Abstract
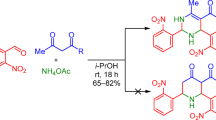
New substituted 2,4,6-triphenylpyrimidine containing the N,N-dimethylamino group in the para-position of the phenyl ring located at the fourth atom of the diazine ring has been obtained. The synthesis is based on the initial preparation of chalcone and its subsequent reaction with ammonium acetate and aldehyde. The thermal, optical, electrochemical, and electrophysical properties of the synthesized pyrimidine have been studied in detail.







Similar content being viewed by others
REFERENCES
S. Achelle, J. Rodríguez-López, and F. R. Guen, “Photoluminescence properties of aryl-, arylvinyl-, and arylethynylpyrimidine derivatives,” Chemistry Select 3, 1852–1886 (2018).
M. Fecková, P. le Poul, F. Bureš, F. Robin-le Guen, and S. Achelle, “Nonlinear optical properties of pyrimidine chromophores,” Dyes Pigm. 182, 108659 (2020).
G. N. Lipunova, E. V. Nosova, V. N. Charushin, and O. N. Chupakhin, “Functionalized quinazolines and pyrimidines for optoelectronic materials,” Curr. Org. Synth. 15, 793–814 (2018).
E. V. Verbitskiy, G. L. Rusinov, O. N. Chupakhin, and V. N. Charushin, “Design of fluorescent sensors based on azaheterocyclic push-pull systems towards nitroaromatic explosives and related compounds: A review,” Dyes Pigm. 180, 108414 (2020).
P. Ganesan, D. G. Chen, J. L. Liao, W. C. Li, Y. N. Lai, D. Luo, and Y. Chi, “Isomeric spiro-[acridine-9,9'-fluorene]-2, 6-dipyridylpyrimidine based TADF emitters: insights into photophysical behaviors and OLED performances,” J. Mater. Chem. 6, 10088–10100.
Q. Zhang, S. Sun, W. Liu, P. Leng, X. Lv, Y. Wang, H. Chen, S. Ye, S. Zhuang, and L. Wang, “integrating TADF luminogens with AIE characteristics using a novel acridine-carbazole hybrid as donor for high-performance and low efficiency roll-off OLEDs,” J. Mater. Chem. 7, 9487–9495.
S. Achelle, J. Rodríguez-López, F. Bureš, and F. Robin-le Guen, “Tuning the photophysical properties of push-pull azaheterocyclic chromophores by protonation: A brief overview of a French-Spanish-Czech Project,” Chem. Record 20, 440–451 (2020).
S. R. Forrest and M. E. Thompson, “Introduction: Organic electronics and optoelectronics,” Chem. Rev. 107, 923–925 (2007).
K. Itami, D. Yamazaki, and J. Yoshida, “Pyrimidine-core extended π-systems: General synthesis and interesting fluorescent properties,” J. Am. Chem. Soc. 126, 15396–15397 (2004).
M. Mastalir, M. Glatz, E. Pittenauer, G. Allmaier, and K. Kirchner, “Rhenium-catalyzed dehydrogenative coupling of alcohols and amines to afford nitrogen-containing aromatics and more,” Org. Lett. 21, 1116–1120 (2019).
M. Mastalir, M. Glatz, E. Pittenauer, G. Allmaier, and K. Kirchner, “Sustainable synthesis of quinolines and pyrimidines catalyzed by manganese PNP pincer complexes,” J. Am. Chem. Soc. 138, 15543–15546 (2016).
Z. C. Wu and D. L. Boger, “Synthesis, characterization, and cycloaddition reactivity of a monocyclic aromatic 1,2,3,5-tetrazine,” J. Am. Chem. Soc. 141, 16388–16397 (2019).
Funding
This work was supported by the Russian Federation Presidential Council for the State Support of Young Scientists and Leading Scientific Schools, MK-4033.2022.1.3.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors of this work declare that they have no conflicts of interest.
Additional information
Translated by O. Polyakov
Rights and permissions
About this article
Cite this article
Slobodinyuk, D.G., Lunegov, I.V., Mayorova, O.A. et al. A New Synthetic Approach to Obtaining 2,4,6-Trisubstituted Pyrimidine and Studying Its Thermal, Optical, Electrochemical, and Electrophysical Properties. Polym. Sci. Ser. D 16, 537–542 (2023). https://doi.org/10.1134/S1995421223030401
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1995421223030401