Abstract
Background
Gastric cancer (GC) is threatening public health as there are at least one million new cases reported each year. Rhomboid domain-containing protein 1 (SEC61G) has been identified to regulate tumor cell survival, metastasis and cellular response by regulating glycolysis. However, the function of SEC61G in gastric cancer is not fully understood.
Objectives
To investigate the effect of SEC61G on proliferation, epidermal growth factor receptor (EGFR) and glycolysis levels on GC, as well as the sensitivity of GC to etoposide.
Results
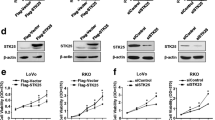
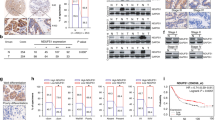
SEC61G is highly expressed in gastric cancer tissues and gastric cell lines. SEC61G knockdown suppresses the cell viability and EdU incorporation. Meanwhile, SEC61G knockdown attenuated the phosphorylation of EGFR and AKT. SEC61G knockdown suppressed the migration, invasion, glucose uptake, lactate release and ATP production, while elevated oxygen consumption rate (OCR), promoting the sensitivity of gastric cancer to etoposide.
Conclusions
SEC61G knockdown weakened the proliferation, EGFR phosphorylation and glycolysis of gastric cancer cells, as well as increased the sensitivity of gastric cancer to etoposide, which make SEC61G a promising therapeutic target for gastric cancer treatment.






Similar content being viewed by others
Availability of data and materials
All data generated or analyzed during this study are included in this published article. The data sets used and/or analyzed during the present study are available from the corresponding author on reasonable request.
References
Chandrashekar DS, Karthikeyan SK, Korla PK, Patel H, Shovon AR, Athar M, Netto GJ, Qin ZS, Kumar S, Manne U (2022) UALCAN: an update to the integrated cancer data analysis platform. Neoplasia 25:18–27. https://doi.org/10.1016/j.neo.2022.01.001
Dragovich T, Campen C (2009) Anti-EGFR-targeted therapy for esophageal and gastric cancers: an evolving concept. J Oncol. https://doi.org/10.1155/2009/804108
Fouzder C, Mukhuty A, Mukherjee S, Malick C, Kundu R (2021) Trigonelline inhibits Nrf2 via EGFR signalling pathway and augments efficacy of Cisplatin and Etoposide in NSCLC cells. Toxicol Vitro 70:105038. https://doi.org/10.1016/j.tiv.2020.105038
Gong Y, Hu Y, Huang J, Wang H (2022) RAB11A aggravates PDGF-BB-stimulated proliferation, migration, and inflammation of airway smooth muscle cells via affecting the NF-κB and PI3K/AKT pathways. Allergol Immunopathol 50(4):147–154. https://doi.org/10.15586/aei.v50i4.635
Huang J, Dai W, Xiao D, Xiong Q, Liu C, Hu J, Ge F, Yu X, Li S (2022) Acetylation-dependent SAGA complex dimerization promotes nucleosome acetylation and gene transcription. Nat Struct Mol Biol 29(3):261–273. https://doi.org/10.1038/s41594-022-00736-4
Li T, Fan J, Wang B, Traugh N, Chen Q, Liu JS, Li B, Liu XS (2017) TIMER: a web server for comprehensive analysis of tumor-infiltrating immune cells. Can Res 77(21):e108–e110. https://doi.org/10.1158/0008-5472.CAN-17-0307
Liu B, Liu J, Liao Y, Jin C, Zhang Z, Zhao J, Liu K, Huang H, Cao H, Cheng Q (2019) Identification of SEC61G as a novel prognostic marker for predicting survival and response to therapies in patients with glioblastoma. Med Sci Monitor 25:3624–3635. https://doi.org/10.12659/MSM.916648
Lollies A, Hartmann S, Schneider M, Bracht T, Weiss A, Arnolds J, Klein-Hitpass L, Sitek B, Hansmann M, Küppers R (2018) An oncogenic axis of STAT-mediated BATF3 upregulation causing MYC activity in classical Hodgkin lymphoma and anaplastic large cell lymphoma. Leukemia 32(1):92–101. https://doi.org/10.1038/leu.2017.203
Lu Z, Zhou L, Killela P, Rasheed AB, Di C, Poe WE, McLendon RE, Bigner DD, Nicchitta C, Yan H (2009) Glioblastoma proto-oncogene SEC61γ is required for tumor cell survival and response to endoplasmic reticulum stress. Can Res 69(23):9105–9111. https://doi.org/10.1158/0008-5472.CAN-09-2775
Lu T, Zhang Q, Wu X, Liu S (2022) KLF8 enhances acute myeloid leukemia cell growth and glycolysis via AKT/mTOR pathway. Trop J Pharmaceut Res 21(6):1169–1175. https://doi.org/10.4314/tjpr.v21i6.5
Ma J, He Z, Zhang H, Zhang W, Gao S, Ni X (2021) SEC61G promotes breast cancer development and metastasis via modulating glycolysis and is transcriptionally regulated by E2F1. Cell Death Dis 12(6):550. https://doi.org/10.1038/s41419-021-03797-3
Montecucco A, Zanetta F, Biamonti G (2015) Molecular mechanisms of etoposide. EXCLI J 14:95. https://doi.org/10.17179/excli2015-561
Paul D, Chanukuppa V, Reddy PJ, Taunk K, Adhav R, Srivastava S, Santra MK, Rapole S (2016) Global proteomic profiling identifies etoposide chemoresistance markers in non-small cell lung carcinoma. J Proteomics 138:95–105. https://doi.org/10.1016/j.jprot.2016.02.008
Tsukamoto Y, Uchida T, Karnan S, Noguchi T, Nguyen L, Tanigawa M, Takeuchi I, Matsuura K, Hijiya N, Nakada C (2008) Genome-wide analysis of DNA copy number alterations and gene expression in gastric cancer. J Pathol 216(4):471–482. https://doi.org/10.1002/path.2424
Wang D, Wang B, Wang R, Zhang Z, Lin Y, Huang G, Lin S, Jiang Y, Wang W, Wang L (2017) High expression of EGFR predicts poor survival in patients with resected T3 stage gastric adenocarcinoma and promotes cancer cell survival. Oncol Lett 13(5):3003–3013. https://doi.org/10.3892/ol.2017.5827
Wang Q, Zhang J, Ye J, Guo J (2022) Vitexin exerts anti-tumor and anti-angiogensis effects on cervical cancer through VEGFA/VEGFR2 pathway. Euro J Gynaecol Oncol 43(4):86–91. https://doi.org/10.22514/ejgo.2022.036
Xiang X, Su Y, Li G-l, Ma L, Zhou C, Ma R (2020) Phase II study of preoperative intra-arterial epirubicin, etoposide, and oxaliplatin combined with oral S-1 chemotherapy for the treatment of Borrmann Type 4 gastric cancer. J Gastric Cancer 20(4):395. https://doi.org/10.5230/jgc.2020.20.e40
Yu HG, Ai YW, Yu LL, Zhou XD, Liu J, Li JH, Xu XM, Liu S, Chen J, Liu F (2008) Phosphoinositide 3-kinase/Akt pathway plays an important role in chemoresistance of gastric cancer cells against etoposide and doxorubicin induced cell death. Int J Cancer 122(2):433–443. https://doi.org/10.1002/ijc.23049
Zhang W, Gou P, Dupret J-M, Chomienne C, Rodrigues-Lima F (2021) Etoposide, an anticancer drug involved in therapy-related secondary leukemia: Enzymes at play. Transl Oncol 14(10):101169. https://doi.org/10.1016/j.tranon.2021.101169
Author information
Authors and Affiliations
Contributions
Conceptualization, methodology, and writing—original draft were performed by Dengfeng Wu; formal analysis, resources, and investigation were performed by Chunying Fang; formal analysis, visualization and data curation were performed by Yazhi Chen; project administration, supervision, and validation were performed by Xuefeng Xu; validation, supervision, and writing—review & editing were performed by Xiongbo Wu and Sijie Chen. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
Dengfeng Wu declares that he/she has no conflict of interest. Chunying Fang declares that he/she has no conflict of interest. Yazhi Chen declares that he/she has no conflict of interest. Xuefeng Xu declares that he/she has no conflict of interest. Xiongbo Wu declares that he/she has no conflict of interest. Sijie Chen declares that he/she has no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wu, D., Fang, C., Chen, Y. et al. SEC61G knockdown enhances the sensitivity of gastric cancer cells to etoposide through EGFR and glycolytic-mediated pathways. Mol. Cell. Toxicol. (2023). https://doi.org/10.1007/s13273-023-00403-9
Accepted:
Published:
DOI: https://doi.org/10.1007/s13273-023-00403-9