Abstract
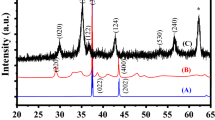
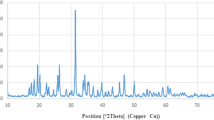
TiO2 nanoparticles are widely used in industry, pharmacology, and medicine. Recently, it has been proposed to use TiO2 nanoparticles for targeted delivery of chemotherapeutic drugs, which can lead to an increase in the local concentration of TiO2 and influence the processes occurring in the tumor, including entosis (the invasion of one tumor cell into another). In this work, we studied the effect of TiO2 nanoparticles (particles of the mineral anatase less than 25 nm in size and particles consisting of a mixture of anatase and rutile less than 75 nm in size at a concentration of 1, 10, or 100 μg/mL for 72 h) on the process of entosis in human breast MCF-7 adenocarcinoma cell culture. Cultivation of cells in the presence of different types of TiO2 nanoparticles slows down proliferation and causes a decrease in the number of entoses. Elemental analysis by analytical electron microscopy revealed the presence of TiO2 in the endosomal compartment, in the cytosol and in the extracellular space. Immunochemical staining showed that both types of TiO2 nanoparticles at a concentration of 10 μg/mL disrupt the formation of adhesive contacts, including those in the early stages of entosis. The impact of anatase particles induces the translocation of the p53 protein into the cell nuclei. In general, the TiO2 nanoparticles used in the work inhibit entosis in MCF-7 culture cells, disrupting adhesive contacts and interfering with the process of invasion. However, it can be assumed that disruption of adhesive contacts can enhance the mobility of tumor cells and metastasis.








Similar content being viewed by others
REFERENCES
Biola-Clier, M., Gaillard, J.-C., Rabilloud, T., Armengaud, J., and Carriere, M., Titanium dioxide nanoparticles alter the cellular phosphoproteome in A549 cells, Nanomaterials, 2020, vol. 10, p. 185. https://doi.org/10.3390/nano10020185
Chumakov, P.M., Versatile functions of p53 protein in multicellular organisms, Biochemistry (Moscow), 2007, vol. 72, no. 13, p. 1399. https://doi.org/10.1134/s0006297907130019
Ding, L., Li, J., Huang, R., Liu, Z., Li, C., Yao, S., Wang, J., Qi, D., Li, N., and Pi, J., Salvianolic acid B protects against myocardial damage caused by nanocarrier TiO2; and synergistic anti-breast carcinoma effect with curcumin via codelivery system of folic acid-targeted and polyethylene glycol-modified TiO2 nanoparticles, Int. J. Nanomed., 2016, vol. 11, p. 5709. https://doi.org/10.2147/IJN.S107767
Durgan, J., Tseng, Y.Y., Hamann, J.C., Domart, M.C., Collinson, L., Hall, A., Overholtzer, M., and Florey, O., Mitosis can drive cell cannibalism through entosis, Elife, 2017, vol. 6, p. e27134. https://doi.org/10.7554/eLife.27134
Fage, S.W., Muris, J., Jakobsen, S.S., and Thyssen, J.P., Titanium: a review on exposure, release, penetration, allergy, epidemiology, and clinical reactivity, Contact Dermatitis, 2016, vol. 74, p. 323. https://doi.org/10.1111/cod.12565
Frohlich, E., Cellular targets and mechanisms in the cytotoxic action of non-biodegradable engineered nanoparticles, Curr. Drug Metab., 2013, vol. 14, no. 9, p. 976. https://doi.org/10.2174/1389200211314090004
Garanina, A.S., Kisurina-Evgenieva, O.P., Erokhina, M.V., Smirnova, E.A., Factor, V.M., and Onishchenko, G.E., Consecutive entosis stages in human substrate-dependent cultured cells, Sci. Rep., 2017, vol. 7, p. 12555. https://doi.org/10.1038/s41598-017-12867-6
Hanot-Roy, M., Tubeuf, E., Guilbert, A., Bado-Nilles, A., Vigneron, P., Trouiller, B., Braun, A., and Lacroix, G., Oxidative stress pathways involved in cytotoxicity and genotoxicity of titanium dioxide (TiO2) nanoparticles on cells constitutive of alveolo-capillary barrier in vitro, Toxicol. In Vitro, 2016, vol. 33, p. 125. https://doi.org/10.1016/j.tiv.2016.01.013
Hurum, D.C., Gray, K.A., Rajh, T., and Thurnauer, M.C., Recombination pathways in the degussa P25 formulation of TiO2: surface versus lattice mechanisms, J. Phys. Chem., 2005, vol. 109, p. 977. https://doi.org/10.1021/jp045395d
Krajcovic, M., Johnson, N.B., Sun, Q., Normand, G., Hoover, N., Yao, E., Richardson, A.L., King, R.W., Cibas, E.S., Schnitt, S.J., Brugge, J.S., and Overholtzer, M., A non-genetic route to aneuploidy in human cancers, Nat. Cell Biol., 2011, vol. 13, p. 324. https://doi.org/10.1038/ncb2174
Krishna, S. and Overholtzer, M., Mechanisms and consequences of entosis, Cell Mol. Life Sci., 2016, vol. 73, p. 2379. https://doi.org/10.1007/s00018-016-2207-0
Kisurina-Evgenieva, O.P., Khashba, L.A., Ma-michev, I.A., Savitskaya, M.A., and Onishchenko, G.E., Entosis and cell cycle in tumor cell culture, Cell Tissue Biol., 2019, vol. 13, p. 8. https://doi.org/10.1134/S1990519X19010073
Lagopati, N., Tsilibary, E.-P., Falaras, P., Papazafiri, P., Pavlatou, E.A., Kotsopoulou, E., and Kitsiou1, P., Effect of nanostructured TiO2 crystal phase on photoinduced apoptosis of breast cancer epithelial cells, Int. J. Nanomed., 2014, vol. 9, p. 3219. https://doi.org/10.2147/IJN.S62972
Liang, J., Niu, Z., Zhang, B., Yu, X., Zheng, Y., Wang, C., Ren, H., Wang, M., Ruan, B., Qin, H., Zhang, X., Gu, S., Sai, X., Tai, Y., Gao, L., et al., p53-dependent elimination of aneuploid mitotic offspring by entosis, Cell Death. Differ., 2021, vol. 28, p. 799. https://doi.org/10.1038/s41418-020-00645-3
Overholtzer, M. and Brugge, J.S., The cell biology of cell-in-cell structures, Nat. Rev. Mol. Cell Biol., 2008, vol. 9, p. 796. https://doi.org/10.1038/nrm2504
Overholtzer, M., Mailleux, A.A., Mouneimne, G., Normand, G., Schnitt, S.J., King, R.W., Cibas, E.S., and Brugge, J.S., A nonapoptotic cell death process, entosis, that occurs by cell-in-cell invasion, Cell, 2007, vol. 131, p. 966. https://doi.org/10.1016/j.cell.2007.10.040
Rossi, E.M., Pylkkänen, L., Koivisto, A.J., Vippola, M., Jensen, K.A., Miettinen, M., Sirola, K., Nykäsenoja, H., Karisola, P., Stjernvall, T., Vanhala, E., Kiilunen, M., Pasanen, P., Mäkinen, M., Hämeri, K., et al., Airway exposure to silica-coated TiO2 nanoparticles induces pulmonary neutrophilia in mice, Toxicol. Sci., 2010, vol. 113, p. 422. https://doi.org/10.1093/toxsci/kfp254
Sayes, C.M., Wahi, R., Kurian, P.A., Liu, Y., West, J.L., Ausman, K.D., Warheit, D.B., and Colvin, V.L., Correlating nanoscale titania structure with toxicity: a cytotoxicity and inflammatory response study with human dermal fibroblasts and human lung epithelial cells, Toxicol. Sci., 2006, vol. 92, p. 174. https://doi.org/10.1093/TOXSCI/KFJ197
Sayes, C.M., Wahi, R., Kurian, P.A., Liu, Y., West, J.L., Ausman, K.D., Warheit, D.B., and Colvin, V.L., Correlating nanoscale titania structure with toxicity: a cytotoxicity and inflammatory response study with human dermal fibroblasts and human lung epithelial cells, Toxicol. Sci., 2006, vol. 92, p. 174. https://doi.org/10.1093/toxsci/kfj197
Setyawati, M.I., Tay, C.Y., Chia, S.L., Goh, S.L., Fang, W., Neo, M.J., Chong, H.C., Tan, S.M., Loo, S.C.J., Ng, K.W., Xie J.P., Ong, C.N., Tan, N.S., and Leong, D.T., Titanium dioxide nanomaterials cause endothelial cell leakiness by disrupting the homophilic interaction of VE–cadherin, Nat. Commun., 2013, vol. 4, p. 1673. https://doi.org/10.1038/ncomms2655
Sun, Q., Cibas, E.S., Huang, H., Hodgson, L., and Overholtzer, M., Induction of entosis by epithelial cadherin expression, Cell Res., 2014, vol. 24, p. 1288. https://doi.org/10.1038/cr.2014.137
Sund, J., Palomäki, J., Ahonen, N., Savolainen, K., Alenius, H, and Puustinen, A., Phagocytosis of nano-sized titanium dioxide triggers changes in protein acetylation, J. Proteomics, 2014, vol. 108, p. 469. https://doi.org/10.1016/j.jprot.2014.06.011
Sydor, M.J., Anderson, D.S., Steele, H.B.B., Ross, J.B.A., and Holian, A., Effects of titanium dioxide and zinc oxide nano-materials on lipid order in model membranes, Biochim. Biophys. Acta, Biomembr., 2020, vol. 1862, p. 183313. https://doi.org/10.1016/j.bbamem.2020.183313
Trang, N.T.K., Dong, V.P., and Hoon, Y., Cellular effects of ultraviolet-radiated reduced-titanium dioxide nanoparticles on human hypopharyngeal adenocarcinoma cells, J. Nanosci. Nanotechnol., 2021, vol. 21, p. 3656. https://doi.org/10.1166/jnn.2021.19172
Wang, J., Zhou, G., Chen, C., Yu, H., Wang, T., Ma, Y., Jia, C., Gao, Y., Li, B., Sun, J., Li, Y., Fang, J., Zhao, Y., and Chai, Z., Acute toxicity and biodistribution of different sized titanium dioxide particles in mice after oral administration, Toxicol. Lett., 2007, vol. 168, p. 176. https://doi.org/10.1016/j.toxlet.2006.12.001
Wang, Y., Cui, H., Zhou, J., Li, F., Wang, J., Chen, M., and Liu, Q., Cytotoxicity, DNA damage, and apoptosis induced by titanium dioxide nanoparticles in human non-small cell lung cancer A549 cells, Environ. Sci. Pollut. Res. Int., 2015, vol. 22, p. 5519. https://doi.org/10.1007/s11356-014-3717-7
Warheit, D.B., Webb, T.R., Sayes, C.M., Colvin, V.L., and Reed, K.L., Pulmonary instillation studies with nanoscale TiO2 rods and dots in rats: toxicity is not dependent upon particle size and surface area, Toxicol. Sci., 2006, vol. 91, p. 227. https://doi.org/10.1093/toxsci/kfj140
Zeng, L., Pan, Y., Tian, Y., Wang, X., Ren, W., Wang, S., Lu, G., and Wu, A., Doxorubicin-loaded NaYF 4: Yb/Tm–TiO 2 inorganic photosensitizers for NIR-triggered photodynamic therapy and enhanced chemotherapy in drug-resistant breast cancers, Biomaterials, 2015, vol. 57, p. 93. https://doi.org/10.1016/j.biomaterials.2015.04.006
Funding
The work was carried out at the expense of the budgetary funds of Moscow State University on the planned topic “Mechanisms of Cell Survival and Death” (121032300098-5).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The authors declare that they have no conflicts of interest. The work did not include experiments involving animals or human beings.
Rights and permissions
About this article
Cite this article
Kisurina-Evgenieva, O.P., Savitskaya, M.A., Smeshnova, D.S. et al. Titanium-Dioxide Nanoparticles Suppress Entosis in MCF-7 Human Breast Adenocarcinoma Culture. Cell Tiss. Biol. 17, 531–542 (2023). https://doi.org/10.1134/S1990519X23050073
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S1990519X23050073