Abstract
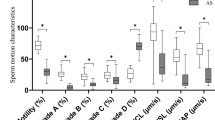
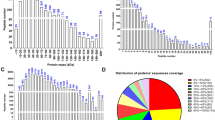
Low acrosin activity (LAA) is associated with sperm function anomaly and poor outcomes of in vitro fertilization. In this study, we confirm that 993 semen samples with LAA had a reduced sperm motility and low in vitro fertilization rate in comparison with 1332 normal controls (NC). Proteomic comparison between 11 LAA and 11 NC sperm samples identified 35 upregulated and 99 downregulated proteins in the LAA group. Indeed, proteomic data showed that acrosome enzymes Spam1 and Acrosin were among the downregulated proteins in the LAA group, which was validated by quantitative PCR and immunefluorescent staining of sperm cells. The KEEG pathway analysis revealed a deficiency of GSH and Gln biosynthesis in LAA sperm cells. Immunofluorescent staining of sperms and quantitative PCR verified downregulation of GLUL and GCLC, the key enzymes for GSH and Gln biosynthesis. Moreover, the results of ELISA assay confirmed low levels of GSH and Gln in LAA sperm cells. Mechanistic studies showed that addition of 10 mM H2O2 to semen samples led to a significant reduction of acrosin activity and sperm motility, most possibly by triggering premature acrosome release. In contrast, the presence of 20 mM GSH blocked the oxidative effects of H2O2. Since GSH counteracts the oxidative stress and Gln participates in TCA cycling, their deficiency may affect the redox balance as well as energy production of sperm cells. These findings shed new light on the pathological mechanisms of infertility associated with LAA. Male infertility patients could benefit from GSH supplement by improvement of acrosin activity and other sperm functions.









Similar content being viewed by others
References
Abou-Haila A, Tulsiani DR (2000) Mammalian sperm acrosome: formation, contents, and function. Arch Biochem Biophys 379:173–182
Agarwal A, Loughlin KR (1991) Acrosin activity in patients with idiopathic infertility. Arch Androl 27:97–101
Amaral A (2022) Energy metabolism in mammalian sperm motility. Wires Mech Dis 4:1569
Baba D, Kashiwabara S, Honda A, Yamagata K, Wu Q, Ikawa M, Okabe M, Baba T (2002) Mouse sperm lacking cell surface hyaluronidase PH-20 can pass through the layer of cumulus cells and fertilize the egg. J Biol Chem 277:30310–30314
Beygi Z, Forouhari S, Mahmoudi E, Hayat SMG, Nourimand F (2021) Role of oxidative stress and antioxidant supplementation in male fertility. Curr Mol Med 21:265–282
Carlsen E, Giwercman A, Keiding N, Skakkebaek NE (1992) Evidence for decreasing quality of semen during past 50 years. BMJ 305:609–613
Chen F, Gao L, Zhou H, Guo L, Chen Q, Gan Y, Sun X, Li Q, Wang K (2019) The association between sperm head elongation and semen quality. Andrology 7:840–845
Coticchio G (2013) Polarization microscopy and rescue ICSI. Reprod Biomed Online 26:222–227
Crosby JA, Barros C (1999) Effect of recombinant boar beta-acrosin on sperm binding to intact zona pellucida during in vitro fertilization. Biol Reprod 61:1535–1540
Cruzat V, Macedo Rogero M, Noel Keane K, Curi R, Newsholme P (2018) Glutamine: metabolism and immune function, supplementation and clinical translation. Nutrients 10:1564
Cui YH, Zhao RL, Wang Q, Zhang ZY (2000) Determination of sperm acrosin activity for evaluation of male fertility. Asian J Androl 2:229–232
Dunbar BS, Dudkiewicz AB, Bundman DS (1985) Proteolysis of specific porcine zona pellucida glycoproteins by boar acrosin. Biol Reprod 32:619–630
Dutta S, Majzoub A, Agarwal A (2019a) Oxidative stress and sperm function: a systematic review on evaluation and management. Arab J Urol 17:87–97
Dutta S, Majzoub A, Agarwal A (2019b) Oxidative stress and sperm function: a systematic review on evaluation and management. Arab J Urol 24:87–97
Fathi Najafi T, Latifnejad Roudsari R, Namvar F, Ghavami Ghanbarabadi V, Hadizadeh Talasaz Z, Esmaeli M (2015) Air pollution and quality of sperm: a meta-analysis. Iran Red Crescent Med J 17:26930
Fléchon JE (2016) The acrosome of eutherian mammals. Cell Tissue Res 363:147–157
Flohé L (2013) The fairytale of the GSSG/GSH redox potential. Biochim Biophys Acta 1830:3139–3142
Fujii J, Tsunoda S (2011) Redox regulation of fertilisation and the spermatogenic process. Asian J Androl 13:420–423
Garg A, Kumaresan A, Ansari MR (2009) Effects of hydrogen peroxide (H2O2) on fresh and cryopreserved buffalo sperm functions during incubation at 37 degrees C in vitro. Reprod Domest Anim 44:907–912
Gerhard I, Fröhlich E, Eggert-Kruse W, Klinga K, Runnebaum B (1989) Relationship of sperm acrosin activity to semen and clinical parameters in infertile patients. Andrologia 21:146–154
Hirose M, Honda A, Fulka H, Tamura-Nakano M, Matoba S, Tomishima T, Mochida K, Hasegawa A, Nagashima K, Inoue K, Ohtsuka M, Baba T, Yanagimachi R, Ogura A (2020) Acrosin is essential for sperm penetration through the zona pellucida in hamsters. Proc Natl Acad Sci USA 117:2513–2518
Ito C, Toshimori K (2016) Acrosome markers of human sperm. Anat Sci Int 91:128–142
Levine H, Jørgensen N, Martino-Andrade A, Mendiola J, Weksler-Derri D, Mindlis I, Pinotti R, Swan SH (2017) Temporal trends in sperm count: a systematic review and meta-regression analysis. Hum Reprod Update 23:646–659
Li R, Wu X, Zhu Z, Lv Y, Zheng Y, Lu H, Zhou K, Wu D, Zeng W, Dong W, Zhang T (2022) Polyamines protect boar sperm from oxidative stress in vitro. J Anim Sci 100:69–84
Lu JC, Li Z, Xia SJ (2018) Expert consensus on the standardized assessment of Chinese male fertility. China Medical Science and Technology Press, Beijing
Mao HT, Yang WX (2013) Modes of acrosin functioning during fertilization. Gene 526:75–79
Miki K (2007) Energy metabolism and sperm function. Soc Reprod Fertil Suppl 65:309–325
Minhas S, Bettocchi C, Boeri L, Capogrosso P, Carvalho J, Cilesiz NC, Cocci A, Corona G, Dimitropoulos K, Gül M, Hatzichristodoulou G, Hugh Jones T, Kadioglu A, Martínez Salamanca JI, Milenkovic U, Modgil V, Russo GI, Serefoglu EC, Tharakan T, Verze P, Salonia A (2021) European Association of Urology Guidelines on Male Sexual and Reproductive Health: 2021 Update on Male Infertility. Eur Urol 80:603–620
Mohanty G, Samanta L (2018) Redox regulation & sperm function: a proteomic insight. Indian J Med Res 148:84–91
Park S, Kim YH, Jeong PS, Park C, Lee JW, Kim JS, Wee G, Song BS, Park BJ, Kim SH, Sim BW, Kim SU, Triggs-Raine B, Baba T, Lee SR, Kim E (2019) SPAM1/HYAL5 double deficiency in male mice leads to severe male subfertility caused by a cumulus-oocyte complex penetration defect. FASEB J 33:14440–14449
Ricci E, Al Beitawi S, Cipriani S, Candiani M, Chiaffarino F, Viganò P, Noli S, Parazzini F (2017) Semen quality and alcohol intake: a systematic review and meta-analysis. Reprod Biomed Online 34:38–47
Schlegel PN, Sigman M, Collura B, De Jonge CJ, Eisenberg ML, Lamb DJ, Mulhall JP, Niederberger C, Sandlow JI, Sokol RZ, Spandorfer SD, Tanrikut C, Treadwell JR, Oristaglio JT, Zini A (2021) Diagnosis and treatment of infertility in men: AUA/ASRM guideline part II. J Urol 205:44–51
Sengupta P, Borges E Jr, Dutta S, Krajewska-Kulak E (2018) Decline in sperm count in European men during the past 50 years. Hum Exp Toxicol 37:247–255
World Health Organization (2010) WHO laboratory manual for the examination and processing of human semen, 5th edn. World Health Organization, Geneva
Xu F, Zhu H, Zhu W, Fan L (2018) Human sperm acrosomal status, acrosomal responsiveness, and acrosin are predictive of the outcomes of in vitro fertilization: a prospective cohort study. Reprod Biol 18:344–354
Yang S, Lian G (2020) ROS and diseases: role in metabolism and energy supply. Mol Cell Biochem 467:1–12
Yoo HC, Yu YC, Sung Y, Han JM (2020) Glutamine reliance in cell metabolism. Exp Mol Med 52:1496–1516
Zhu Z, Kawai T, Umehara T, Hoque SAM, Zeng W, Shimada M (2019) Negative effects of ROS generated during linear sperm motility on gene expression and ATP generation in boar sperm mitochondria. Free Radic Biol Med 141:159–171
Acknowledgements
The authors thank the patients participated in this study and the physicians/technicians contributed to the study.
Funding
This work was supported by the Shuangchuang Program of Jiangsu Province (Shi-Wen Jiang); the Wuxi Leading Talents in Medical and Health Profession Program (Shi-Wen Jiang); the Fenghuanchao Program (Shi-Wen Jiang) of the Wuxi Taihu Lake Talent Plan; and research funding from Lianyungang Maternal and Child Health Hospital.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Ethical approval and informed consent
This retrospective study was approved by the Ethics Committee of Women’s Hospital of Jiangnan University (No:2021-01-0927-27), and written informed consent was signed by all study subjects. All procedures involving human participants were carried out per institutional and national ethical standards as well as the 1964 Helsinki declaration and its later amendments.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Lin, M., Ling, P., He, Q. et al. Low acrosin activity is associated with decreased Spam1/acrosin expression and GSH deficiency-caused premature acrosome release of human sperm cells. Cell Tissue Res 394, 529–545 (2023). https://doi.org/10.1007/s00441-023-03826-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00441-023-03826-x