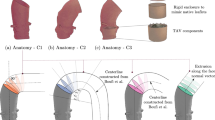
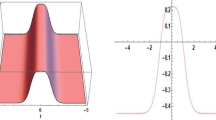
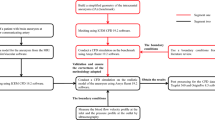
Abstract
The impact of the distribution in space of the inlet velocity in the numerical simulations of the hemodynamics in the thoracic aorta is systematically investigated. A real healthy aorta geometry, for which in-vivo measurements are available, is considered. The distribution is modeled through a truncated cone shape, which is a suitable approximation of the real one downstream of a trileaflet aortic valve during the systolic part of the cardiac cycle. The ratio between the upper and the lower base of the truncated cone and the position of the center of the upper base are selected as uncertain parameters. A stochastic approach is chosen, based on the generalized Polynomial Chaos expansion, to obtain accurate response surfaces of the quantities of interest in the parameter space. The selected parameters influence the velocity distribution in the ascending aorta. Consequently, effects on the wall shear stress are observed, confirming the need to use patient-specific inlet conditions if interested in the hemodynamics of this region. The surface base ratio is globally the most important parameter. Conversely, the impact on the velocity and wall shear stress in the aortic arch and descending aorta is almost negligible.








Similar content being viewed by others
References
Capellini, K., E. Vignali, E. Costa, E. Gasparotti, M. E. Biancolini, L. Landini, V. Positano, and S. Celi. Computational fluid dynamic study for aTAA hemodynamics: an integrated image-based and radial basis functions mesh morphing approach. J. Biomech. Eng. 140(11):111007, 2018.
Capellini, K., E. Gasparotti, U. Cella, E. Costa, B. M. Fanni, C. Groth, S. Porziani, M. E. Biancolini, and S. Celi. A novel formulation for the study of the ascending aortic fluid dynamics with in vivo data. Med. Eng. Phys. 91:68–78, 2021.
Gallo, D., G. De Santis, F. Negri, D. Tresoldi, R. Ponzini, D. Massai, M. A. Deriu, P. Segers, B. Verhegghe, G. Rizzo, and U. Morbiducci. On the use of in vivo measured flow rates as boundary conditions for image-based hemodynamic models of the human aorta: Implications for indicators of abnormal flow. Ann. Biomed. Eng. 40(3):729–741, 2012.
Morbiducci, U., R. Ponzini, D. Gallo, C. Bignardi, and G. Rizzo. Inflow boundary conditions for image-based computational hemodynamics: impact of idealized versus measured velocity profiles in the human aorta. J. Biomech. 46(1):102–109, 2013.
Boccadifuoco, A., A. Mariotti, S. Celi, N. Martini, and M. V. Salvetti. Impact of uncertainties in outflow boundary conditions on the predictions of hemodynamic simulations of ascending thoracic aortic aneurysms. Comput. Fluids. 165:96–115, 2018.
Boccadifuoco, A., A. Mariotti, K. Capellini, S. Celi, and M. V. Salvetti. Validation of numerical simulations of thoracic aorta hemodynamics: comparison with In Vivo measurements and stochastic sensitivity analysis. Cardiovasc. Eng. Technol. 9(4):688–706, 2018.
Antonuccio, M. N., A. Mariotti, S. Celi, M. V. Salvetti: Effects of the distribution in space of the velocity-inlet condition in hemodynamic simulations of the thoracic aorta. In: Lecture Notes in Computer Science (Including Subseries Lecture Notes in Artificial Intelligence and Lecture Notes in Bioinformatics), vol. 12108, pp. 63– 74 (2020)
Antonuccio, M. N., A. Mariotti, B. M. Fanni, K. Capellini, C. Capelli, E. Sauvage, and S. Celi. Effects of uncertainty of outlet boundary conditions in a patient-specific case of aortic coarctation. Ann. Biomed. Eng. 49(12):3494–3507, 2021. https://doi.org/10.1007/s10439-021-02841-9.
Mariotti, A., A. Boccadifuoco, S. Celi, and M. V. Salvetti. Hemodynamics and stresses in numerical simulations of the thoracic aorta: stochastic sensitivity analysis to inlet flow-rate waveform. Comput. Fluids. 230:105123, 2021.
Youssefi, P., A. Gomez, C. Arthurs, R. Sharma, M. Jahangiri, and C. A. Figueroa. Impact of patient-specific inflow velocity profile on hemodynamics of the thoracic aorta. J. Biomech. Eng. 140(1):1011002, 2018.
San, O., and A. E. Staples. An improved model for reduced-order physiological fluid flows. J. Mech. Med. Biol. 12(3):1250052, 2012.
Haugen, B. O., S. Berg, K. M. Brecke, H. Torp, S. A. Slørdahl, T. Skjærpe, and S. O. Samstad. Blood flow velocity profiles in the aortic annulus: a 3-dimensional freehand color flow doppler imaging study. J. Am. Soc. Echocardiogr. 15(4):328–333, 2002.
Sigovan, M., P. Dyverfeldt, J. Wrenn, E. E. Tseng, D. Saloner, and M. D. Hope. Extended 3D approach for quantification of abnormal ascending aortic flow. Magn. Reson. Imaging. 33:695–700, 2015.
Xu, L., L. Yin, Y. Liu, and F. Liang. A computational study on the influence of aortic valve disease on hemodynamics in dilated aorta. Math. Biosci. Eng. 17(1):606–626, 2020.
Chandra, S., S. S. Raut, A. Jana, R. W. Biederman, M. Doyle, S. C. Muluk, and E. A. Finol. Fluid-structure interaction modeling of abdominal aortic aneurysms: the impact of patient-specific inflow conditions and fluid/solid coupling. ASME J. Biomech. Eng. 135(8):081001, 2013.
Lodi Rizzini, M., D. Gallo, G. De Nisco, F. D’Ascenzo, C. Chiastra, P. P. Bocchino, F. Piroli, G. M. De Ferrari, and U. Morbiducci. Does the inflow velocity profile influence physiologically relevant flow patterns in computational hemodynamic models of left anterior descending coronary artery? Med. Eng. Phys. 82:58–69, 2020.
Madhavan, S., and E. M. C. Kemmerling. The effect of inlet and outlet boundary conditions in image-based CFD modeling of aortic flow. BioMed. Eng. OnLine. 17:66, 2018.
Armour, C. H., B. Guo, S. Pirola, S. Saitta, Y. Liu, Z. Dong, and X. Y. Xu. The influence of inlet velocity profile on predicted flow in type b aortic dissection. Biomech. Model. Mechanobiol. 20(2):481–490, 2021.
Updegrove, A., N. M. Wilson, J. Merkow, H. Lan, A. L. Marsden, and S. C. Shadden. SimVascular: an open source pipeline for cardiovascular simulation. Ann. Biomed. Eng. 45:525–541, 2016.
Masri, A., L. G. Svensson, B. P. Griffin, and M. Y. Desai. Contemporary natural history of bicuspid aortic valve disease: a systematic review. Heart. 103(17):1323–1330, 2017.
Xu, L., T. Yang, L. Yin, Y. Kong, Y. Vassilevski, and F. Liang. Numerical simulation of blood flow in aorta with dilation: a comparison between laminar and LES modeling methods. CMES Comput. Model. Eng. Sci. 124(2):509–526, 2020.
Whiting, C. H., and K. E. Jansen. A stabilized finite element method for the incompressible Navier-Stokes equations using a hierarchical basis. Int. J. Numer. Methods Fluids. 35(1):93–116, 2001.
Esmaily Moghadam, M., Y. Bazilevs, T.-Y. Hsia, I. E. Vignon-Clementel, and A. L. Marsden. A comparison of outlet boundary treatments for prevention of backflow divergence with relevance to blood flow simulations. Comput. Mech. 48(3):277–291, 2011.
Jansen, K. E., C. H. Whiting, and G. M. Hulbert. A generalized-\(\alpha\) method for integrating the filtered Navier-Stokes equations with a stabilized finite element method. Comput. Methods Appl. Mech. Eng. 190(3–4):305–319, 2000.
Arbia, G., I. E. Vignon-Clementel, T.-Y. Hsia, and J.-F. Gerbeau. Modified Navier-Stokes equations for the outflow boundary conditions in hemodynamics. Eur. J. Mech. B-Fluids. 60:175–188, 2016.
Garcia, J., A. J. Barker, and M. Markl. The role of imaging of flow patterns by 4D flow MRI in aortic stenosis. JACC: Cardiovasc. Imaging. 12(2):252–266, 2019.
Saikrishnan, N., L. Mirabella, and A. P. Yoganathan. Bicuspid aortic valves are associated with increased wall and turbulence shear stress levels compared to trileaflet aortic valves. Biomech Model Mechanobiol. 14(3):577–588, 2015.
Jayendiran, R., F. Condemi, S. Campisi, M. Viallon, P. Croisille, and S. Avril. Computational prediction of hemodynamical and biomechanical alterations induced by aneurysm dilatation in patient-specific ascending thoracic aortas. Int. J. Numer. Methods Biol. 36(6):3326, 2020.
Hoeijmakers, M., I. Waechter-Stehle, J. Weese, and F. Van de Vosse. Combining statistical shape modeling, CFD, and meta-modeling to approximate the patient-specific pressure-drop across the aortic valve in real-time. Int. J. Numer. Methods Biol. 36(10):3387, 2020.
Liu, J., J. A. Shar, and P. Sucosky. Wall shear stress directional abnormalities in BAV aortas: toward a new hemodynamic predictor of aortopathy? Front. Physiol. 9:993, 2018.
Lenz, A., J. Petersen, C. Riedel, J. M. Weinrich, H. Kooijman, B. P. Schoennagel, G. Adam, Y. von Kodolitsch, H. Reichenspurner, E. Girdauskas, et al. 4d flow cardiovascular magnetic resonance for monitoring of aortic valve repair in bicuspid aortic valve disease. J. Cardiovasc. Magn. Reson. 22:1–10, 2020.
Qin, J. J., P. Obeidy, M. Gok, A. Gholipour, and S. M. Grieve. 4d-flow MRI derived wall shear stress for the risk stratification of bicuspid aortic valve aortopathy: a systematic review. Front. Cardiovasc. Med. 9:1075833, 2022.
Lorenz, R., J. Bock, A. Barker, F. von Knobelsdorff-Brenkenhoff, W. Wallis, J. Korvink, M. Bissell, J. Schulz-Menger, and M. Markl. 4d flow magnetic resonance imaging in bicuspid aortic valve disease demonstrates altered distribution of aortic blood flow helicity. Magn. Resonan. Med. 71(4):1542–1553, 2014.
Mahadevia, R., A. J. Barker, S. Schnell, P. Entezari, P. Kansal, P. W. Fedak, S. C. Malaisrie, P. McCarthy, J. Collins, J. Carr, et al. Bicuspid aortic cusp fusion morphology alters aortic three-dimensional outflow patterns, wall shear stress, and expression of aortopathy. Circulation. 129(6):673–682, 2014.
Kalinowski, M. E., M. Szulik, S. Pawlak, B. Rybus-Kalinowska, M. Zembala, Z. Kalarus, and T. Kukulski. The impact of the aortic cusps fusion pattern and valve disease severity on the aortic wall mechanics in patients with bicuspid aortic valve. Int. J. Cardiovasc. Imaging. 36:1429–1436, 2020.
Vignali, E., E. Gasparotti, S. Celi, and S. Avril. Fully-coupled FSI computational analyses in the ascending thoracic aorta using patient-specific conditions and anisotropic material properties. Front. Physiol. 12:1740, 2021.
Calò, K., K. Capellini, G. De Nisco, V. Mazzi, E. Gasparotti, D. Gallo, S. Celi, and U. Morbiducci. Impact of wall displacements on the large-scale flow coherence in ascending aorta. J. Biomech. 154:111620, 2023.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest regarding the content of this article.
Ethical Approval
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1975, as revised in 2000. Informed consent was obtained from all patients for being included in the study.
Additional information
Associate Editor Sarah Vigmostad, Ph.D oversaw review of this article.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Mariotti, A., Celi, S., Antonuccio, M.N. et al. Impact of the Spatial Velocity Inlet Distribution on the Hemodynamics of the Thoracic Aorta. Cardiovasc Eng Tech 14, 713–725 (2023). https://doi.org/10.1007/s13239-023-00682-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s13239-023-00682-2