Abstract
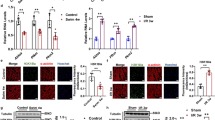
Exercise preconditioning (EP) is a line of scientific inquiry into the short-term biochemical mediators of cardioprotection in the heart. This study examined the involvement of autophagy induced by energy metabolism in myocardial remodelling by EP and myocardial protection. A total of 120 healthy male Sprague Dawley (SD) rats were randomly divided into six groups. Plasma cTnI, HBFP staining and electrocardiographic indicators were examined in the context of myocardial ischemic/hypoxic injury and protection. Western blotting and fluorescence double labelling were used to investigate the relationship between energy metabolism and autophagy in EP-resistant myocardial injury caused by exhaustive exercise. Compared with those in the C group, the levels of myocardial ischemic/hypoxic injury were significantly increased in the EE group. Compared with those in the EE group, the levels of myocardial ischemic/hypoxic injury were significantly decreased in the EEP + EE and LEP + EE groups. Compared with that in the EE group, the level of GLUT4 in the sarcolemma was significantly increased, and the colocalization of GLUT4 with the sarcolemma was significantly increased in the EEP + EE and LEP + EE groups (P < 0.05). LC3-II and LC3-II/LC3-I levels of the EEP + EE group were significantly elevated compared with those in the EE group (P < 0.05). The levels of p62 were significantly decreased in the EEP + EE and LEP + EE groups compared with the EE group (P < 0.05). EP promotes GLUT4 translocation and induced autophagy to alleviate exhaustive exercise-induced myocardial ischemic/hypoxic injury.











Similar content being viewed by others
Data availability
The data used to support the fndings of this study are available from the corresponding author upon request.
Code availability
Not applicable.
Abbreviations
- AMPK:
-
AMP-activated protein kinase
- ANOVA:
-
One-way analysis of variance
- cTnI:
-
Cardiac troponin I
- ECG:
-
Electrocardiogram
- EP:
-
Exercise preconditioning
- EEP:
-
Early exercise preconditioning
- GLUT4:
-
Glucose transporter 4
- HBFP:
-
Hematoxylin basic fuchsin picric acid
- IEP:
-
Induction of exercise preconditioning
- IHA:
-
Ischemia hypoxia area
- IOD:
-
Integral optical density
- LEP:
-
Late exercise preconditioning
- LC3:
-
Microtubule-associated protein 1 light chain 3
- MOD:
-
Mean optical density
- PEP:
-
Protection of exercise preconditioning
References
Abel ED (2004) Glucose transport in the heart. Front Biosci 9:201–215
Apple FS, Jaffe AS, Collinson P et al (2015) IFCC educational materials on selected analytical and clinical applications of high sensitivity cardiac troponin assays. Clin Biochem 48:201–203
Aviv Y, Shaw J, Gang H et al (2011) Regulation of autophagy in the heart: “you only live twice.” Antioxid Redox Signal 14(11):2245–2250
Cho DK, Choi DH, Cho JY (2017) Effect of treadmill exercise on skeletal muscle autophagy in rats with obesity induced by a high-fat diet. J Exerc Nutr Biochem 21(3):26–34
Dai Y, Wang Z, Quan M et al (2018) Asiatic acid protests against myocardial ischemia/reperfusion injury via modulation of glycometabolism in rat cardiomyocyte. Drug Des Dev Ther 12:3573–3582
Domenech R, Macho P, Schwarze H et al (2002) Exercise induces early and late myocardial preconditioning in dogs. Cardiovasc Res 55(3):561–566
Gillen JB, Gibala MJ (2014) Is high-intensity interval training a time-efficient exercise strategy to improve health and fitness? Appl Physiol Nutr Metab 39(3):409–412
Gurusamy N, Lekli I, Gorbunov NV et al (2009) Cardioprotection by adaptation to ischaemia augments autophagy in association with BAG-1 protein. J Cell Mol Med 13(2):373–387
Gustafsson AB, Gottlieb RA (2008) Heart mitochondria: gates of life and death. Cardiovasc Res 77(2):334–343
Hao Z, Pan SS, Shen YJ et al (2014) Exercise preconditioning-induced early and late phase of cardioprotection is associated with protein kinase C epsilon translocation. Circ J 78(7):1636–1645
Hardie DG, Ross FA, Hawley SA (2012) AMPK: a nutrient and energy sensor that maintains energy homeostasis. Nat Rev Mol Cell Biol 13(4):251–262
He C, Bassik MC, Moresi V et al (2012) Exercise-induced BCL2-regulated autophagy is required for muscle glucose homeostasis. Nature 481(7382):511–515
Heather LC, Pates KM, Atherton HJ et al (2013) Differential translocation of the fatty acid transporter, FAT/CD36, and the glucose transporter, GLUT4, coordinates changes in cardiac substrate metabolism during ischemia and reperfusion. Circ Heart Fail 6(5):1058–1066
Huang Y, Liu HT, Yuan Y et al (2021a) Exercise preconditioning increases Beclin1 and induces autophagy to promote early myocardial protection via intermittent myocardial ischemia-hypoxia. Int Heart J 62(2):407–415
Huang Y, Pan SS, Guo YP et al (2021b) Comparison of myocardial ischemic/hypoxic staining techniques for evaluating the alleviation of exhaustive exercise-induced myocardial injury by exercise preconditioning. J Mol Histol 52(2):373–383
Jaswal JS, Keung W, Wang W et al (2011) Targeting fatty acid and carbohydrate oxidation—a novel therapeutic intervention in the ischemic and failing heart. Biochim Biophys Acta 1813(7):333–1350
Ji L, Zhang X, Liu W et al (2013) AMPK-regulated and Akt-dependent enhancement of glucose uptake is essential in ischemic preconditioning-alleviated reperfusion injury. PLoS ONE 8(7):e69910
Karlsen T, Aamot IL, Haykowsky M et al (2017) High intensity interval training for maximizing health outcomes. Prog Cardiovasc Dis 60(1):67–77
Lee Y, Lee HY, Gustafsson AB (2012) Regulation of autophagy by metabolic and stress signaling pathways in the heart. J Cardiovasc Pharmacol 60(2):118–124
Lee Y, Kwon I, Jang Y et al (2017) Potential signaling pathways of acute endurance exercise-induced cardiac autophagy and mitophagy and its possible role in cardioprotection. J Physiol Sci 67(6):639–654
Levine B, Klionsky DJ (2004) Development by self-digestion: molecular mechanisms and biological functions of autophagy. Dev Cell 6(4):463–477
Li JY, Pan SS, Wang JY et al (2019) Changes in autophagy levels in rat myocardium during exercise preconditioning-initiated cardioprotective effects. Int Heart J 60(2):419–428
Liu LX, Arany Z (2014) Maternal cardiac metabolism in pregnancy. Cardiovasc Res 101(4):545–553
Liu HT, Pan SS (2019) Late exercise preconditioning promotes autophagy against exhaustive exercise-induced myocardial injury through the activation of the AMPK-mTOR-ULK1 pathway. Biomed Res Int 2019:5697380
Lu J, Pan SS (2017) Elevated C-type natriuretic peptide elicits exercise preconditioning-induced cardioprotection against myocardial injury probably via the up-regulation of NPR-B. J Physiol Sci 67(4):475–487
Luiken JJ, Glatz JF, Neumann D (2015) Cardiac contraction-induced GLUT4 translocation requires dual signaling input. Trends Endocrinol Metab (TEM) 26(8):404–410
Maejima Y, Isobe M, Jn S (2016) Regulation of autophagy by Beclin 1 in the heart. J Mol Cell Cardiol 95:19–25
Mizushima N, Komatsu M (2011) Autophagy: renovation of cells and tissues. Cell 147(4):728–741
Ni HM, Williams JA, Jaeschke H et al (2013) Zonated induction of autophagy and mitochondrial spheroids limits acetaminophen-induced necrosis in the liver. Redox Biol 1:427–432
Parra VM, Macho P, Sanchez G et al (2015) Exercise preconditioning of myocardial infarct size in dogs is triggered by calcium. J Cardiovasc Pharmacol 65(3):276–281
Pehmøller C, Treebak JT, Birk JB et al (2009) Genetic disruption of AMPK signaling abolishes both contraction- and insulin-stimulated TBC1D1 phosphorylation and 14-3-3 binding in mouse skeletal muscle. Am J Physiol Endocrinol Metab 297(3):E665-675
Pohl C, Dikic I (2019) Cellular quality control by the ubiquitin-proteasome system and autophagy. Science 366(6467):818–822
Quindry JC, Franklin BA (2021) Exercise preconditioning as a cardioprotective phenotype. Am J Cardiol 148:8–15
Richter EA, Hargreaves M (2013) Exercise, GLUT4, and skeletal muscle glucose uptake. Physiol Rev 93(3):993–1017
Ruze A, Chen BD, Liu F et al (2019) Macrophage migration inhibitory factor plays an essential role in ischemic preconditioning-mediated cardioprotection. Clin Sci (London, England) 133(5):665–680
Sheena A, Mohan SS, Haridas NP et al (2011) Elucidation of the glucose transport pathway in glucose transporter 4 via steered molecular dynamics simulations. PLoS ONE 6(10):e25747
Shen YJ, Pan SS, Zhuang T et al (2011) Exercise preconditioning initiates late cardioprotection against isoproterenol-induced myocardial injury in rats independent of protein kinase C. J Physiol Sci 61(1):13–21
Shepherd RE, Gollnick PD (1976) Oxygen uptake of rats at different work intensities. Pflugers Arch 362(3):219–222
Shintani T, Klionsky DJ (2004) Autophagy in health and disease: a double-edged sword. Science (New York, NY) 306(5698):990–995
Smenes BT, Bækkerud FH, Slagsvold KH et al (2018) Acute exercise is not cardioprotective and may induce apoptotic signalling in heart surgery: a randomized controlled trial. Interact Cardiovasc Thorac Surg 27(1):95–101
Su Y, Wang Y, Xu P et al (2021) Study on the time-effectiveness of exercise preconditioning on heart protection in exhausted rats. Chin J Physiol 64(2):97–105
Thygesen K, Alpert JS, Jaffe AS et al (2018) Fourth universal definition of myocardial infarction. J Am Coll Cardiol 72(18):2231–2264
Till M, Kolter T, Eckel J (1997) Molecular mechanisms of contraction-induced translocation of GLUT4 in isolated cardiomyocytes. Am J Cardiol 80:85A-89A
Verboven M, Cuypers A, Deluyker D et al (2019) High intensity training improves cardiac function in healthy rats. Sci Rep 9(1):5612
von Lewinski D, Gasser R, Rainer PP et al (2010) Functional effects of glucose transporters in human ventricular myocardium. Eur J Heart Fail 12(2):106–113
Wan DF, Pan SS, Tong YS et al (2021) Exercise preconditioning promotes autophagy to cooperate for cardioprotection by increasing LC3 lipidation-associated proteins. Front Physiol 12:599892
Williams RP, Manou-Stathopoulou V, Redwood SR et al (2014) ‘Warm-up Angina’: harnessing the benefits of exercise and myocardial ischaemia. Heart (British Cardiac Society) 100(2):106–114
Wong AP, Niedzwiecki A, Rath M (2016) Myocardial energetics and the role of micronutrients in heart failure: a critical review. Am J Cardiovasc Dis 6(3):81–92
Yamashita N, Hoshida S, Otsu K et al (1999) Exercise provides direct biphasic cardioprotection via manganese superoxide dismutase activation. J Exp Med 189(11):1699–1706
Yuan Y, Pan SS (2018) Parkin mediates mitophagy to participate in cardioprotection induced by late exercise preconditioning but Bnip3 does not. J Cardiovasc Pharmacol 71(5):303–316
Yuan JQ, Yuan Y, Pan SS et al (2020) Altered expression levels of autophagy-associated proteins during exercise preconditioning indicate the involvement of autophagy in cardioprotection against exercise-induced myocardial injury. J Physiol Sci 70(1):10
Funding
This work was supported by National Natural Science Foundation of China (No. 31471136).
Author information
Authors and Affiliations
Contributions
S-SP received National Natural Science Foundation of China and designed the study. YPG conducted experiments, performed the data analysis and drafted the manuscript. T-RC, YH, DFW and YST conducted experiments. Authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflicts of interest.
Ethical approval
All animal experiments were performed according to the Laboratory Animal Guideline for Ethical Review of Animal Welfare (GB/T 35892-2018, China) and approved by the Ethics Committee of Science Research at Shanghai University of Sport, China.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Guo, YP., Pan, SS., Chen, TR. et al. Exercise preconditioning promotes myocardial GLUT4 translocation and induces autophagy to alleviate exhaustive exercise-induced myocardial injury in rats. J Mol Histol 54, 453–472 (2023). https://doi.org/10.1007/s10735-023-10152-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10735-023-10152-7