Abstract
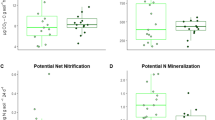
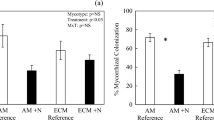
Forest mycorrhizal type is getting more attention as a potentially significant factor controlling carbon (C) and nitrogen (N) cycling. Ectomycorrhizal (ECM) forests are frequently reported to have lower N availability and higher soil C storage than arbuscular mycorrhizal (AM) forests. However, it is still unclear whether such characteristics stem from the low organic matter quality inherent in the ECM forest or other biotic and abiotic factors, such as competition for N between ECM fungi and free-living microbes. We conducted soil and litter reciprocal transplant experiments between AM-symbiotic black locust and ECM-symbiotic oak forests to separate the effects of organic matter quality and forest type (i.e., factors including ECM fungal presence and soil physicochemical properties) on decomposition rates and N availability. We hypothesized that the forest type, rather than organic matter quality, is a more determinant factor for available N content but not organic matter decomposition rate. Forest type had a more substantial effect not only on nitrate content but also on decomposition rate than organic matter quality. Since the litter decomposition rate was higher when placed in the oak forest, the higher soil C accumulation in the oak than in the black locust forests may be caused by greater C input rather than the slower decomposition in the oak than black locust forest. In addition, nitrate content was determined by forest type, suggesting the suppression of nitrate content by ECM fungal presence. This study suggests that the forest type with different mycorrhizal associations can affect biogeochemical cycling independent of organic matter quality.




Similar content being viewed by others
Data availability
Enquiries about data availability should be directed to the authors.
References
Aerts R (1997) Climate, leaf litter chemistry and leaf litter decomposition in terrestrial ecosystems: a triangular relationship. Oikos 79:439–449
Averill C, Turner BL, Finzi AC (2014) Mycorrhiza-mediated competition between plants and decomposers drives soil carbon storage. Nature 505:543–545. https://doi.org/10.1038/nature12901
Bengtsson G, Bengtson P, Månsson KF (2003) Gross nitrogen mineralization-, immobilization-, and nitrification rates as a function of soil C/N ratio and microbial activity. Soil Biol Biochem 35:143–154. https://doi.org/10.1016/S0038-0717(02)00248-1
Berg B, Berg MP, Bottner P et al (1993) Litter mass loss rates in pine forests of Europe and Eastern United States: some relationships with climate and litter quality. Biogeochemistry 20:127–159. https://doi.org/10.1007/BF00000785
Berlemont R, Martiny AC (2013) Phylogenetic distribution of potential cellulases in bacteria. Appl Environ Microbiol 79:1545–1554. https://doi.org/10.1128/AEM.03305-12
Bödeker ITM, Lindahl BD, Olson Ã, Clemmensen KE (2016) Mycorrhizal and saprotrophic fungal guilds compete for the same organic substrates but affect decomposition differently. Funct Ecol 30:1967–1978. https://doi.org/10.1111/1365-2435.12677
Bremner JM, Jenkinson DS (1960) Determination of organic carbon in soil. J Soil Sci 11:394–402. https://doi.org/10.1111/j.1365-2389.1960.tb01093.x
Brzostek ER, Dragoni D, Brown ZA, Phillips RP (2015) Mycorrhizal type determines the magnitude and direction of root-induced changes in decomposition in a temperate forest. New Phytol 206:1274–1282. https://doi.org/10.1111/nph.13303
Callahan BJ, Mcmurdie PJ, Rosen MJ et al (2016) DADA2: high-resolution sample inference from Illumina amplicon data. Nat Methods 13:581. https://doi.org/10.1038/nmeth.3869
Carteron A, Cichonski F, Laliberté E (2021) Ectomycorrhizal stands accelerate decomposition to a greater extent than arbuscular mycorrhizal stands in a northern deciduous forest. Ecosystems. https://doi.org/10.1007/s10021-021-00712-x
Chalot M, Brun A (1998) Physiology of organic nitrogen acquisition by ectomycorrhizal fungi and ectomycorrhizas. FEMS Microbiol Rev 22:21–44. https://doi.org/10.1111/j.1574-6976.1998.tb00359.x
Cheng JM, Wan HE (2002) Vegetation construction and soil and water conservation in the Loess Plateau of China. China Forestry Publishing House, Beijing
Clemmensen KE, Finlay RD, Dahlberg A et al (2014) Carbon sequestration is related to mycorrhizal fungal community shifts during long term succession in boreal forests. New Phytol 205:1525–1536
Cornelissen J, Aerts R, Cerabolini B et al (2001) Carbon cycling traits of plant species are linked with mycorrhizal strategy. Oecologia 129:611–619. https://doi.org/10.1007/s004420100752
Courty PE, Buee M, Diedhiou AG et al (2010) The role of ectomycorrhizal communities in forest ecosystem processes: new perspectives and emerging concepts. Soil Biol Biochem 42:679–698. https://doi.org/10.1016/J.Soilbio.2009.12.006
Cullings K, Ishkhanova G, Ishkhanov G, Henson J (2010) Induction of saprophytic behavior in the ectomycorrhizal fungus Suillus granulatus by litter addition in a Pinus contorta (lodgepole pine) stand in Yellowstone. Soil Biol Biochem 42:1176–1178. https://doi.org/10.1016/j.soilbio.2010.03.010
Fernandez CW, Kennedy PG (2016) Revisiting the Gadgil effect: do interguild fungal interactions control carbon cycling in forest soils? New Phytol 209:1382–1394. https://doi.org/10.1111/nph.13648
Fernandez CW, See CR, Kennedy PG (2020) Decelerated carbon cycling by ectomycorrhizal fungi is controlled by substrate quality and community composition. New Phytol 226:569–582. https://doi.org/10.1111/nph.16269
Gadgil RL, Gadgil PD (1971) Mycorrhiza and litter decomposition. Nature 233:133
He QY, Yan MJ, Miyazawa Y et al (2020) Sap flow changes and climatic responses over multiple-year treatment of rainfall exclusion in a sub-humid black locust plantation. For Ecol Manage 457:117730. https://doi.org/10.1016/j.foreco.2019.117730
Hodge A, Campbell CD, Fitter AH (2001) An arbuscular mycorrhizal fungus accelerates decomposition and acquires nitrogen directly from organic material. Nature 413:297–299. https://doi.org/10.1038/35095041
Isobe K, Ise Y, Kato H et al (2020) Consequences of microbial diversity in forest nitrogen cycling: diverse ammonifiers and specialized ammonia oxidizers. ISME J 14:12–25. https://doi.org/10.1038/s41396-019-0500-2
Iwaoka C, Imada S, Taniguchi T et al (2018) The impacts of soil fertility and salinity on soil nitrogen dynamics mediated by the soil microbial community beneath the halophytic shrub tamarisk. Microb Ecol 75:985–996. https://doi.org/10.1007/s00248-017-1090-z
Janusz G, Pawlik A, Sulej J et al (2017) Lignin degradation: microorganisms, enzymes involved, genomes analysis and evolution. FEMS Microbiol Rev 41:941–962. https://doi.org/10.1093/femsre/fux049
Kjeldahl J (1883) A new method for the determination of nitrogen in organic matter. Fresenius’ Z Anal Chem 22:366–382. https://doi.org/10.1007/BF01338151
Kohler A, Kuo A, Nagy LG et al (2015) Convergent losses of decay mechanisms and rapid turnover of symbiosis genes in mycorrhizal mutualists. Nat Genet 47:410–415. https://doi.org/10.1038/ng.3223
Koide RT, Wu T (2003) Ectomycorrhizas and retarded decomposition in a Pinus resinosa plantation. New Phytol 158:401–407. https://doi.org/10.1046/j.1469-8137.2003.00732.x
Lang AK, Jevon FV, Vietorisz CR et al (2021) Fine roots and mycorrhizal fungi accelerate leaf litter decomposition in a northern hardwood forest regardless of dominant tree mycorrhizal associations. New Phytol 230:316–326. https://doi.org/10.1111/nph.17155
Li YY, Shao MA (2006) Change of soil physical properties under long-term natural vegetation restoration in the Loess Plateau of China. J Arid Environ 64:77–96. https://doi.org/10.1016/j.jaridenv.2005.04.005
Li T, Ren B, Wang D, Liu G (2015) Spatial variation in the storages and age-related dynamics of forest carbon sequestration in different climate zones-evidence from black locust plantations on the Loess Plateau of China. PLoS ONE 10:e0121862. https://doi.org/10.1371/journal.pone.0121862
Li Z, Zeng Z, Tian D et al (2020) Global patterns and controlling factors of soil nitrification rate. Glob Chang Biol 26:4147–4157. https://doi.org/10.1111/gcb.15119
Lin G, Mccormack ML, Ma C, Guo D (2016) Similar below-ground carbon cycling dynamics but contrasting modes of nitrogen cycling between arbuscular mycorrhizal and ectomycorrhizal forests. New Phytol. https://doi.org/10.1111/nph.14206
Mushinski RM, Payne ZC, Raff JD et al (2020) Nitrogen cycling microbiomes are structured by plant mycorrhizal associations with consequences for nitrogen oxide fluxes in forests. Glob Chang Biol 27:1068–1082. https://doi.org/10.1111/gcb.15439
Oksanen AJ, Blanchet FG, Friendly M et al (2016) Package ‘vegan’(Version 2.4-0)
Orwin KH, Kirschbaum MUF, St John MG, Dickie IA (2011) Organic nutrient uptake by mycorrhizal fungi enhances ecosystem carbon storage: a model-based assessment. Ecol Lett 14:493–502. https://doi.org/10.1111/j.1461-0248.2011.01611.x
Phillips RP, Fahey TJ (2006) Tree species and mycorrhizal associations influence the magnitude of rhizosphere effects. Ecology 87:1302–1313
Phillips RP, Brzostek E, Midgley MG (2013) The mycorrhizal-associated nutrient economy: a new framework for predicting carbon–nutrient couplings in temperate forests. New Phytol 199:41–51. https://doi.org/10.1111/nph.12221
Põlme S, Abarenkov K, Henrik Nilsson R et al (2020) FungalTraits: a user-friendly traits database of fungi and fungus-like stramenopiles. Fungal Divers 105:1. https://doi.org/10.1007/s13225-020-00466-2
Prosser JI, Nicol GW (2012) Archaeal and bacterial ammonia-oxidisers in soil: the quest for niche specialisation and differentiation. Trends Microbiol 20:523–531. https://doi.org/10.1016/j.tim.2012.08.001
Qiu L, Zheng F, Yin R (2012) SWAT-based runoff and sediment simulation in a small watershed, the loessial hilly-gullied region of China: capabilities and challenges. Int J Sediment Res 27:226–234. https://doi.org/10.1016/S1001-6279(12)60030-4
Romero-Olivares AL, Morrison EW, Pringle A, Frey SD (2021) Linking genes to traits in fungi. Microb Ecol 82:145–155. https://doi.org/10.1007/s00248-021-01687-x
Seyfried GS, Midgley MG, Phillips RP, Yang WH (2022) Data for refining the role of nitrogen mineralization in mycorrhizal nutrient syndromes. Biogeochemistry. https://doi.org/10.1007/s10533-023-01038-7
Shimadzu (2013) 680°C combusion catalytic oxidation method measurement principles
Smith SE, Read DJ (2008) Mycorrhizal symbiosis. Elsevier, New York
Smith SE, Smith FA (2011) Roles of arbuscular mycorrhizas in plant nutrition and growth: new paradigms from cellular to ecosystem scales. Annu Rev Plant Biol 62:227–250. https://doi.org/10.1146/annurev-arplant-042110-103846
Smith GR, Wan J (2019) Resource-ratio theory predicts mycorrhizal control of litter decomposition. New Phytol 223:1595–1606. https://doi.org/10.1111/nph.15884
Soudzilovskaia NA, Vaessen S, Barcelo M et al (2020) FungalRoot: global online database of plant mycorrhizal associations. New Phytol 227:955–966. https://doi.org/10.1111/nph.16569
Suzuki MT, Giovannoni SJ (1996) Bias caused by template annealing in the amplification of mixtures of 16S rRNA genes. Appl Environ Microbiol 62:2–8
Tateno R, Tokuchi N, Yamanaka N et al (2007) Comparison of litterfall production and leaf litter decomposition between an exotic black locust plantation and an indigenous oak forest near Yan’an on the Loess Plateau, China. For Ecol Manage 241:84–90. https://doi.org/10.1016/j.foreco.2006.12.026
Tatsumi C, Taniguchi T, Du S et al (2019) The steps in the soil nitrogen transformation process vary along an aridity gradient via changes in the microbial community. Biogeochemistry 144:15–29. https://doi.org/10.1007/s10533-019-00569-2
Tatsumi C, Taniguchi T, Du S et al (2020) Soil nitrogen cycling is determined by the competition between mycorrhiza and ammonia-oxidizing prokaryotes. Ecology 101:e02963. https://doi.org/10.1002/ecy.2963
Tatsumi C, Hyodo F, Taniguchi T et al (2021) Arbuscular mycorrhizal community in roots and nitrogen uptake patterns of understory trees beneath ectomycorrhizal and non-ectomycorrhizal overstory trees. Front Plant Sci 11:1–13. https://doi.org/10.3389/fpls.2020.583585
Taylor BR, Parkinson D, Parsons WFJ (1989) Nitrogen and lignin content as predictors of litter decay rates: a microcosm test. Ecology 70:97–104. https://doi.org/10.2307/1938416
Toju H, Tanabe AS, Yamamoto S, Sato H (2012) High-coverage ITS primers for the DNA-based identification of ascomycetes and basidiomycetes in environmental samples. PLoS ONE 7:e40863. https://doi.org/10.1371/journal.pone.0040863
Urakawa R, Ohte N, Shibata H et al (2016) Factors contributing to soil nitrogen mineralization and nitrification rates of forest soils in the Japanese archipelago. For Ecol Manage 361:382–396. https://doi.org/10.1016/j.foreco.2015.11.033
Wang Y, Qian P (2009) Conservative fragments in bacterial 16S rRNA genes and primer design for 16S ribosomal DNA amplicons in metagenomic studies. PLoS ONE. https://doi.org/10.1371/journal.pone.0007401
Yao H, Gao Y, Nicol GW et al (2011) Links between ammonia oxidizer community structure, abundance, and nitrification potential in acidic soils. Appl Environ Microbiol 77:4618–4625. https://doi.org/10.1128/AEM.00136-11
Yin H, Wheeler E, Phillips RP (2014) Root-induced changes in nutrient cycling in forests depend on exudation rates. Soil Biol Biochem 78:213–221. https://doi.org/10.1016/j.soilbio.2014.07.022
Yuan ZY, Chen HYH (2009) Global trends in senesced-leaf nitrogen and phosphorus. Glob Ecol Biogeogr 18:532–542. https://doi.org/10.1111/j.1466-8238.2009.00474.x
Zak DR, Pellitier PT, Argiroff WA et al (2019) Viewpoint exploring the role of ectomycorrhizal fungi in soil carbon dynamics. New Phytol 223:33
Acknowledgements
We greatly thank to members of the Institute of Soil and Water Conservation of Chinese Academy of Science (CAS), the Arid Land Research Center (ALRC) of Tottori University, and the Field Science, Education and Research Center (FSERC) of Kyoto University, for cooperation and logistics in both of field survey and laboratory analysis. This study was financially supported in part by JSPS-KAKENHI (Grant No.15H05113, 20KK0134, 20J00656), Grant-in-Aid for JSPS Research Fellow (Grant No. 17J07686), JSPS-NSFC Bilateral Joint Research Projects (No. 41411140035, 41171419), and Fund of Joint Research Program of Arid Land Research Center, Tottori University.
Funding
This study was funded by Japan Society for the Promotion of Science (20J00656, 17J07686, 20KK0134, 15H05113, 41171419); National Natural Science Foundation of China (41411140035).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors have not disclosed any competing interests.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Responsible Editor: Fiona Soper.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Tatsumi, C., Taniguchi, T., Hyodo, F. et al. Mycorrhizal type affects forest nitrogen availability, independent of organic matter quality. Biogeochemistry 165, 327–340 (2023). https://doi.org/10.1007/s10533-023-01087-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10533-023-01087-y