Abstract
Background
Current evidence shows that ML385, a new type of nuclear factor erythroid 2-related factor 2 inhibitor, exerts a good inhibitory effect on tumors. However, whether ML385 can regulate the biological behavior of thyroid cancer remains puzzling.
Objectives
We aimed to observe the regulation of ML385 on biological characteristics such as proliferation, apoptosis, migration, and invasion of thyroid tumor cells in vitro and clarify the molecular mechanism of regulating glucose metabolism of tumor cells to lay a foundation for ML385 as a therapeutic tumor drug.
Results
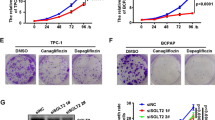
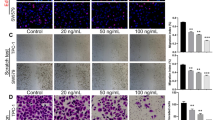
ML385 effectively reduced the viability, proliferation, invasion, migration, glucose consumption, lactate production, adenosine triphosphate level, and extracellular acidification rate of TPC-1 cells and concentration-dependently promoted TPC-1 cell apoptosis, indicating that ML385 inhibited the biological behavior thyroid cancer cell and aerobic glycolysis in vitro. Moreover, the above cellular behaviors were not significantly altered when 2-DG was added to ML385 treatment, suggesting that the inhibition of glycolysis by 2-DG may partially block the effect of ML385 on thyroid cancer cells.
Conclusions
ML385 inhibits the biological behavior of thyroid cancer cells by impairing aerobic glycolysis.




Similar content being viewed by others
Data availability
The datasets generated during and/or analyzed during the current study are available from the corresponding author on reasonable request.
References
Abbaszadeh Z, Çeşmeli S, Biray AÇ (2020) Crucial players in glycolysis: cancer progress. Gene 726:144158
Bartman CR et al (2023) Slow TCA flux and ATP production in primary solid tumours but not metastases. Nature 614:349–357
Bikas A et al (2015) Glucose-deprivation increases thyroid cancer cells sensitivity to metformin. Endocr Relat Cancer 22:919–932
Cabanillas ME, Ryder M, Jimenez C (2019) Targeted therapy for advanced thyroid cancer: kinase inhibitors and beyond. Endocr Rev 40:1573–1604
Chelakkot C, Chelakkot VS, Shin Y, Song K (2023) Modulating glycolysis to improve cancer therapy. Int J Mol Sci. https://doi.org/10.3390/ijms24032606
Chen DW et al (2023) Thyroid cancer. Lancet 401:1531–1544
Cykowiak M, Krajka-Kuźniak V (2021) Role of Nrf2 in pancreatic cancer. Antioxidants (basel). https://doi.org/10.3390/antiox11010098
Dai YQ, Bai Y, Gu J, Fan BY (2022) Stanniocalcin1 knockdown induces ferroptosis and suppresses glycolysis in prostate cancer via the Nrf2 pathway. Neoplasma 69:1396–1405
Farkhondeh T et al (2021) Roles of Nrf2 in gastric cancer: targeting for therapeutic strategies. Molecules. https://doi.org/10.3390/molecules26113157
Ganapathy-Kanniappan S, Geschwind JF (2013) Tumor glycolysis as a target for cancer therapy: progress and prospects. Mol Cancer 12:152
Gong Z et al (2021) The knockdown of Nrf2 suppressed tumor growth and increased the sensitivity to lenvatinib in anaplastic thyroid cancer. Oxid Med Cell Longev 2021:3900330
He F, Antonucci L, Karin M (2020) NRF2 as a regulator of cell metabolism and inflammation in cancer. Carcinogenesis 41:405–416
Huang J et al (2022) FTO suppresses glycolysis and growth of papillary thyroid cancer via decreasing stability of APOE mRNA in an N6-methyladenosine-dependent manner. J Exp Clin Cancer Res 41:42
Ji L et al (2023) The NRF2 antagonist ML385 inhibits PI3K-mTOR signaling and growth of lung squamous cell carcinoma cells. Cancer Med 12:5688–5702
Koppenol WH, Bounds PL, Dang CV (2011) Otto Warburg’s contributions to current concepts of cancer metabolism. Nat Rev Cancer 11:325–337
Laha D, Nilubol N, Boufraqech M (2020) New therapies for advanced thyroid cancer. Front Endocrinol (lausanne) 11:82
Li J, Wang D, Liu Y, Zhou Y (2022a) Role of NRF2 in colorectal cancer prevention and treatment. Technol Cancer Res Treat 21:15330338221105736
Li X et al (2022b) Lactate metabolism in human health and disease. Signal Transduct Target Ther 7:305
Lin Y et al (2020) Gallic acid alleviates gouty arthritis by inhibiting NLRP3 inflammasome activation and pyroptosis through enhancing Nrf2 signaling. Front Immunol 11:580593
Luo J et al (2022) Anti-anaplastic thyroid cancer (ATC) effects and mechanisms of PLX3397 (Pexidartinib), a multi-targeted tyrosine kinase inhibitor (TKI). Cancers (basel). https://doi.org/10.3390/cancers15010172
Menegon S, Columbano A, Giordano S (2016) The dual roles of NRF2 in cancer. Trends Mol Med 22:578–593
Rabinowitz JD, Enerbäck S (2020) Lactate: the ugly duckling of energy metabolism. Nat Metab 2:566–571
Sánchez-Ortega M, Carrera AC, Garrido A (2021) Role of NRF2 in lung cancer. Cells. https://doi.org/10.3390/cells10081879
Schmidlin CJ et al (2021) The intricacies of NRF2 regulation in cancer. Semin Cancer Biol 76:110–119
Singh A et al (2016) Small molecule inhibitor of NRF2 selectively intervenes therapeutic resistance in KEAP1-deficient NSCLC tumors. ACS Chem Biol 11:3214–3225
Tuttle RM (2018) Controversial issues in thyroid cancer management. J Nucl Med 59:1187–1194
Wang Z et al (2022) Dexmedetomidine attenuates myocardial ischemia/reperfusion-induced ferroptosis via AMPK/GSK-3β/Nrf2 axis. Biomed Pharmacother 154:113572
Zhang D et al (2014) 2-Deoxy-D-glucose targeting of glucose metabolism in cancer cells as a potential therapy. Cancer Lett 355:176–183
Zhang HS et al (2019a) Nrf2 promotes breast cancer cell migration via up-regulation of G6PD/HIF-1α/Notch1 axis. J Cell Mol Med 23:3451–3463
Zhang T et al (2019b) Targeting the ROS/PI3K/AKT/HIF-1α/HK2 axis of breast cancer cells: combined administration of Polydatin and 2-Deoxy-d-glucose. J Cell Mol Med 23:3711–3723
Zhang Y, Xin Z, Dong B, Xue W (2022) Combination of the NRF2 inhibitor and autophagy inhibitor significantly inhibited tumorigenicity of castration-resistant prostate cancer. Comput Math Methods Med 2022:4182401
Acknowledgements
The authors have no acknowledgments.
Funding
No funding was received.
Author information
Authors and Affiliations
Contributions
WZ and HY conceived the project and designed the experiments. WZ, HY, HL, and HH performed the experiments and analyzed the data. WZ wrote and revised the manuscript. All the authors have accepted responsibility for the entire content of this manuscript and approved its submission.
Corresponding author
Ethics declarations
Conflict of interest
Wentian Zheng declares that they have no conflict of interest. Huan Yang declares that they have no conflict of interest. Hehua Lin declares that they have no conflict of interest. Hanxing Huang declares that they have no conflict of interest.
Ethical approval
This article contains no studies with human participants or animals performed by authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zheng, W., Yang, H., Lin, H. et al. ML385 suppresses the proliferation, migration, and invasion of thyroid carcinoma cells by impairing aerobic glycolysis. Mol. Cell. Toxicol. (2023). https://doi.org/10.1007/s13273-023-00395-6
Accepted:
Published:
DOI: https://doi.org/10.1007/s13273-023-00395-6