Abstract
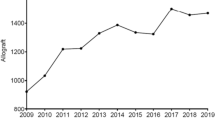
Musculoskeletal allografts represent an important practice in orthopedic surgeries and the demand for them has been growing. For this reason, in order to reduce clinical risk and to more efficiently manage the increase of allograft usage and also to optimize timing of the surgeries, the thawing and washing processes with aseptic technique were centralized in the department of Hospital Pharmacy. This study describe the design and execution of an adapted Media Fill Test (MFT) to demonstrate aseptic thawing and washing of allografts. For this specific and innovative setting, to better simulate the actual processing steps, a surrogate system was developed to simulate the tendon allograft. The aseptic technique of four operators was assessed and an initial performance validation and the first revalidation were described. All MFT were completed successfully, with no observation of turbidity. The readapted MFT shown in this study can provide insight into this innovative and growing field to other health professionals who want to implement this service.



Similar content being viewed by others
Data Availability
Data sharing not applicable to this article as no datasets were generated or analyzed during the current study.
References
Centers for Disease Control and Prevention (CDC) (2002) Update: allograft-associated bacterial infections–United States. MMWR Morb Mortal Wkly Rep 51(10):207–210
DeDino F, Kim V, DiNello D, Whitmer S, Adamo M, Starkey D, Evans K, Walker M, Thut D, Shields S, Van’t Hof W (2020) A cell therapy media fill protocol for validation of aseptic processing of cord blood. Cell Gene Therapy Insights 6:1529–1537
European Directorate for the Quality of Medicines & HealthCare of the Council of Europe (EDQM) (2019) The Guide to the quality and safety of tissues and cells for human application. 4th edn. ISBN 978-92-871-8945-5
European Commission (2022) The Rules Governing Medicinal Products in the European Union Volume 4 EU Guidelines for Good Manufacturing Practice for Medicinal Products for Human and Veterinary Use. https://health.ec.europa.eu/system/files/2022-08/20220825_gmp-an1_en_0.pdf. Accessed 23 January 2023
European Commission (2006) Commission Directive 2006/86/EC of 24 October 2006 implementing Directive 2004/23/EC of the European Parliament and of the Council as regards traceability requirements, notification of serious adverse reactions and events and certain technical requirements for the coding, processing, preservation, storage and distribution of human tissues and cells. http://data.europa.eu/eli/dir/2006/86/oj. Accessed 23 January 2023
Fu SH, Liu JY, Huang CC, Lin Fl, Yang RS, Hou CH (2017) Quality control processes in allografting: a twenty-year retrospective review of a hospital-based bone bank in Taiwan. PLoS ONE 12(10):e0184809. https://doi.org/10.1371/journal.pone.0184809
ISO Technical Committee (2015b) ISO 14644-2:2015, Cleanrooms and associated controlled environments: part 2: monitoring to provide evidence of cleanroom performance related to air cleanliness by particle concentration. https://www.iso.org/obp/ui/#iso:std:iso:14644:-1:ed-2:v1:en. Accessed 23 January 2023
ISO Technical Committee (2015a) ISO 14644-1:2015, Cleanrooms and associated controlled environments: part 1: classification of air cleanliness by particle concentration. https://www.iso.org/obp/ui/#iso:std:iso:14644:-1:ed-2:v1:en. Accessed 23 January 2023
Khuu HM, Patel N, Carter CS, Murray PR, Read EJ (2006) Sterility testing of cell therapy products: parallel comparison of automated methods with a CFR-compliant method. Transfusion 46(12):2071–2082. https://doi.org/10.1111/j.1537-2995.2006.01041.x
Lomas R, Chandrasekar A, Board TN (2013) Bone allograft in the U.K.: perceptions and realities. Hip Int 23(5):427–433. https://doi.org/10.5301/hipint.5000018
Lotfi R, Rojewski MT, Zeplin PH, Funk W, Pullig O, Nöth U, Schrezenmeier H (2020) Validation of microbiological testing of cellular medicinal products containing antibiotics. Transfus Med Hemother 47(2):144–151. https://doi.org/10.1159/000501284
Ossato A, Mezzadrelli V, Montagner G, Trojan D, Giovagnoni G, Giannini M, Trabucchi C, Angelini C, Realdon F, Cipriano L, Realdon N, Zuppini T, Tessari R (2023) A retrospective hospital benefit and cost analysis of the management of human tissues for orthopaedic allografts. Eur J Hosp Pharm. https://doi.org/10.1136/ejhpharm-2023-003744
Robertson A, Nutton RW, Keating JF (2006) Current trends in the use of tendon allografts in orthopaedic surgery. J Bone Joint Surg Br 88(8):988–992. https://doi.org/10.1302/0301-620X.88B8.17555
Secretariat of the Pharmaceutical Inspection Convention (PIC/S). Recommendation on the validation of aseptic processes PI 007 – 6 (2011) https://www.gmpcompliance.org/files/guidemgr/PI007-6.Recommendation-on-Aseptic-Processes.pdf. Accessed 24 July 2023
Serra M, Roseti L, Bassi A (2015) Media fill for validation of a good manufacturing practice-compliant cell production process. Methods Mol Biol 1283:161–169. https://doi.org/10.1007/7651_2014_104
Sigward E, Fourgeaud M, Vazquez R, Guerrault-Moro M-N, Brossard D, Crauste-Manciet S (2012) Aseptic simulation test challenged with microorganisms for validation of operators for aseptic compounding in hospital pharmacies. Am J Health-Sys Pharm 69(15):1218–1224. https://doi.org/10.2146/ajhp110295
UNE EN 17141:2021 (2021) Cleanrooms and associated controlled environments: biocontamination control is classified in these ICS categories. https://www.en-standard.eu/une-en-17141-2021-cleanrooms-and-associated-controlled-environments-biocontamination-control/. Accessed 23 January 2023
UNI EN ISO 13408-1:2015 (2015) Aseptic processing of health care products is classified in these ICS categories. https://www.en-standard.eu/bs-en-iso-13408-1-2015-aseptic-processing-of-health-care-products-general-requirements/. Accessed 23 January 2023
Vaishnav S, Vangsness CT Jr, Dellamaggiora R (2009) New techniques in allograft tissue processing. Clin Sports Med 28(1):127–141. https://doi.org/10.1016/j.csm.2008.08.002
Acknowledgements
None.
Funding
The authors did not receive support from any organization for the submitted work.
Author information
Authors and Affiliations
Contributions
OA and RT contributed to the study concepts, study design; GG, GM, SAF, RF and RT contributed to the experimental studies and data acquisition; GC and SM contributed to the data analysis; OA, GC, RT contributed to the manuscript preparation and RN and TZ contributed to the manuscript editing and review; All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
The authors declare that they have no known competing financial.
interests or personal relationships that could have influenced the work reported in this paper.
Conflict of interest
The authors have no conflict of interest to declare.
Ethical approval
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ossato, A., Gasperoni, C., Stella, M. et al. Validation of orthopedic allograft preparation process: a new application of media fill test. Cell Tissue Bank (2023). https://doi.org/10.1007/s10561-023-10107-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10561-023-10107-y