Abstract
Background
Saline nasal irrigation is an effective therapy for relieving common cold symptoms. This study aimed to investigate and explore the efficacy of physiological seawater nasal irrigation (PSNI) on children with mild and asymptomatic infection with Omicron.
Methods
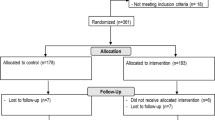
This randomized controlled trial was conducted in Shanghai, China, and 403 children with mild and asymptomatic infection with Omicron were included. These children were allocated into the PSNI group and the control group. The primary outcome was the duration of viral shedding (DVS), and the secondary outcome was the change in clinical symptoms.
Results
The median age of all participants was 5.59 (6.26) years old. The DVS was significantly shorter in the PSNI group [2.40 (1.13)] than in the control group [3.09 (2.14)] (P = 0.014). The multivariable Cox regression model also showed that patients in the PSNI group had an increased probability of shorter DVS compared with patients in the control group [hazard ratio (HR), 1.27; 95% confidence interval (CI), 1.04–1.55; P = 0.017]. Subgroup analysis suggested that the DVS of patients without full vaccination was significantly reduced in the PSNI group. The proportions of runny nose and stuffy nose were apparently reduced in the first three days in the PSNI group or the control group, but there was no evidence showing that PSNI contributes to the benefit compared with the control group.
Conclusion
PSNI can reduce the DVS of patients with mild and asymptomatic infection with SARS-CoV-2 Omicron BA.2 variant.



Similar content being viewed by others
Data availability
The de-identified individual participant dataset used in this article will be made available with publication upon reasonable request.
References
Yin Y, Lin J, Yuan S, Tong S, Chen E, Zheng J, et al. The relationship between early isolation and the duration of viral shedding of mild and asymptomatic infection with SARS-CoV-2 Omicron BA.2 variant. J Infect. 2022;85:e184–6.
Price AM, Olson SM, Newhams MM, Halasa NB, Boom JA, Sahni LC, et al. BNT162b2 protection against the omicron variant in children and adolescents. N Engl J Med. 2022;386:1899–909.
Meinhardt J, Radke J, Dittmayer C, Franz J, Thomas C, Mothes R, et al. Olfactory transmucosal SARS-CoV-2 invasion as a port of central nervous system entry in individuals with COVID-19. Nat Neurosci. 2021;24:168–75.
Moein ST, Hashemian SM, Mansourafshar B, Khorram-Tousi A, Tabarsi P, Doty RL. Smell dysfunction: a biomarker for COVID-19. Int Forum Allergy Rhinol. 2020;10:944–50.
Wang M, Bu X, Fang G, Luan G, Huang Y, Akdis CA, et al. Distinct expression of SARS-CoV-2 receptor ACE2 correlates with endotypes of chronic rhinosinusitis with nasal polyps. Allergy. 2021;76:789–803.
DeGeorge KC, Ring DJ, Dalrymple SN. Treatment of the Common Cold. Am Fam Physician. 2019;100:281–9.
Baxter AL, Schwartz KR, Johnson RW, Kuchinski AM, Swartout KM, Srinivasa Rao ASR, et al. Rapid initiation of nasal saline irrigation to reduce severity in high-risk COVID+ outpatients. Ear Nose Throat J. 2022;1455613221123737.
Yuan L, Zhu H, Zhou M, Ma J, Liu X, Wu K, et al. Nasal irrigation efficiently attenuates SARS-CoV-2 Omicron infection, transmission and lung injury in the Syrian hamster model. iScience. 2022;25:105475.
Štanfel D, Kalogjera L, Ryazantsev SV, Hlača K, Radtsig EY, Teimuraz R, et al. The Role of Seawater and Saline Solutions in Treatment of Upper Respiratory Conditions. Mar Drugs. 2022;20:330.
Zhang Y, Li W. Effects of oral desloratadine citrate disodium combined with physiological seawater nasal irrigation on IgE levels, IL-4, IL-6, IL-13 and IFN-γ expression and treatment of intermittent allergic rhinitis. Cell Mol Biol (Noisy-le-grand). 2020;66:54–8.
Atar Y, Karaketir S, Aydogdu I, Sari H, Bircan HS, Uyar Y, et al. Comparison of isotonic seawater nasal spray containing chamomile liquid extract and other isotonic seawater nasal washing solutions for allergic rhinitis. Ann Otol Rhinol Laryngol. 2022;131:427–34.
Cegolon L, Mastrangelo G, Emanuelli E, Camerotto R, Spinato G, Frezza D. Early Negativization of SARS-CoV-2 Infection by Nasal Spray of Seawater plus Additives: The RENAISSANCE Open-Label Controlled Clinical Trial. Pharmaceutics. 2022;14:2502.
Yin Y, Lin J, Yuan S, Tong S, He Y, Dong B,etl al.: Vaccination status for mild and asymptomatic infections with SARS-CoV-2 Omicron BA.2 variant in Shanghai. Journal of Medical Virology. 2023;95:e28767.
King D, Mitchell B, Williams CP, Spurling GK. Saline nasal irrigation for acute upper respiratory tract infections. Cochrane Database Syst Rev. 2015;2015:Cd006821.
Trimble AT, Whitney Brown A, Laube BL, Lechtzin N, Zeman KL, Wu J, et al. Hypertonic saline has a prolonged effect on mucociliary clearance in adults with cystic fibrosis. J Cyst Fibros. 2018;17:650–6.
Süslü N, Bajin MD, Süslü AE, Oğretmenoğlu O. Effects of buffered 2.3%, buffered 0.9%, and nonbuffered 0.9% irrigation solutions on nasal mucosa after septoplasty. Eur Arch Otorhinolaryngol. 2009;266:685–9.
Keojampa BK, Nguyen MH, Ryan MW. Effects of buffered saline solution on nasal mucociliary clearance and nasal airway patency. Otolaryngol Head Neck Surg. 2004;131:679–82.
Bastier PL, Lechot A, Bordenave L, Durand M, de Gabory L. Nasal irrigation: from empiricism to evidence-based medicine. A review. Eur Ann Otorhinolaryngol Head Neck Dis. 2015;132:281–5.
Machado RRG, Glaser T, Araujo DB, Petiz LL, Oliveira DBL, Durigon GS, et al. Inhibition of severe acute respiratory syndrome coronavirus 2 replication by hypertonic saline solution in lung and kidney epithelial cells. ACS Pharmacol Transl Sci. 2021;4:1514–27.
Casale M, Rinaldi V, Sabatino L, Moffa A, Ciccozzi M. Could nasal irrigation and oral rinse reduce the risk for COVID-19 infection? Int J Immunopathol Pharmacol. 2020;34:2058738420941757.
Burton MJ, Clarkson JE, Goulao B, Glenny AM, McBain AJ, Schilder AG,et al. Antimicrobial mouthwashes (gargling) and nasal sprays administered to patients with suspected or confirmed COVID-19 infection to improve patient outcomes and to protect healthcare workers treating them. Cochrane Database Syst Rev. 2020;9:Cd013627.
Huijghebaert S, Hoste L, Vanham G. Essentials in saline pharmacology for nasal or respiratory hygiene in times of COVID-19. Eur J Clin Pharmacol. 2021;77:1275–93.
Spinato G, Fabbris C, Costantini G, Conte F, Scotton PG, Cinetto F, et al. The effect of isotonic saline nasal lavages in improving symptoms in SARS-CoV-2 infection: a case-control study. Front Neurol. 2021;12:794471.
Ramalingam S, Graham C, Dove J, Morrice L, Sheikh A. A pilot, open labeled, randomized controlled trial of hypertonic saline nasal irrigation and gargling for the common cold. Sci Rep. 2019;9:1015.
Cevik M, Tate M, Lloyd O, Maraolo AE, Schafers J, Ho A. SARS-CoV-2, SARS-CoV, and MERS-CoV viral load dynamics, duration of viral shedding, and infectiousness: a systematic review and meta-analysis. Lancet Microbe. 2021;2:e13-22.
Funding
Health and Family Planning Research Project of Pudong New Area Health Committee (PW2021E-06).
Author information
Authors and Affiliations
Contributions
LJL, ZF, LYB: conceptualization, data curation, formal analysis, writing–original draft. YSH, WJH: data curation. ZJ, ZL: data curation, formal analysis, writing–review and editing. HY, CJ, YY: conceptualization, supervision, writing–review and editing. LJL, ZF, LYB contributed equally to this study. All authors critically reviewed the manuscript. All authors had full access to all the data in the study and had final responsibility for the decision to submit for publication.
Corresponding authors
Ethics declarations
Conflict of interest
No financial or non-financial benefits have been received or will be received from any party related directly or indirectly to the subject of this article.
Ethical approval
The study was approved by the ethics committee of the Shanghai Children's Medical Center and conducted according to the Declaration of Helsinki guidelines (SCMCIRB-K2022036-1).
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, JL., Zhang, F., Li, YB. et al. Efficacy of physiological seawater nasal irrigation for the treatment of children with SARS-CoV-2 Omicron BA.2 variant infection: a randomized controlled trial. World J Pediatr (2023). https://doi.org/10.1007/s12519-023-00749-z
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s12519-023-00749-z