Abstract
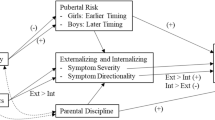
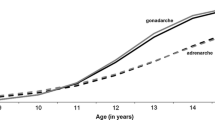
This study tested interactions among puberty-related genetic risk, prenatal substance use, harsh discipline, and pubertal timing for the severity and directionality (i.e., differentiation) of externalizing and internalizing problems and adolescent substance use. This is a companion paper to Marceau et al. (2021) which examined the same influences in developmental cascade models. Data were from the Avon Longitudinal Study of Parents and Children (ALSPAC) cohort (n = 4504 White boys, n = 4287 White girls assessed from the prenatal period through 18.5 years). We hypothesized generally that later predictors would strengthen the influence of puberty-related genetic risk, prenatal substance use exposure, and pubertal risk on psychopathology and substance use (two-way interactions), and that later predictors would strengthen the interactions of earlier influences on psychopathology and substance use (three-way interactions). Interactions were sparse. Although all fourteen interactions showed that later influences can exacerbate or trigger the effects of earlier ones, they often were not in the expected direction. The most robust moderator was parental discipline, and differing and synergistic effects of biological and socially-relevant aspects of puberty were found. In all, the influences examined here operate more robustly in developmental cascades than in interaction with each other for the development of psychopathology and transitions to substance use.




Similar content being viewed by others
Data Availability
Data for the current study is available from the ALSPAC study: http://www.bristol.ac.uk/alspac/researchers/access/. Derived variables and associated code for variables created in this study are returned to the ALSPAC executive team, and are available/distributed by ALSPAC. Data analytic scripts or additional details are freely available upon author request.
References
Bahji A, Mazhar MN, Hudson CC, Nadkarni P, MacNeil BA, Hawken E (2019) Prevalence of substance use disorder comorbidity among individuals with eating disorders: a systematic review and meta-analysis. Psychiatry Res 27:358–366
Barker DJ (1990) The fetal and infant origins of adult disease. BMJ (Clin Res ed) 301(6761):1111
Blanco C, Flórez-Salamanca L, Secades‐Villa R, Wang S, Hasin DS (2018) Predictors of initiation of nicotine, alcohol, cannabis, and cocaine use: results of the national epidemiologic survey on alcohol and related conditions (NESARC). Am J Addict 27(6):477–484
Bonadio FT, Evans SC, Cho GY, Callahan KP, Chorpita BF, Mental H, Weisz JR, the Research Network on Youth Mental H (2022) Whose Outcomes come out? Patterns of caregiver- and youth-reported outcomes based on caregiver-youth baseline discrepancies. J Clin Child Adolesc Psychol 51(4):469–483
Bowen E, Heron J, Waylen A, Wolke D, Team AS (2005) Domestic violence risk during and after pregnancy: findings from a british longitudinal study. BJOG Int J Obstet Gynaecol 112(8):1083–1089
Boyd A, Golding J, Macleod J, Lawlor DA, Fraser A, Henderson J, Molloy L, Ness A, Ring S, Davey Smith G (2013) Cohort Profile: the ‘Children of the 90s’—the index offspring of the avon longitudinal study of parents and children. Int J Epidemiol 42(1):111–127
Corley RP, Beltz AM, Wadsworth SJ, Berenbaum SA (2015) Genetic influences on pubertal development and links to behavior problems. Behav Genet 45(3):294–312
Cousminer DL, Berry DJ, Timpson NJ, Ang W, Thiering E, Byrne EM, Taal HR, Huikari V, Bradfield JP, Kerkhof M, Groen-Blokhuis MM, Kreiner-Moller E, Marinelli M, Holst C, Leinonen JT, Perry JR, Surakka I, Pietilainen O, Kettunen J, Anttila V, Kaakinen M, Sovio U, Pouta A, Das S, Lagou V, Power C, Prokopenko I, Evans DM, Kemp JP, St Pourcain B, Ring S, Palotie A, Kajantie E, Osmond C, Lehtimaki T, Viikari JS, Kahonen M, Warrington NM, Lye SJ, Palmer LJ, Tiesler CM, Flexeder C, Montgomery GW, Medland SE, Hofman A, Hakonarson H, Guxens M, Bartels M, Salomaa V, Murabito JM, Kaprio J, Sorensen TI, Ballester F, Bisgaard H, Boomsma DI, Koppelman GH, Grant SF, Jaddoe VW, Martin NG, Heinrich J, Pennell CE, Raitakari OT, Eriksson JG, Smith GD, Hypponen E, Jarvelin MR, McCarthy MI, Ripatti S, Widen E (2013) Genome-wide association and longitudinal analyses reveal genetic loci linking pubertal height growth, pubertal timing and childhood adiposity. Hum Mol Genet 22(13):2735–2747
Cribbie RA (2007) Multiplicity control in structural equation modeling. Struct Equ Model: Multidiscip J 14(1):98–112
Culbert KM, Burt SA, Klump KL (2017) Expanding the developmental boundaries of etiologic effects: the role of adrenarche in genetic influences on disordered eating in males. J Abnorm Psychol 126(5):593
Day FR, Thompson DJ, Helgason H, Chasman DI, Finucane H, Sulem P, Ruth KS, Whalen S, Sarkar AK, Albrecht E (2017) Genomic analyses identify hundreds of variants associated with age at menarche and support a role for puberty timing in cancer risk. Nat Genet 49(6):834
De Los Reyes A, Augenstein TM, Wang M, Thomas SA, Drabick DA, Burgers DE, Rabinowitz J (2015) The validity of the multi-informant approach to assessing child and adolescent mental health. Psychol Bull 141(4):858
Dick DM, Agrawal A, Keller MC, Adkins A, Aliev F, Monroe S, Hewitt JK, Kendler KS, Sher KJ (2015) Candidate gene–environment interaction research: reflections and recommendations. Perspect Psychol Sci 10(1):37–59
Elks CE, Perry JR, Sulem P, Chasman DI, Franceschini N, He C, Lunetta KL, Visser JA, Byrne EM, Cousminer DL, Gudbjartsson DF, Esko T, Feenstra B, Hottenga JJ, Koller DL, Kutalik Z, Lin P, Mangino M, Marongiu M, McArdle PF, Smith AV, Stolk L, van Wingerden SH, Zhao JH, Albrecht E, Corre T, Ingelsson E, Hayward C, Magnusson PK, Smith EN, Ulivi S, Warrington NM, Zgaga L, Alavere H, Amin N, Aspelund T, Bandinelli S, Barroso I, Berenson GS, Bergmann S, Blackburn H, Boerwinkle E, Buring JE, Busonero F, Campbell H, Chanock SJ, Chen W, Cornelis MC, Couper D, Coviello AD, d’Adamo P, de Faire U, de Geus EJ, Deloukas P, Doring A, Smith GD, Easton DF, Eiriksdottir G, Emilsson V, Eriksson J, Ferrucci L, Folsom AR, Foroud T, Garcia M, Gasparini P, Geller F, Gieger C, Gudnason V, Hall P, Hankinson SE, Ferreli L, Heath AC, Hernandez DG, Hofman A, Hu FB, Illig T, Jarvelin MR, Johnson AD, Karasik D, Khaw KT, Kiel DP, Kilpelainen TO, Kolcic I, Kraft P, Launer LJ, Laven JS, Li S, Liu J, Levy D, Martin NG, McArdle WL, Melbye M, Mooser V, Murray JC, Murray SS, Nalls MA, Navarro P, Nelis M, Ness AR, Northstone K, Oostra BA, Peacock M, Palmer LJ, Palotie A, Pare G, Parker AN, Pedersen NL, Peltonen L, Pennell CE, Pharoah P, Polasek O, Plump AS, Pouta A, Porcu E, Rafnar T, Rice JP, Ring SM, Rivadeneira F, Rudan I, Sala C, Salomaa V, Sanna S, Schlessinger D, Schork NJ, Scuteri A, Segre AV, Shuldiner AR, Soranzo N, Sovio U, Srinivasan SR, Strachan DP, Tammesoo ML, Tikkanen E, Toniolo D, Tsui K, Tryggvadottir L, Tyrer J, Uda M, van Dam RM, van Meurs JB, Vollenweider P, Waeber G, Wareham NJ, Waterworth DM, Weedon MN, Wichmann HE, Willemsen G, Wilson JF, Wright AF, Young L, Zhai G, Zhuang WV, Bierut LJ, Boomsma DI, Boyd HA, Crisponi L, Demerath EW, van Duijn CM, Econs MJ, Harris TB, Hunter DJ, Loos RJ, Metspalu A, Montgomery GW, Ridker PM, Spector TD, Streeten EA, Stefansson K, Thorsteinsdottir U, Uitterlinden AG, Widen E, Murabito JM, Ong KK, Murray A (2010) Thirty new loci for age at menarche identified by a meta-analysis of genome-wide association studies. Nat Genet 42(12):1077–1085
Ellis BJ, Boyce WT, Belsky J, Bakermans-Kranenburg MJ, van Ijzendoorn MH (2011) Differential susceptibility to the environment: an evolutionary–neurodevelopmental theory. Dev Psychopathol 23(01):7–28
Essex MJ, Klein MH, Cho E, Kraemer HC (2003) Exposure to maternal depression and marital conflict: gender differences in children’s later mental health symptoms. J Am Acad Child Adolesc Psychiatry 42(6):728–737
Fraser A, Macdonald-Wallis C, Tilling K, Boyd A, Golding J, Davey Smith G, Henderson J, Macleod J, Molloy L, Ness A (2013) Cohort profile: the avon longitudinal study of parents and children: ALSPAC mothers cohort. Int J Epidemiol 42(1):97–110
Ge X, Natsuaki MN, Jin R, Biehl MC (2011) A contextual amplification hypothesis: Pubertal timing and girls’ emotional and behavioral problems. In: Kerr M, Stattin H, Engels RCME, Overbeek G, Andershed AK (eds) Understanding girls’ problem behavior: how girls’ delinquency develops in the context of maturity and health, co-occurring problems, and relationships. Wiley, Hoboken, pp 11–30
Goodman R (1997) The strengths and difficulties questionnaire: a research note. J Child Psychol Psychiatry 38(5):581–586
Goodman R, Meltzer H, Bailey V (1998) The strengths and difficulties questionnaire: a pilot study on the validity of the self-report version. Eur Child Adolesc Psychiatry 7(3):125–130
Grumbach MM, Styne DM (2003) Puberty: ontogeny, neuroendocrinology, physiology, and disorders. In: Larsen PR, Kronenberg HM, Melmed S, Polonsky KS (eds) Williams textbook of endocrinology. Elsevier, Amsterdam, pp 1115–1286
Harden KP, Hill JE, Turkheimer E, Emery RE (2008) Gene-environment correlation and interaction in peer effects on adolescent alcohol and tobacco use. Behav Genet 38(4):339–347
Horvath G, Knopik VS, Marceau K (2020) Polygenic influences on pubertal timing and tempo and depressive symptoms in boys and girls. J Res Adolesc 30(1):78–94
Hummel A, Shelton KH, Heron J, Moore L, van den Bree MBM (2013) A systematic review of the relationships between family functioning, pubertal timing and adolescent substance use. Addiction (Abingdon England) 108(3):487–496
Irner TB (2012) Substance exposure in utero and developmental consequences in adolescence: a systematic review. Child Neuropsychol 18(6):521–549
Johnson PO, Neyman J (1936) Tests of certain linear hypotheses and their application to some educational problems. Stat Res Memoirs 1:57–93
Jordan CJ, Andersen SL (2017) Sensitive periods of substance abuse: early risk for the transition to dependence. Dev Cogn Neurosci 25:29–44
Klump KL, Perkins PS, Burt SA, McGue M, Iacono WG (2007) Puberty moderates genetic influences on disordered eating. Psychol Med 37(5):627–634
Klump KL, Keel PK, Sisk C, Burt SA (2010) Preliminary evidence that estradiol moderates genetic influences on disordered eating attitudes and behaviors during puberty. Psychol Med 40(10):1745–1753
Klump KL, Culbert KM, Slane JD, Burt SA, Sisk CL, Nigg JT (2012) The effects of puberty on genetic risk for disordered eating: evidence for a sex difference. Psychol Med 42(3):627–637
Klump KL, Fowler N, Mayhall L, Sisk CL, Culbert KM, Burt SA (2018) Estrogen moderates genetic influences on binge eating during puberty: disruption of normative processes? J Abnorm Psychol 127(5):458
Legleye S, Karila L, Beck F, Reynaud M (2007) Validation of the CAST, a general population Cannabis abuse screening test. J Subst Use 12(4):233–242
Long JA (2019) Interactions: comprehensive, user-friendly toolkit for probing interactions
Maier R, Visscher P, Robinson M, Wray N (2018) Embracing polygenicity: a review of methods and tools for psychiatric genetics research. Psychol Med 48(7):1055–1067
Maisonet M, Christensen KY, Rubin C, Holmes A, Flanders WD, Heron J, Ong KK, Golding J, McGeehin MA, Marcus M (2010) Role of prenatal characteristics and early growth on pubertal attainment of british girls. Pediatrics 126(3):e591–e600
Marceau K, Neiderhiser J (2020) Generalist genes and specialist environments for adolescent internalizing and externalizing problems: a test of severity and directionality. Dev Psychopathol 34(1):379–386
Marceau K, Abar C, Jackson K (2015) Parental knowledge is a contextual amplifier of associations of pubertal maturation and substance use. J Youth Adolesc 44(9):1720–1734
Marceau K, Zahn-Waxler C, Shirtcliff EA, Schreiber JE, Hastings P, Klimes-Dougan B (2015) Adolescents’, mothers’, and fathers’ gendered coping strategies during conflict: youth and parent influences on conflict resolution and psychopathology. Dev Psychopathol 27(4pt1):1025–1044
Marceau K, Rolan E, Leve LD, Ganiban JM, Reiss D, Shaw DS, Natsuaki MN, Egger HL, Neiderhiser JM (2019) Parenting and prenatal risk as moderators of genetic influences on conduct problems during middle childhood. Dev Psychol 55(6):1164
Marceau K, Kirisci L, Tarter RE (2019) Correspondence of pubertal neuroendocrine and Tanner stage changes in boys and associations with substance use. Child Dev 90(6):e763–e782
Marceau K, Brick LA, Knopik VS, Reijneveld S (2020) Developmental pathways from genetic, prenatal, parenting and emotional/behavioral risk to cortisol reactivity and adolescent substance use: a TRAILS study. J Youth Adolesc 49:17–31
Marceau K, Nair N, Rogers ML, Jackson KM (2020) Lability in parent-and child-based sources of parental monitoring is differentially associated with adolescent substance use. Prev Sci 21(4):568–579
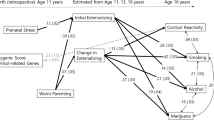
Marceau K, Horvath G, Loviska AM, Knopik VS (2021) Developmental cascades from polygenic and prenatal substance use to adolescent substance use: leveraging severity and directionality of externalizing and internalizing problems to understand pubertal and harsh discipline-related risk. Behav Genet 51:559–579
Marceau K, Brick LA, Pasman JA, Knopik VS, Reijneveld SA (2022) Interactions between genetic, prenatal, cortisol, and parenting influences on adolescent substance use and frequency: a TRAILS study. Eur Addict Res 28(3):176–185
Marceau K, Yu L, Knopik VS, Ganiban JM, Neiderhiser JM (2022) Intergenerational transmission of psychopathology: an examination of symptom severity and directionality. Dev Psychopathol 34(5):1767–1780
McNeil TF, Cantor-Graae E, Sjöström K (1994) Obstetric complications as antecedents of schizophrenia: empirical effects of using different obstetric complication scales. J Psychiatry Res 28(6):519–530
Mendle J, Ferrero J (2012) Detrimental psychological outcomes associated with pubertal timing in adolescent boys. Dev Rev 32(1):49–66
Mendle J, Turkheimer E, Emery RE (2007) Detrimental psychological outcomes associated with early pubertal timing in adolescent girls. Dev Rev 27(2):151–171
Molnar DS, Levitt A, Eiden RD, Schuetze P (2014) Prenatal cocaine exposure and trajectories of externalizing behavior problems in early childhood: examining the role of maternal negative affect. Dev Psychopathol 26(02):515–528
Morie KP, Crowley MJ, Mayes LC, Potenza MN (2019) Prenatal drug exposure from infancy through emerging adulthood: results from neuroimaging. Drug Alcohol Depend. https://doi.org/10.1016/j.drugalcdep.2019.01.032. (Drug Alcohol Depend 19839–19853)
Morris NM, Udry JR (1980) Validation of a self-administered instrument to assess stage of adolescent development. J Youth Adolesc 9(3):271–280
Munn-Chernoff MA, Johnson EC, Chou YL, Coleman JR, Thornton LM, Walters RK, Yilmaz Z, Baker JH, Hübel C, Gordon S (2021) Shared genetic risk between eating disorder‐and substance‐use‐related phenotypes: evidence from genome‐wide association studies. Addict Biol 26(1):e12880
Neiderhiser JM, Marceau K, De Araujo-Greecher M, Ganiban JM, Mayes LC, Shaw DS, Reiss D, Leve LD (2016) Estimating the roles of genetic risk, perinatal risk, and marital hostility on early childhood adjustment: medical records and self-reports. Behav Genet 46(3):334–352
Pasman JA, Smit K, Vollebergh WA, Nolte IM, Hartman CA, Abdellaoui A, Verweij KJ, Maciejewski D, Vink JM (2021) Interplay between genetic risk and the parent environment in adolescence and substance use in young adulthood: a TRAILS study. Dev Psychopathol 35(1):396–409
Porath AJ, Fried PA (2005) Effects of prenatal cigarette and marijuana exposure on drug use among offspring. Neurotoxicol Teratol 27(2):267–277
Prom-Wormley EC, Ebejer J, Dick DM, Bowers MS (2017) The genetic epidemiology of substance use disorder: a review. Drug Alcohol Depend 180:241–259
Purcell S, Neale B, Todd-Brown K, Thomas L, Ferreira MA, Bender D, Maller J, Sklar P, De Bakker PI, Daly MJ (2007) PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 81(3):559–575
Rhee SH, Hewitt JK, Young SE, Corley RP, Crowley TJ, Stallings MC (2003) Genetic and environmental influences on substance initiation, use, and problem use in adolescents. Arch Gen Psychiatry 60(12):1256–1264
Rosseel Y (2012) lavaan: an R package for structural equation modeling. J Stat Softw 48(2):36
Salbach-Andrae H, Klinkowski N, Lenz K, Lehmkuhl U (2009) Agreement between youth-reported and parent-reported psychopathology in a referred sample. Eur Child Adolesc Psychiatry 18(3):136–143
Saunders JB, Aasland OG, Babor TF, De la Fuente JR, Grant M (1993) Development of the alcohol use disorders identification test (AUDIT): WHO collaborative project on early detection of persons with harmful alcohol consumption-II. Addiction 88(6):791–804
Ullsperger JM, Nikolas MA (2017) A meta-analytic review of the association between pubertal timing and psychopathology in adolescence: are there sex differences in risk? Psychol Bull 143(9):903
Zuckerman M (1999) Diathesis-stress models. American Psychological Association, Washington
Acknowledgements
We are extremely grateful to all the families who took part in this study, the midwives for their help in recruiting them, and the whole ALSPAC team, which includes interviewers, computer and laboratory technicians, clerical workers, research scientists, volunteers, managers, receptionists and nurses.
Funding
The UK Medical Research Council and Wellcome (Grant ref: 217065/Z/19/Z) and the University of Bristol provide core support for ALSPAC. This publication is the work of the authors, who will serve as guarantors for the contents of this paper. A comprehensive list of grants funding is available on the ALSPAC website (http://www.bristol.ac.uk/alspac/external/documents/grant-acknowledgements.pdf). GWAS data was generated by Sample Logistics and Genotyping Facilities at Wellcome Sanger Institute and LabCorp (Laboratory Corporation of America) using support from 23andMe. This research was specifically funded by the National Institute on Drug Abuse (K01 DA039288, Marceau).
Author information
Authors and Affiliations
Contributions
KM conceived of the study, analyzed the data, and drafted the manuscript. GH and AL were instrumental in construction of key study variables. VK was instrumental in conceptualizing the study. All authors edited the manuscript and approved of the final manuscript prior to submission.
Corresponding author
Ethics declarations
Conflict of interest
Kristine Marceau, Amy M. Loviska, Gregor Horvath and Valerie S. Knopik declare that they have no conflict of interest.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Ethical approval for the study was obtained from the ALSPAC Ethics and Law Committee and the Local Research Ethics Committees. Ethical approval for the present data analysis was also obtained from the Purdue University IRB.
Informed Consent
Informed consent for the use of data collected via questionnaires and clinics was obtained from participants following the recommendations of the ALSPAC Ethics and Law Committee at the time.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Edited by Stephen Petrill.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Marceau, K., Loviska, A., Horvath, G. et al. Interactions Between Genetic, Prenatal Substance Use, Puberty, and Parenting are Less Important for Understanding Adolescents’ Internalizing, Externalizing, and Substance Use than Developmental Cascades in Multifactorial Models. Behav Genet 54, 181–195 (2024). https://doi.org/10.1007/s10519-023-10164-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10519-023-10164-9