. TBI only and TBI + HL rats exhibited typical cortical impact damage (F(3,95) = 51.75, p < 0.0001) and peri-impact cell loss compared to control group (F(3,238) = 47.34, p < 0.0001). Most notably, TBI + HL rats showed significant alterations in WM area measured via the corpus callosum (F(3, 95) = 3.764, p = 0.0133). Worsened behavioral outcomes displayed by TBI + HL rats compared to TBI alone suggest HL contributes to TBI functional deficits. While an intact white matter, such as the corpus callosum, may lessen the consequent functional deficits associated with TBI by enhancing hemispheric communications, there are likely alternative cellular and molecular pathways mitigating TBI-associated inflammatory or oxidative stress responses. Here, we showed that the environmental condition of the patient, i.e., HL, participates in white matter integrity and behavioral outcomes, suggesting its key role in the disease diagnosis to aptly treat TBI patients.

Similar content being viewed by others
Data Availability
All research data are available from Cesar V. Borlongan upon reasonable request.
Abbreviations
- TBI:
-
Traumatic brain injury
- HL:
-
Homelessness
- CC:
-
Corpus callosum
- WM:
-
White matter
- GM:
-
Gray matter
- BW:
-
Beam walk
- EBST:
-
Elevated body swing test
- FA:
-
Forelimb akinesia
- PG:
-
Paw grasp
References
Gardner RC, Yaffe K. Epidemiology of mild traumatic brain injury and neurodegenerative disease. Mol Cell Neurosci. 2015;66(Pt B):75–80.
Chapman JC, Diaz-Arrastia R. Military traumatic brain injury: a review. Alzheimers Dement. 2014;10(3 Suppl):S97-104.
Hoge CW, et al. Mild traumatic brain injury in U.S. soldiers returning from Iraq. N Engl J Med. 2008;358(5):453–63.
Tanielian TL, et al. Editors, Invisible wounds of war: psycho- logical and cognitive injuries, their consequences, and services to assist recovery. Santa Monica, CA: Rand Corporation. 2008, Rand Corporation: Santa Monica, CA.
Defense and Veterans Brain Injury Center. Available from http://dvbic.dcoe.mil/dod-worldwide-numbers-tbi.
Elder GA. Update on TBI and cognitive impairment in military veterans. Curr Neurol Neurosci Rep. 2015;15(10):68.
Vasterling JJ, Dikmen S. Mild traumatic brain injury and posttraumatic stress disorder: clinical and conceptual complexities. J Int Neuropsychol Soc. 2012;18(3):390–3.
Monsour M, Ebedes D, Borlongan CV. A review of the pathology and treatment of TBI and PTSD. Exp Neurol. 2022;351:114009.
Tsai J, Rosenheck RA. Risk factors for homelessness among US veterans. Epidemiol Rev. 2015;37:177–95.
US Department of Housing and Urban Development, The 2013 Annual Homeless Assessment Report (AHAR) to Congress: part 1, point-in-time estimates of homelessness. 2013, US Department of Housing and Urban Development: Washington, D.C.
US Department of Veterans Affairs, Profile of veterans. 2013. Washington, DC: US Department of Veterans Affairs; 2011.
Hwang SW, et al. The effect of traumatic brain injury on the health of homeless people. CMAJ. 2008;179(8):779–84.
To MJ, et al. Healthcare utilization, legal incidents, and victimization following traumatic brain injury in homeless and vulnerably housed individuals: a prospective cohort study. J Head Trauma Rehabil. 2015;30(4):270–6.
Cusimano MD, et al. Cognitive dysfunction, brain volumes, and traumatic brain injury in homeless persons. Neurotrauma Rep. 2021;2(1):136–48.
McMillan TM, et al. Head injury and mortality in the homeless. J Neurotrauma. 2015;32(2):116–9.
Lafferty B. Traumatic brain injury: a factor in the causal pathway to homelessness? J Nurse Pract. 2010;6:358–62.
Solliday-McRoy C, et al. Neuropsychological functioning of homeless men. J Nerv Ment Dis. 2004;192(7):471–8.
Goering PN, et al. The At Home/Chez Soi trial protocol: a pragmatic, multi-site, randomised controlled trial of a Housing First intervention for homeless individuals with mental illness in five Canadian cities. BMJ Open. 2011;1(2):e000323.
Stergiopoulos V, et al. Neurocognitive impairment in a large sample of homeless adults with mental illness. Acta Psychiatr Scand. 2015;131(4):256–68.
Topolovec-Vranic J, et al. The high burden of traumatic brain injury and comorbidities amongst homeless adults with mental illness. J Psychiatr Res. 2017;87:53–60.
Noel F, et al. A longitudinal study of suicidal ideation among homeless, mentally ill individuals. Soc Psychiatry Psychiatr Epidemiol. 2016;51(1):107–14.
Brisson D, et al. A systematic review of the association between poverty and biomarkers of toxic stress. J Evid Based Soc Work (2019). 2020;17(6):696–713.
Hernandez-Ontiveros DG, et al. Microglia activation as a biomarker for traumatic brain injury. Front Neurol. 2013;4:30.
Lozano D, et al. Neuroinflammatory responses to traumatic brain injury: etiology, clinical consequences, and therapeutic opportunities. Neuropsychiatr Dis Treat. 2015;11:97–106.
Altumbabic M. Peeling J, Del Bigio MR. Intracerebral hemorrhage in the rat: effects of hematoma aspiration. Stroke. 1998;29(9):1917–22; discussion 1922–3.
Borlongan CV, Sanberg PR. Elevated body swing test: a new behavioral parameter for rats with 6-hydroxydopamine-induced hemiparkinsonism. J Neurosci. 1995;15(7 Pt 2):5372–8.
Glover LE, et al. Immediate, but not delayed, microsurgical skull reconstruction exacerbates brain damage in experimental traumatic brain injury model. PLoS ONE. 2012;7(3):e33646.
Hayashi T, et al. Quantitative analyses of matrix metalloproteinase activity after traumatic brain injury in adult rats. Brain Res. 2009;1280:172–7.
Yu S, et al. Severity of controlled cortical impact traumatic brain injury in rats and mice dictates degree of behavioral deficits. Brain Res. 2009;1287:157–63.
Tajiri N, et al. Intravenous transplants of human adipose-derived stem cell protect the brain from traumatic brain injury-induced neurodegeneration and motor and cognitive impairments: cell graft biodistribution and soluble factors in young and aged rats. J Neurosci. 2014;34(1):313–26.
Hinkley LB, et al. The role of corpus callosum development in functional connectivity and cognitive processing. PLoS ONE. 2012;7(8):e39804.
Filley CM, Kelly JP. White matter and cognition in traumatic brain injury. J Alzheimers Dis. 2018;65(2):345–62.
Rutgers DR, et al. White matter abnormalities in mild traumatic brain injury: a diffusion tensor imaging study. AJNR Am J Neuroradiol. 2008;29(3):514–9.
Berginstrom N, et al. White matter hyperintensities increases with traumatic brain injury severity: associations to neuropsychological performance and fatigue. Brain Inj. 2020;34(3):415–20.
Johnson AD, et al. Effects of stressful life events on cerebral white matter hyperintensity progression. Int J Geriatr Psychiatry. 2017;32(12):e10–7.
Taylor WD, et al. Evidence of white matter tract disruption in MRI hyperintensities. Biol Psychiatry. 2001;50(3):179–83.
Moen KG, et al. Traumatic axonal injury: the prognostic value of lesion load in corpus callosum, brain stem, and thalamus in different magnetic resonance imaging sequences. J Neurotrauma. 2014;31(17):1486–96.
Magalhaes R, et al. White matter changes in microstructure associated with a maladaptive response to stress in rats. Transl Psychiatry. 2017;7(1):e1009.
Stubbs JL, et al. Differential age-associated brain atrophy and white matter changes among homeless and precariously housed individuals compared with the general population. BMJ Neurol Open. 2023;5(1):e000349.
Monsour M, Borlongan CV. No one left behind: inclusion of individuals experiencing homelessness in TBI stem cell therapy. Med Hypotheses. 2023;170:111002.
Acosta SA, et al. Influence of post-traumatic stress disorder on neuroinflammation and cell proliferation in a rat model of traumatic brain injury. PLoS ONE. 2013;8(12):e81585.
Greenberg JM, et al. Resilience in homeless veterans: clinical and cognitive correlates. Psychiatr Rehabil J. 2019;42(3):314–22.
Haarbauer-Krupa J, et al. Epidemiology of chronic effects of traumatic brain injury. J Neurotrauma. 2021;38(23):3235–47.
Fluiter K, et al. Inhibition of the membrane attack complex of the complement system reduces secondary neuroaxonal loss and promotes neurologic recovery after traumatic brain injury in mice. J Immunol. 2014;192(5):2339–48.
Bellander BM, et al. Complement activation in the human brain after traumatic head injury. J Neurotrauma. 2001;18(12):1295–311.
Schmidt OI, et al. Closed head injury–an inflammatory disease? Brain Res Brain Res Rev. 2005;48(2):388–99.
Braun M, et al. White matter damage after traumatic brain injury: a role for damage associated molecular patterns. Biochim Biophys Acta Mol Basis Dis. 2017;1863(10 Pt B):2614–2626.
Calcia MA, et al. Stress and neuroinflammation: a systematic review of the effects of stress on microglia and the implications for mental illness. Psychopharmacology. 2016;233(9):1637–50.
Schramm E, Waisman A. Microglia as central protagonists in the chronic stress response. Neurol Neuroimmunol Neuroinflamm. 2022;9(6).
Dowd JB, Goldman N. Do biomarkers of stress mediate the relation between socioeconomic status and health? J Epidemiol Community Health. 2006;60(7):633–9.
Etter K. Building foundations of resilience. 2015, Early Childhood Colorado Partnership.
Shanks TRW, Robinson C. Assets, economic opportunity and toxic stress: a framework for understanding child and educational outcomes. Econ Educ Rev. 2013;33:154–70.
Blanch AK, Shern DL, Staverman SM. Toxic stress, behavioral health, and the next major era in public health. 2014, Mental Health America.
National Scientific Council on the Developing Child. Early experiences can alter gene expression and affect long-term development: working paper no 10. 2010; Available from: http://www.developingchild.net.
Hackman DA, Farah MJ, Meaney MJ. Socioeconomic status and the brain: mechanistic insights from human and animal research. Nat Rev Neurosci. 2010;11(9):651–9.
Abdullah MFILB, Ng YP, Side HB. Depression and anxiety among traumatic brain injury patients in Malaysia. Asian J Psychiatr. 2018;37:67–70.
Scholten AC, et al. Prevalence of and risk factors for anxiety and depressive disorders after traumatic brain injury: a systematic review. J Neurotrauma. 2016;33(22):1969–94.
Barker-Collo S, et al. Depression and anxiety across the first 4 years after mild traumatic brain injury: findings from a community-based study. Brain Inj. 2018;32(13–14):1651–8.
Onapa H, et al. The physical and mental health effects of housing homeless people: a systematic review. Health Soc Care Community. 2022;30(2):448–68.
Gold AL, et al. Cortical thickness and subcortical gray matter volume in pediatric anxiety disorders. Neuropsychopharmacology. 2017;42(12):2423–33.
He E, et al. White matter alterations in depressive disorder. Front Immunol. 2022;13:826812.
Lu M, et al. Cerebral white matter changes in young healthy individuals with high trait anxiety: a tract-based spatial statistics study. Front Neurol. 2018;9:704.
Shad MU, Muddasani S, Rao U. Gray matter differences between healthy and depressed adolescents: a voxel-based morphometry study. J Child Adolesc Psychopharmacol. 2012;22(3):190–7.
Perry RE, et al. Developing a neurobehavioral animal model of poverty: drawing cross-species connections between environments of scarcity-adversity, parenting quality, and infant outcome. Dev Psychopathol. 2019;31(2):399–418.
Harland BC, Dalrymple-Alford JC. Enriched environment procedures for rodents: creating a standardized protocol for diverse enrichment to improve consistency across research studies. Bio Protoc. 2020;10(11):e3637.
Ajagbe͓ A, Ajenikoko MK, Ajiboye EO. An overview of the therapeutic effects of environmental enrichment on traumatic brain injury. J Clin Basic Res. 2021;5(4):9–15.
Galaj E, Barrera ED, Ranaldi R. Therapeutic efficacy of environmental enrichment for substance use disorders. Pharmacol Biochem Behav. 2020;188:172829.
Magee WL, et al. Music interventions for acquired brain injury. Cochrane Database Syst Rev. 2017;1(1):CD006787.
Leon M, Woo C. Environmental enrichment and successful aging. Front Behav Neurosci. 2018;12:155.
Belchev Z, et al. Remotely delivered environmental enrichment intervention for traumatic brain injury: study protocol for a randomised controlled trial. BMJ Open. 2021;11(2):e039767.
Pottie K, et al. Clinical guideline for homeless and vulnerably housed people, and people with lived homelessness experience. CMAJ. 2020;192(10):E240–54.
Teixeira L, Russell D, Hobbs T. The SHARE framework: a smarter way to end homelessness. 2018. http://www.homelesshub.ca/resource/share-framework-smarter-way-end-homelessness.
Leijser LM, Siddiqi A, Miller SP. Imaging evidence of the effect of socio-economic status on brain structure and development. Semin Pediatr Neurol. 2018;27:26–34.
Lezak KR, Missig G, Carlezon WA Jr. Behavioral methods to study anxiety in rodents. Dialogues Clin Neurosci. 2017;19(2):181–91.
Kaplan GB, et al. Pathophysiological bases of comorbidity: traumatic brain injury and post-traumatic stress disorder. J Neurotrauma. 2018;35(2):210–25.
Jacotte-Simancas A, et al. Effects of voluntary physical exercise, citicoline, and combined treatment on object recognition memory, neurogenesis, and neuroprotection after traumatic brain injury in rats. J Neurotrauma. 2015;32(10):739–51.
Jasielski P, et al. Application of citicoline in neurological disorders: a systematic review. Nutrients. 2020;12(10).
Gleeson M, et al. The anti-inflammatory effects of exercise: mechanisms and implications for the prevention and treatment of disease. Nat Rev Immunol. 2011;11(9):607–15.
Kosari-Nasab M, et al. Anxiolytic- and antidepressant-like effects of silymarin compared to diazepam and fluoxetine in a mouse model of mild traumatic brain injury. Toxicol Appl Pharmacol. 2018;338:159–73.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C.V.B. holds leadership position with University of South Florida, a patent holder, and has patent applications on stem cell biology and its therapeutic applications, consultant to a number of stem cell–based companies, and research funding from the NIH. All the other authors declared no potential conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Monsour, M., Lee, JY. & Borlongan, C. An Understated Comorbidity: The Impact of Homelessness on Traumatic Brain Injury. Neurotherapeutics 20, 1446–1456 (2023). https://doi.org/10.1007/s13311-023-01419-8
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13311-023-01419-8