Abstract
Hepatocellular carcinoma (HCC) treatment is a major challenge. Although andrographolide (Andro) has an anti-proliferation effect on HCC, its underlying mechanism is not yet elucidated, and whether Andro can inhibit HCC metastasis has not been reported. The present study aimed to clarify whether Andro inhibits SK-Hep-1 cell proliferation and HCC metastasis, and the mechanisms. The results showed that Andro significantly reduced the survival of HCC cells and tumor weight and volume in tumor-bearing nude mice. Andro also triggered apoptosis of HCC cells and upregulated MIR22HG, Cleaved Caspase 9/7/3 expression levels, and downregulated BCL-2 mRNA, BCL-2 expression levels. Knockdown of MIR22HG or overexpression of HuR attenuated the effects of Andro on the signal transduction of mitochondrial apoptotic pathway and proliferation ability in HCC cells. Moreover, Andro significantly reduced the invasive ability of the cells and the level of HCC cell lung metastasis, upregulated miR-22-3p expression level and downregulated HMGB1 and MMP-9 expression levels. MIR22HG or miR-22-3p knockdown attenuated the effects of Andro on the signaling of HMGB1/MMP-9 pathway and invasive ability in HCC cells, while the overexpression of HMGB1 attenuated the inhibitory effects of Andro on the MMP-9 expression level and invasive ability in HCC cells. Thus, the regulation of MIR22HG-HuR/BCL-2 and MIR22HG/HMGB1 signaling pathways is involved in the anti-HCC proliferation and metastasis effects of Andro. This study provided a new pharmacological basis for Andro in HCC treatment and, for the first time, identified a natural product molecule capable of positively regulating MIR22HG, which has a robust biological function.
Graphical abstract












Similar content being viewed by others
Abbreviations
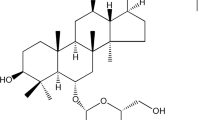
- Andro:
-
Andrographolide
- HCC:
-
Hepatocellular carcinoma
- CCK-8:
-
Cell counting kit-8
- LDH:
-
Lactate dehydrogenase
- EdU:
-
5-Ethynyl-20-deoxyuridine
- PI:
-
Propidium iodide
- TUNEL:
-
Terminal deoxynucleotidyl transferase-mediated biotinylated UTP nick end labeling
- MMP:
-
Matrix metalloproteinase
- JC-1:
-
5,5′,6,6′-Tetrachloro-1,1′,3,3′-tetraethylbenzimidazolylcarbocyanine iodide
- LncRNAs:
-
Long non-coding RNAs
- BCL-2:
-
B-cell leukemia/lymphoma 2
- MIR22HG:
-
Human miR-22 host gene
- HuR:
-
Human antigen R
- ALT:
-
Glutamic-pyruvic transaminase
- AST:
-
Glutamic oxaloacetic transaminase
- HMGB1:
-
High-mobility group protein B1
- BUN:
-
Blood urea nitrogen
- CREA:
-
Creatinine
- Cyt C:
-
Cytochrome C
References
Torre LA, Bray F, Siegel RL, Ferlay J, Lortet-Tieulent J, Jemal A (2015) Global cancer statistics, 2012. CA Cancer J Clin 65(2):87–108
Sartorius K, Sartorius B, Aldous C, Govender PS, Madiba TE (2015) Global and country underestimation of hepatocellular carcinoma (HCC) in 2012 and its implications. Cancer Epidemiol 39(3):284–290
de Lope CR, Tremosini S, Forner A, Reig M, Bruix J (2012) Management of HCC. J Hepatol 56(Suppl 1):S75-87
Aino H, Sumie S, Niizeki T, Kuromatsu R, Tajiri N, Nakano M, Satani M, Yamada S, Okamura S, Shimose S, Sumie H, Torimura T, Sata M (2014) Clinical characteristics and prognostic factors for advanced hepatocellular carcinoma with extrahepatic metastasis. Mol Clin Oncol 2(3):393–398
Natsuizaka M, Omura T, Akaike T, Kuwata Y, Yamazaki K, Sato T, Karino Y, Toyota J, Suga T, Asaka M (2005) Clinical features of hepatocellular carcinoma with extrahepatic metastases. J Gastroenterol Hepatol 20(11):1781–1787
Kopp F, Mendell JT (2018) Functional classification and experimental dissection of long noncoding RNAs. Cell 172(3):393–407
Beermann J, Piccoli MT, Viereck J, Thum T (2016) Non-coding RNAs in development and disease: background, mechanisms, and therapeutic approaches. Physiol Rev 96(4):1297–1325
Lavorgna G, Vago R, Sarmini M, Montorsi F, Salonia A, Bellone M (2016) Long non-coding RNAs as novel therapeutic targets in cancer. Pharmacol Res 110:131–138
Takahashi K, Yan I, Haga H, Patel T (2014) Long noncoding RNA in liver diseases. Hepatology 60(2):744–753
Zheng J, Xiong D, Sun X, Wang J, Hao M, Ding T, Xiao G, Wang X, Mao Y, Fu Y, Shen K, Wang J (2012) Signification of hypermethylated in cancer 1 (HIC1) as tumor suppressor gene in tumor progression. Cancer Microenviron 5(3):285–293
Zhao X, He M, Wan D, Ye Y, He Y, Han L, Guo M, Huang Y, Qin W, Wang MW, Chong W, Chen J, Zhang L, Yang N, Xu B, Wu M, Zuo L, Gu J (2003) The minimum LOH region defined on chromosome 17p13.3 in human hepatocellular carcinoma with gene content analysis. Cancer Lett 190(2):221–232
Zhang DY, Zou XJ, Cao CH, Zhang T, Lei L, Qi XL, Liu L, Wu DH (2018) Identification and functional characterization of long non-coding RNA MIR22HG as a tumor suppressor for hepatocellular carcinoma. Theranostics 8(14):3751–3765
Liu PL, Tsai JR, Hwang JJ, Chou SH, Cheng YJ, Lin FY, Chen YL, Hung CY, Chen WC, Chen YH, Chong IW (2010) High-mobility group box 1-mediated matrix metalloproteinase-9 expression in non-small cell lung cancer contributes to tumor cell invasiveness. Am J Respir Cell Mol Biol 43(5):530–538
Wu Y, Zhou Y, Huan L, Xu L, Shen M, Huang S, Liang L (2019) LncRNA MIR22HG inhibits growth, migration and invasion through regulating the miR-10a-5p/NCOR2 axis in hepatocellular carcinoma cells. Cancer Sci 110(3):973–984
Shi L, Zhang G, Zheng Z, Lu B, Ji L (2017) Andrographolide reduced VEGFA expression in hepatoma cancer cells by inactivating HIF-1α: the involvement of JNK and MTA1/HDCA. Chem Biol Interact 273:228–236
Ji L, Shen K, Jiang P, Morahan G, Wang Z (2011) Critical roles of cellular glutathione homeostasis and jnk activation in andrographolide-mediated apoptotic cell death in human hepatoma cells. Mol Carcinog 50(8):580–591
Pearngam P, Kumkate S, Okada S, Janvilisri T (2019) Andrographolide inhibits cholangiocarcinoma cell migration by down-regulation of claudin-1 via the p-38 signaling pathway. Front Pharmacol 10:827
Dai L, Wang G, Pan W (2017) Andrographolide inhibits proliferation and metastasis of SGC7901 gastric cancer cells. Biomed Res Int 2017:6242103
Dai Y, Chen SR, Chai L, Zhao J, Wang Y, Wang Y (2019) Overview of pharmacological activities of Andrographis paniculata and its major compound andrographolide. Crit Rev Food Sci Nutr 59(sup1):S17-s29
Zhang L, Bao M, Liu B, Zhao H, Zhang Y, Ji X, Zhao N, Zhang C, He X, Yi J, Tan Y, Li L, Lu C (2020) Effect of andrographolide and its analogs on bacterial infection: a review. Pharmacology 105(3–4):123–134
Qu J, Liu Q, You G, Ye L, Jin Y, Kong L, Guo W, Xu Q, Sun Y (2022) Advances in ameliorating inflammatory diseases and cancers by andrographolide: pharmacokinetics, pharmacodynamics, and perspective. Med Res Rev 42(3):1147–1178
Chua LS (2014) Review on liver inflammation and antiinflammatory activity of Andrographis paniculata for hepatoprotection. Phytother Res 28(11):1589–1598
Zhou J, Zhang S, Ong CN, Shen HM (2006) Critical role of pro-apoptotic Bcl-2 family members in andrographolide-induced apoptosis in human cancer cells. Biochem Pharmacol 72(2):132–144
Xu J, Shao T, Song M, Xie Y, Zhou J, Yin J, Ding N, Zou H, Li Y, Zhang J (2020) MIR22HG acts as a tumor suppressor via TGFβ/SMAD signaling and facilitates immunotherapy in colorectal cancer. Mol Cancer 19(1):51
Cui Z, An X, Li J, Liu Q, Liu W (2018) LncRNA MIR22HG negatively regulates miR-141-3p to enhance DAPK1 expression and inhibits endometrial carcinoma cells proliferation. Biomed Pharmacother 104:223–228
Su W, Feng S, Chen X, Yang X, Mao R, Guo C, Wang Z, Thomas DG, Lin J, Reddy RM, Orringer MB, Chang AC, Yang Z, Beer DG, Chen G (2018) Silencing of long noncoding RNA MIR22HG triggers cell survival/death signaling via oncogenes YBX1, MET, and p21 in lung cancer. Cancer Res 78(12):3207–3219
Deng X, Ye D, Hua K, Song H, Luo Q, Munankarmy A, Liu D, Zhou B, Zheng W, Zhou X, Ji C, Wang X, Yu Y, Fang L (2021) MIR22HG inhibits breast cancer progression by stabilizing LATS2 tumor suppressor. Cell Death Dis 12(9):810
Abdelmohsen K, Gorospe M (2010) Posttranscriptional regulation of cancer traits by HuR. Wiley Interdiscip Rev RNA 1(2):214–229
Kim J, Abdelmohsen K, Yang X, De S, Grammatikakis I, Noh JH, Gorospe M (2016) LncRNA OIP5-AS1/cyrano sponges RNA-binding protein HuR. Nucleic Acids Res 44(5):2378–2392
Abdelmohsen K, Lal A, Kim HH, Gorospe M (2007) Posttranscriptional orchestration of an anti-apoptotic program by HuR. Cell Cycle 6(11):1288–1292
Ishimaru D, Ramalingam S, Sengupta TK, Bandyopadhyay S, Dellis S, Tholanikunnel BG, Fernandes DJ, Spicer EK (2009) Regulation of Bcl-2 expression by HuR in HL60 leukemia cells and A431 carcinoma cells. Mol Cancer Res 7(8):1354–1366
Ayupova DA, Singh M, Leonard EC, Basile DP, Lee BS (2009) Expression of the RNA-stabilizing protein HuR in ischemia-reperfusion injury of rat kidney. Am J Physiol Renal Physiol 297(1):F95-f105
Green DR, Kroemer G (2004) The pathophysiology of mitochondrial cell death. Science 305(5684):626–629
Danial NN, Korsmeyer SJ (2004) Cell death: critical control points. Cell 116(2):205–219
Slee EA, Adrain C, Martin SJ (1999) Serial killers: ordering caspase activation events in apoptosis. Cell Death Differ 6(11):1067–1074
Kuribayashi K, Mayes PA, El-Deiry WS (2006) What are caspases 3 and 7 doing upstream of the mitochondria? Cancer Biol Ther 5(7):763–765
França GS, Vibranovski MD, Galante PA (2016) Host gene constraints and genomic context impact the expression and evolution of human microRNAs. Nat Commun 7:11438
Patel JB, Appaiah HN, Burnett RM, Bhat-Nakshatri P, Wang G, Mehta R, Badve S, Thomson MJ, Hammond S, Steeg P, Liu Y, Nakshatri H (2011) Control of EVI-1 oncogene expression in metastatic breast cancer cells through microRNA miR-22. Oncogene 30(11):1290–1301
Wang J, Xiang G, Zhang K, Zhou Y (2012) Expression signatures of intragenic miRNAs and their corresponding host genes in myeloid leukemia cells. Biotechnol Lett 34(11):2007–2015
Ellerman JE, Brown CK, de Vera M, Zeh HJ, Billiar T, Rubartelli A, Lotze MT (2007) Masquerader: high mobility group box-1 and cancer. Clin Cancer Res 13(10):2836–2848
Tang D, Kang R, Zeh HJ 3rd, Lotze MT (2010) High-mobility group box 1 and cancer. Biochim Biophys Acta 1799(1–2):131–140
Funding
This study was supported by National Natural Science Foundation of China (No. 82260793); China Postdoctoral Science Foundation funded project (No. 2020M680834); Jiangxi Provincial Natural Science Foundation (No. 20212BAB216002); The Key R & D Project of Ganzhou Science and Technology Plan Project (2022B-SF8897); The Start-up Fund of Gannan Medical University (No. QD201821); The Science and Technology Project of the Education Department of Jiangxi Province (No. GJJ211517).
Author information
Authors and Affiliations
Contributions
Conceived and designed the experiments: YJ, YL. Performed the experiments: YL, JH, YJ. Analyzed the data: JH, DM, YS, WY. Contributed reagents/materials/analysis tools: YJ, YL. Wrote and revised the paper: YJ, YL.
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no competing financial interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Order of corresponding authors was incorrect and corrected in this version.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Luo, Y., Hu, J., Jiao, Y. et al. Andrographolide anti-proliferation and metastasis of hepatocellular carcinoma through LncRNA MIR22HG regulation. J Nat Med 78, 123–145 (2024). https://doi.org/10.1007/s11418-023-01752-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11418-023-01752-4