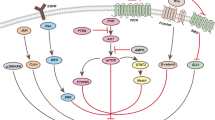
Abstract
Anthracycline antitumor agents, such as doxorubicin (DOX), are effective in the treatment of solid tumors and hematological malignancies, but anthracycline-induced cardiotoxicity (AIC) limits their application as chemotherapeutics. Dexrazoxane (DEX) has been adopted to prevent AIC. Using a chronic AIC mouse model, we demonstrated that DEX is insufficient to reverse DOX-induced cardiotoxicity. Although therapies targeting autophagy have been explored to prevent AIC, but whether novel autophagy inhibitors could alleviate or prevent AIC in clinically relevant models needs further investigation. Here, we show that genetic ablation of Atg7, a key regulator in the early phase of autophagy, protected mice against AIC. We further demonstrated that SAR405, a novel autophagy inhibitor, attenuated DOX-induced cytotoxicity. Intriguingly, the combination of DEX and SAR405 protected cells against DOX-induced cardiotoxicity in vivo. Using the cardiomyocyte cell lines AC16 and H9c2, we determined that autophagy was initiated during AIC. Our results suggest that inhibition of autophagy at its early phase with SAR405 combined with DEX represents an effective therapeutic strategy to prevent AIC.





Similar content being viewed by others
Data availability
The data that has been used is confidential. The data referred to in this study were obtained from the corresponding authors.
References
Armenian SH, Lacchetti C, Barac A, et al. Prevention and monitoring of cardiac dysfunction in survivors of adult cancers: American Society of Clinical Oncology clinical practice guideline. J Clin Oncol: Off J Am Soc Clin Oncol. 2017;35:893–911.
Asselin BL, Devidas M, Chen L, et al. Cardioprotection and Safety of dexrazoxane in patients treated for newly diagnosed T-cell acute lymphoblastic leukemia or advanced-stage lymphoblastic non-Hodgkin lymphoma: a report of the Children’s Oncology Group Randomized Trial Pediatric Oncology Group 9404. J Clin Oncol: Off J Am Soc Clin Oncol. 2016;34:854–62.
Cardinale D, Colombo A, Bacchiani G, et al. Early detection of anthracycline cardiotoxicity and improvement with heart failure therapy. Circulation. 2015;131:1981–8.
Catanzaro MP, Weiner A, Kaminaris A, et al. Doxorubicin-induced cardiomyocyte death is mediated by unchecked mitochondrial fission and mitophagy. FASEB J: Off Publ Fed Am Soc Exp Biol. 2019;33:11096–108.
Chang HM, Okwuosa TM, Scarabelli T, Moudgil R, Yeh ETH. Cardiovascular complications of cancer therapy: best practices in diagnosis, prevention, and management: part 2. J Am Coll Cardiol. 2017;70:2552–65.
Dalby KN, Tekedereli I, Lopez-Berestein G, Ozpolat B. Targeting the prodeath and prosurvival functions of autophagy as novel therapeutic strategies in cancer. Autophagy. 2010;6:322–9.
Davis JL. A practical system for narcotic control within the OR/PACU. J Post Anesth Nurs. 1989;4:32–5.
Gu J, Song ZP, Gui DM, Hu W, Chen YG, Zhang DD. Resveratrol attenuates doxorubicin-induced cardiomyocyte apoptosis in lymphoma nude mice by heme oxygenase-1 induction. Cardiovasc Toxicol. 2012;12:341–9.
Gu J, Hu W, Song ZP, Chen YG, Zhang DD, Wang CQ. Resveratrol-induced autophagy promotes survival and attenuates doxorubicin-induced cardiotoxicity. Int Immunopharmacol. 2016;32:1–7.
Gu J, Fan YQ, Zhang HL, et al. Resveratrol suppresses doxorubicin-induced cardiotoxicity by disrupting E2F1 mediated autophagy inhibition and apoptosis promotion. Biochem Pharmacol. 2018;150:202–13.
Levy JMM, Towers CG, Thorburn A. Targeting autophagy in cancer. Nat Rev Cancer. 2017;17:528–42.
Li DL, Wang ZV, Ding G, et al. Doxorubicin blocks cardiomyocyte autophagic flux by inhibiting lysosome acidification. Circulation. 2016;133:1668–87.
Li M, Sala V, De Santis MC, et al. Phosphoinositide 3-kinase gamma inhibition protects from anthracycline cardiotoxicity and reduces tumor growth. Circulation. 2018;138:696–711.
Lipshultz SE, Scully RE, Lipsitz SR, et al. Assessment of dexrazoxane as a cardioprotectant in doxorubicin-treated children with high-risk acute lymphoblastic leukaemia: long-term follow-up of a prospective, randomised, multicentre trial. Lancet Oncol. 2010;11:950–61.
Mariño G, Pietrocola F, Kong Y, et al. Dimethyl α-ketoglutarate inhibits maladaptive autophagy in pressure overload-induced cardiomyopathy. Autophagy. 2014;10:930–2.
Nakai A, Yamaguchi O, Takeda T, et al. The role of autophagy in cardiomyocytes in the basal state and in response to hemodynamic stress. Nat Med. 2007;13:619–24.
Pasquier B. SAR405, a PIK3C3/Vps34 inhibitor that prevents autophagy and synergizes with MTOR inhibition in tumor cells. Autophagy. 2015;11:725–6.
Ronan B, Flamand O, Vescovi L, et al. A highly potent and selective Vps34 inhibitor alters vesicle trafficking and autophagy. Nat Chem Biol. 2014;10:1013–9.
Shirakabe A, Zhai P, Ikeda Y, et al. Drp1-dependent mitochondrial autophagy plays a protective role against pressure overload-induced mitochondrial dysfunction and heart failure. Circulation. 2016;133:1249–63.
Song E, Da Eira D, Jani S, et al. Cardiac autophagy deficiency attenuates ANP production and disrupts myocardial-adipose cross talk, leading to increased fat accumulation and metabolic dysfunction. Diabetes. 2021;70:51–61.
Sun X, Meng H, Xiao J, Liu F, Du J, Zeng H. Pretreatment of 3-MA prevents doxorubicin-induced cardiotoxicity through inhibition of autophagy initiation. Toxicology. 2023;490:153512.
Tebbi CK, London WB, Friedman D, et al. Dexrazoxane-associated risk for acute myeloid leukemia/myelodysplastic syndrome and other secondary malignancies in pediatric Hodgkin’s disease. J Clin Oncol: Off J Am Soc Clin Oncol. 2007;25:493–500.
Valentim L, Laurence KM, Townsend PA, et al. Urocortin inhibits Beclin1-mediated autophagic cell death in cardiac myocytes exposed to ischaemia/reperfusion injury. J Mol Cell Cardiol. 2006;40:846–52.
Vejpongsa P, Yeh ET. Topoisomerase 2β: a promising molecular target for primary prevention of anthracycline-induced cardiotoxicity. Clin Pharmacol Ther. 2014;95:45–52.
Vrooman LM, Neuberg DS, Stevenson KE, et al. The low incidence of secondary acute myelogenous leukaemia in children and adolescents treated with dexrazoxane for acute lymphoblastic leukaemia: a report from the Dana-Farber Cancer Institute ALL Consortium. Eur J Cancer (Oxford, England: 1990). 2011;47:1373–9.
Wang Y, Lu X, Wang X, et al. atg7-based autophagy activation reverses doxorubicin-induced cardiotoxicity. Circ Res. 2021;129:e166–82.
Xie Z, Klionsky DJ. Autophagosome formation: core machinery and adaptations. Nat Cell Biol. 2007;9:1102–9.
Xu Z, Jin Y, Gao Z, et al. Autophagic degradation of CCN2 (cellular communication network factor 2) causes cardiotoxicity of sunitinib. Autophagy. 2022;18:1152–73.
Zhang S, Liu X, Bawa-Khalfe T, et al. Identification of the molecular basis of doxorubicin-induced cardiotoxicity. Nat Med. 2012;18:1639–42.
Acknowledgements
We are grateful to Dr. Yangqiu Li, Dr. Aibin He, and Dr. Xufeng Qi for critically reading the manuscript.
Funding
This work was supported by the National Natural Science Foundation of China (Grant No. 81970143 and No. 82270167), the Talent Young Program of Guangdong Province (2021B1515020017), the Municipal School Joint Programme from Guangzhou Science and Technological Project (202201020012), and the Leading Talents Program from The First Affiliated Hospital of Jinan University to HZ.
Author information
Authors and Affiliations
Contributions
Conceptualization: HZ; methodology: XFS, FSL, NHY, SQD, DWY; investigation: HZ, JD, HM, HW; visualization: XFS, NHY, JD; funding acquisition: HZ; project administration: HZ; supervision: HZ, JD, EFS; resources: DWY, EFS; writing original draft: XFS, JD; writing review & editing: HZ, JD, EFS.
Corresponding authors
Ethics declarations
Ethical approval
All animal experiments were performed in accordance with the National Research Council’s Guide for the Care and Use of Laboratory Animals, the National Institute of Health guidelines, and the ARRIVE guidelines on ethics. The study was authorized by the Animal Studies Committee of Jinan University (Guangzhou, China) and performed in accordance with the guidelines issued by the committee.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Highlights
• Autophagy is triggered during DOX-induced cardiotoxicity.
• Atg7 deficiency attenuates DOX-induced cardiac dysfunction in vivo.
• VPS34 inhibitor SAR405 provided protection against DOX-induced cardiotoxicity by inhibiting autophagy in vitro and in vivo.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sun, X., Du, J., Meng, H. et al. Targeting autophagy with SAR405 alleviates doxorubicin-induced cardiotoxicity. Cell Biol Toxicol 39, 3255–3267 (2023). https://doi.org/10.1007/s10565-023-09831-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-023-09831-8