Abstract
Purpose of Review
In this review, the impact of obesity on cardiovascular disease in women and emerging anti-obesity pharmacologic treatments are discussed.
Recent Findings
Robust evidence demonstrates the burden of obesity across the lifespan in women and links obesity to a diverse set of cardiovascular diseases. Female-specific risk factors including sex hormones and pregnancy factors intersect with obesity and cardiovascular risk. Sustained weight loss has potential for cardiovascular benefits. Recent trials demonstrate cardiovascular benefits of emerging agents with weight loss effects including GLP-1 RA and SGLT2 inhibitors in women.
Summary
Treatment and prevention strategies for cardiovascular disease in obese women should include integration of weight management strategies including the targeted use of emerging pharmacologic therapies.
Similar content being viewed by others
Introduction
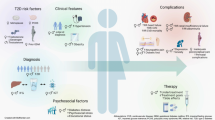
Obesity contributes to the development of a diverse set of cardiovascular diseases. Understanding the burden of obesity and heart disease in women is critical to prevention efforts [1]. Women have high rates of obesity associated with a sex-based accentuation of cardiovascular risk. Importantly, obesity impacts women across the lifespan including in youth, pregnancy, the midlife transition, and in older age (Fig. 1) [2]. In the USA, more than 40% of adult women are obese [3, 4]. Worldwide, obesity leads to 2 million deaths in women, largely related to cardiovascular causes [5, 6]. Health disparities exist in obesity rates among women and in the presence of metabolically unhealthy obesity [7,8,9]. Chronic inflammation in obesity drives adverse systemic processes including vascular dysfunction, insulin resistance, metabolic dysregulation, hypertension, and fibrosis [10].
Shared pathophysiologic mechanisms between obesity and concomitant cardiometabolic conditions including type 2 diabetes (T2DM), hypercholesterolemia, chronic kidney disease, and hypertension suggest that similar therapies may be beneficial. Treatments initially developed for T2DM including glucagon-like peptide-1 receptor agonist (GLP-1 RA) medications and sodium-glucose transporter 2 (SGLT2) inhibitors now have proven benefits for obesity-related cardiometabolic diseases beyond glucose lowering. The intersection of obesity and cardiovascular disease in women heightens the importance of cardiovascular clinicians in the multidisciplinary management of weight loss therapies. The current review discusses obesity-related cardiovascular disease in women with a focus on emerging medication treatments to restore cardiometabolic health and promote weight loss.
Impact of Obesity Across the Life Course in Women
Epidemiology of Obesity in Women
Obesity has a disproportionate influence in women from youth to old age. Most recent US estimates indicate that 18% of female children and 41% of adult women are obese, of whom 11.6% have severe obesity [7]. Rates are higher in both youth and adult women who are Black compared to Non-Hispanic White, Hispanic, and Asian women [8]. Rural residence and a higher burden of unfavorable social determinants of health associate with higher obesity prevalence in women [11, 12]. Increases in severe obesity rates over the past 20 years are higher in women than men [13, 14]. Reflecting underlying population trends, pre-pregnancy obesity rates are also rising with close to a third of people being obese prior to pregnancy in 2019 [15]. During the menopausal period, women develop both higher overall body mass index (BMI) and a shift toward central obesity, a more metabolically toxic fat distribution pattern. The prevalence of obesity remains elevated in older women who are also at the greatest risk of cardiovascular diseases including atrial fibrillation, heart failure, and atherosclerosis.
Pathophysiologic Connections of Obesity and Cardiovascular Disease in Women
A potential conceptual model linking obesity to cardiovascular disease in women includes higher prevalence of cardiometabolic risk factors that contribute to systemic inflammation, vascular dysfunction, and myocardial remodeling [16,17,18,19]. In addition, excess adipose tissue may produce factors that alter cardiovascular homeostasis [20•]. Genetic factors as well as gene-environment interaction are increasingly recognized as important drivers of obesity and adverse metabolic state [21, 22].
Sex differences in both traditional risk factors along with female-specific risk factors may be contributing to the accelerated risk of cardiovascular disease in obese women (Fig. 1) [23]. T2DM appears to confer a stronger risk of cardiovascular disease in women [24,25,26,27]. Obesity attenuates the relative protection from hypertension in younger women [28]. Fat distribution differs by sex with more visceral adiposity in women along with sex-based differences in the metabolic activity of ectopic fat deposits [20, 29,30,31,32,33,34]. Sex hormones, including estrogen, regulate adipose tissue biology, leading to a more unhealthy phenotype in the postmenopausal period [35]. Age-related arterial stiffening is greater in women as is insulin-resistance driven microvascular dysfunction [36,37,38,39,40]. Sex hormones also contribute to endothelial function with a greater decline in vasomotor function in women following menopause [41, 42]. Additional factors related to reproductive health may also contribute to sex-specific obesity risks including hypertensive disorders in pregnancy, infertility, and other pregnancy complications [43, 44].
Association of Obesity and Weight Loss with Cardiovascular Diseases in Women
Obesity and Hypertension in Women
Obesity accelerates the age-related rise in blood pressure [45, 46]. Overall, the presence of obesity is associated with a marked increased risk of incident hypertension in women [28]. Weight gain accelerates age-related increases in blood pressure [47]. Changes in the proportion of estrogen and androgens in the presence of visceral adiposity may also be important in driving obesity-related hypertension in women [16, 48]. A recent study focused on the menopause transition showed an association of higher BMI with both a pattern accelerated change in menopause and with a high blood pressure in the premenopausal period [49••]. Sex-based differences have also been demonstrated in vasodilator function particularly related to altered mineralocorticoid regulation in the endothelium [50, 51]. Weight loss with bariatric surgery produces marked improvement in blood pressure in the early period with attenuation over time [52]. Appropriate treatment of blood pressure is important in the overall management of women with obesity and hypertension.
Obesity and Coronary Heart Disease and Peripheral Artery Disease in Women
A recent umbrella review of meta-analyses defined the risk of coronary heart disease with adiposity [53••]. An increase of BMI by 1 kg/m2 was associated with 4% higher risk of coronary disease in women compared to 6% in men. The presence of obesity in women was associated with a 1.58-fold higher risk of coronary heart disease. Obesity increases the lifetime risk of coronary disease by 85% in women with a 2.53-fold increase for severe obesity. PAD is more prevalent in women with obesity [54]. One study showed that BMI > 35 was associated with 1.35 odds of PAD and 2.98 odds of PAD for BMI > 40 in women suggesting that increasing BMI may be a strong independent risk factor for PAD in women [55]. Mendelian randomization confirms a causal association of adiposity with coronary disease indicating an overall 1.19 relative risk for each 5 kg/m2 increase in genetically predicted BMI [53••]. In addition to conduit vessel atherogenesis, excess adipose tissue may have particular adverse consequences in the microvasculature, a determinant of angina without obstructive coronary disease [56]. About half of the genetic-associated cardiovascular risk related to obesity is mediated by metabolic risk factors including lipids, hypertension, and T2DM [57]. A comprehensive assessment of circulating biomarkers demonstrated higher inflammatory markers and adipokines in women as compared to men [58•]. The constellation of associated risk factors with obesity emphasizes the importance of a comprehensive approach to therapies to mitigate atherosclerotic risk [59, 60]. In patients with T2DM, the rate of achieving risk factor control is lower in women as compared to men [61].
Weight fluctuations portend higher cardiovascular disease risk [62]. Dietary approaches to weight loss generally do not significantly alter coronary heart disease most likely related to the challenges of sustaining the weight loss [63]. In severe obesity, bariatric surgery has been shown to significantly decrease incident coronary artery disease [64, 65]. The greater impact of surgical weight loss is likely due to the lower degree of weight loss with lifestyle modifications vs. surgical interventions (5–10 kg vs. 10–40 kg) [66]. Also, sustained weight loss is more likely with surgical intervention which is important for achieving the beneficial effects of weight loss on atherosclerotic disease.
Obesity and Heart Failure in Women
Both longitudinal observational and genetic studies indicate a causal association of obesity with incident heart failure in women [16, 67,68,69,70] failure in patients across the entire BMI spectrum which showed an increase in the prevalence of heart failure by 5% in men and 7% in women for each 1 kg/m2 increase in BMI [67]. Sex-differences are more pronounced in heart failure with preserved ejection fraction (HFpEF) [71, 72]. In a study of 4 community-based cohorts in the USA, obesity was most strongly associated with HFpEF in women as compared with men [70]. In the Women’s Health Initiative, obesity was an even more potent risk factor for HFpEF in Black American women compared with White women [73]. Inflammation, microvascular dysfunction, and insulin resistance are mechanisms common to obesity and HFpEF that may underlie sex-specific risk [70, 74,75,76,77,78]. Visceral adiposity, that is greater in women particularly following menopause, is associated with left ventricular remodeling and altered myocardial metabolism [79, 80]. Excess visceral adipose tissue was associated with altered exercise hemodynamics in women but not men with HFpEF [81•]. A recent meta-analysis suggested that in women, waist circumference and waist-to-hip ratio were superior predictors of heart failure risk in women pointing to a role for visceral adiposity [82]. Metabolic risk factors that travel with obesity are key drivers of heart failure risk in women consistent with the theme of comprehensive metabolic interventions [83].
In a bariatric surgery study including 77% women, surgical weight loss reduced heart failure rates compared to lifestyle intervention with a hazard ratio of 0.54 with a dose–response relationship between the degree of weight loss at 1 year [66]. In patients with pre-existing CVD, bariatric surgery lowered the rate of recurrent events in women with a HR of 0.6 [84]. New medications with impact on T2DM, obesity, and heart failure will be discussed in detail below.
Obesity and Atrial Fibrillation in Women
Longitudinal studies confirm the importance of obesity as a risk factor for atrial fibrillation accounting for up to one-fifth of atrial fibrillation cases [85,86,87]. In the Women’s Heart Study, higher BMI was associated with heightened atrial fibrillation [85]. Obesity is associated with electrical remodeling in the atrial tissue including fibrosis and fat infiltration [88]. Measures of cardiac remodeling mediate the association of BMI with atrial fibrillation [19]. The association of BMI with atrial fibrillation is enhanced in women [89•].
Multiple randomized studies have evaluated the effect of weight loss on atrial fibrillation burden [90,91,92]. Consistently, weight management strategies combined with risk factor control reduce atrial fibrillation burden as well as improve results after ablation [93].
Obesity and Venous Thromboembolism in Women
Obesity is a pro-inflammatory state, promoting a prothrombotic state. Prior studies show that obesity increases the risk of the first episode of venous thromboembolism (VTE) by 2.33 (95% CI, 1.68–3.24) [94]. Multiple adiposity measures including BMI, waist circumference, and fat percentage were associated with VTE risk in women but men [95••]. Epidemiologic study indicates that up to a quarter of VTE is attributable to elevated BMI in both women and men [96]. Moreover, obesity can increase the risk for VTE recurrence, which may be higher in women compared to men [97]. Hormonal treatments including oral contraceptives and hormone replacement therapy increase VTE risk that is enhanced with concomitant obesity [98]. In addition, VTE incidence is elevated during pregnancy, accounting for 9% of deaths during pregnancy in the USA [99, 100]. Higher BMI is an important risk factor for post pregnancy-related VTE [101].
Obesity and Adverse Pregnancy Outcomes
With the rising prevalence of pre-pregnancy obesity, adverse pregnancy outcomes related to obesity are an important contributor to peri-pregnancy and long-term health in women [15, 44]. There are racial and ethnic disparities in the prevalence of pre-pregnancy obesity [102]. Both gestational diabetes and hypertension disorders of pregnancy have higher incidence with increasing BMI [103, 104]. Furthermore, high-risk obesity characterized by higher waist circumference confers a higher risk of adverse pregnancy outcomes [105•]. Robust epidemiologic evidence links adverse pregnancy outcomes to future development of cardiovascular risk factors and cardiovascular disease [106, 107]. A strategy to promote weight loss after pregnancy could reduce the direct and indirect risks of CVD [108]. A recent American Heart Association Scientific Statement addresses the importance of pre-pregnancy evaluation of obesity [44, 109].
Approaches to Obesity Treatment in Women with CVD
Overview of Anti-Obesity Strategies in Women with CVD
Treatment of obesity has the potential to improve cardiovascular risk factors and reduce the risk of cardiovascular events. Regular evaluation of BMI, cardiovascular risk factors, CVD, and readiness for weight loss is recommended. For individuals with BMI ≥ 30 or > 27 with CVD or risk factors, comprehensive lifestyle therapies are the first step in treatment [110, 111]. For individuals with BMI ≥ 40 or > 35 with CVD, evaluation for bariatric surgery is considered. For all patients with inadequate response to lifestyle therapy or with concomitant CVD, pharmacologic therapies can be considered. The backbone of all initial obesity therapies includes lifestyle and behavioral approaches including nutritional counseling and appropriate exercise augmentation. Similarly, weight loss is recommended for prevention of CVD using counseling and lifestyle approaches.
Studies suggest that to achieve cardiovascular benefits from weight loss in obesity, losing > 10% of body weight is needed [112]. Lifestyle changes with diet and physical activity may result in inadequate weight loss and may be complicated by weight regain [113]. Long-term studies have shown that bariatric surgical procedures typically lead to a sustainable weight loss of 25% and improvements in cardiovascular outcomes [65]. However, surgical approaches are an invasive method that is mainly reserved for cases of severe obesity [111].
There have been recent advances in the development of anti-obesity medications that are altering the treatment landscape. Studies with novel pharmacotherapy approaches for the treatment of obesity are now showing the degree of weight loss that may be relevant to altering CVD course. The focus of the following sections is on recent developments in medication treatment for obesity that is relevant to women with CVD. Earlier medications including orlistat, phentermine, lorcaserin, and naltrexone/buproprion have been reviewed previously, thus they are not the focus of the current review [114, 115].
Glucagon-Like Peptide-1 Receptor Agonists and CVD in Women
Glucagon-like peptide-1 receptor agonist (GLP-1 RA) medications were developed and approved as glucose lowering agents in patients with T2DM [116]. Clinical trials in patients with T2DM demonstrated modest weight loss with all GLP-1 RA as shown in Table 1. The degree of weight loss seems largely related to dosing but may also reflect the extent of brain uptake and action. Both liraglutide and semaglutide have been approved for weight loss based on a series of trials in patients with obesity (Table 1). In the STEP trials, semaglutide resulted in an average weight loss of 12% by week 28 [117••]. The combination of liraglutide with an exercise intervention had enhanced efficacy for maintaining weight loss [118]. A newer T2DM treatment agent, tirzepatide, that combines GLP-1 RA with glucose-dependent insulinotropic polypeptide (GIP) activity, led to a greater degree of weight loss [119••]. In patients with BMI ≥ 30 or ≥ 27 with a risk factor, tirzepatide led to up to 20.9% weight loss with the highest dose.
Given the FDA requirement to evaluate cardiovascular safety of T2DM treatments, there have been dedicated cardiovascular studies for all GLP-1 RA medications [140]. All the cardiovascular outcome studies evaluated composite outcomes of MACE (cardiovascular death, myocardial infarction, and stroke) and enrolled patients with T2DM with a varying prevalence of pre-existing CVD. As shown in Table 1, selected GLP-1 RA’s demonstrated superiority compared to placebo for reduction of MACE. Meta-analyses confirm the benefits of GLP-1 RA’s in reducing individual endpoints including myocardial infarction, stroke, cardiovascular mortality, and all-cause mortality with only moderate heterogeneity across the agents [141, 142]. Among patients with T2DM, the benefit appeared similar for MACE events in individuals with established CVD compared to high burden of risk factors [142]. There was also a suggestion of reduction in hospitalization for heart failure though numbers of events were low and in heart failure with reduced ejection fraction, liraglutide did not show benefits [141]. It seems possible that a greater extent of cardiovascular benefit from GLP-1 RA medications is from the impact on atherosclerotic events. A recent trial (AMPLITUDE-O) looked at the effect of GLP-1 RA, efpeglenatide, for reducing the risk of cardiovascular events in patients with T2DM and CVD and was shown beneficial in reducing MACE compared to placebo [143]. The ongoing SELECT trial is evaluating semaglutide in patients with established CVD without T2DM and preliminary favorable results were reported in a press release [127]. Additional ongoing studies include in patients with atrial fibrillation as well as cardiovascular outcomes with tirzepatide. Multiple potential mechanisms have been proposed for the GLP-1 RA medications including direct vascular effects, alteration of risk factor profile, reduction of inflammation, and plaque stabilization [144,145,146].
Inclusion of women in the cardiovascular outcome trials of GLP-1 RA medications in patients with T2DM ranged from 30 to 46% and reporting on sex-specific results has not been consistent. One meta-analyses indicated similar benefits in women with T2DM with a HR of 0.88 (0.79–0.99, P = 0.03) for MACE compared to placebo [147••]. Another study using registry medication data sources suggested a higher degree of relative risk reduction with GLP-1 RA in women compared to men [148]. In the obesity trials with GLP-1 RA’s, the proportion of women has been higher consistent with many studies of weight loss interventions. In the STEP studies, more than 70% were women, but there is limited information about sex-specific effects. In the SURMOUNT-1 trial with tirzepatide, women comprised 67% of the study participants, but no information was provided in the initial report regarding sex-specific weight loss effects.
The most common adverse effects of GLP-1 RA medications include nausea, vomiting, and diarrhea [149]. In many patients, the intensity is mitigated by gradual dose escalation. The safety during pregnancy is not known and thus not recommended to be used during pregnancy. In general for both T2DM and for obesity management, GLP-1 RA represents a chronic therapeutic agent with evidence of weight regain when the medications are stopped.
Sodium-Glucose Transporter 2 Inhibitors and CVD in Women
Similar to GLP-1 RA medications, sodium-glucose transporter 2 (SGLT2) inhibitors were developed for treatment of T2DM and have expanded into cardiovascular health impact. The role in obesity management is more variable [150, 151]. In trials of patients with T2DM, SGLT2 inhibitors reduce body weight by 1.5–2 kg in a dose-dependent manner that appears maintained over 4 years [152•]. Weight loss with SGLT2 inhibitors appears similar for patients without T2DM [153]. The SGLT2 inhibitor-induced weight loss is much more pronounced when combined with GLP-1 RA suggesting that the combination of glycosuria with appetite suppression may have additive benefits in patients with T2DM [154, 155].
There is robust evidence supporting the use of SGLT2 inhibitors to reduce cardiovascular risk in patients with T2DM and for the treatment of heart failure. Multiple agents demonstrate favorable cardiovascular outcome trials of SGLT2 inhibition in patients with T2DM as shown in Table 1. In meta-analyses, there is reduction of MACE only in patients with established atherosclerotic disease and heart failure outcomes in those with and without established CVD [156•]. A subsequent meta-analysis suggested primary MACE prevention in patients with CKD [157]. Subsequent trials have shown benefit in stable patients with heart failure with reduced and with preserved ejection fraction as well as in patients with chronic kidney disease (Table 1). Studies have shown that SGLT2 inhibitors improve cardiovascular outcomes and HF symptoms in patients with heart failure and preserved ejection fraction (HFpEF) and obesity, and this improvement is accompanied by weight loss [158]. Ongoing trials are evaluating outcomes with unstable coronary disease along with heart failure.
In patients with T2DM, the CV benefits of SGLT2 inhibitors appear similar in women compared to men. In a patient level meta-analysis of the dapagliflozin trials in heart failure for both reduced and preserved ejection fraction, there were no observed sex-based differences in the treatment benefits with a HR for women of 0.74 (0.66–0.84) [159]. Current guideline recommendations for both T2DM with CVD and for heart failure include the use of SGLT2 inhibitors [160]. Potential adverse effects of SGLT2 inhibitors include urinary tract infections and genital mycotic infections in women. The impact during pregnancy is not known and, thus, not recommended. Prior reports suggesting an increase in amputation risk have largely been refuted by more recent evidence suggesting safety in patients with peripheral artery disease [161]. It is important to note that SGLT2 inhibitors are not approved for weight management but for treatment of T2DM and heart failure though the combination with GLP-1 RA for patients with T2DM may augment weight loss.
Incorporating the Treatment of Obesity in Cardiovascular Care of Women
Access to effective weight loss management strategies remains low even among patients with established CVD who may gain the greatest clinical benefits. Barriers include higher cost of medication therapies as well as fragmentation of care of patients with obesity. Multidisciplinary cardiometabolic management will be necessary to augment guideline-based use as appropriate. In the setting of cardiovascular practice, it is important to evaluate BMI for all patients as well as assess readiness for weight management strategies [162]. Cardiovascular clinicians are often the primary provider for patients with obesity and CVD [163•]. For patients with T2DM, the most recent ADA guidelines recommend GLP-1 RA medications added to metformin for patients with established atherosclerotic CVD or high risk indicators for CVD [164]. In patients with chronic coronary artery disease and T2DM, current AHA/ACC guidelines recommend GLP-1 RA or SGLT2 inhibitors as Class I [165]. Currently, estimates of implementation based on these recent guidelines remain low [166]. Weight reduction is greater with GLP-1 RA as compared to SGLT2 inhibitors [167]; however, patients with heart failure and chronic kidney disease have more direct benefit from SGLT2 inhibitors [156, 168]. Combination therapy with both agents may also be used in patients with need for heart failure, weight loss, and kidney protection [169, 170].
A recent study shows increasing prevalence of cardiologists prescribing SGLT-2 inhibitors (12-fold increase) and GLP-1 Ras (fourfold) from 2015 to 2020; however, overall total use by cardiologists remains low [171]. Incorporation of approaches to address cost concerns is also critical to prevent accentuation of health disparities [172]. Inclusion of anti-obesity medication therapies as a strategy for secondary prevention is an emerging facet of comprehensive cardiovascular care.
Conclusions
The global rise in obesity prevalence is a major risk public health problem that affects women throughout their lifespan. Despite the strong association of obesity and CVD and the effect of weight loss on improving cardiovascular outcomes, obesity remains an undertreated condition. This review emphasizes the cardiovascular benefits of emerging therapies with an impact on obesity and approaches to incorporate use in appropriate patients with CVD. Ongoing clinical trials will provide additional information about the cardiovascular health impacts of GLP-1 RA and SGLT2 inhibitors as well as direct impact in patients with established CVD. Overall, there is robust evidence to support the inclusion of pharmacologic agents for weight management in the care of women with cardiovascular diseases.
References
Papers of particular interest, published recently, have been highlighted as: •Of importance •• Of major importance
Vogel B, Acevedo M, Appelman Y, Bairey Merz CN, Chieffo A, Figtree GA, et al. The Lancet women and cardiovascular disease commission: reducing the global burden by 2030. Lancet. 2021;397:2385–438. https://doi.org/10.1016/S0140-6736(21)00684-X.
Boutari C, Mantzoros CS. A 2022 update on the epidemiology of obesity and a call to action: as its twin COVID-19 pandemic appears to be receding, the obesity and dysmetabolism pandemic continues to rage on. Metabolism. 2022;133:155217. https://doi.org/10.1016/j.metabol.2022.155217.
Tsao CW, Aday AW, Almarzooq ZI, Anderson CAM, Arora P, Avery CL, et al. Heart disease and stroke statistics-2023 update: a report from the American Heart Association. Circulation. 2023;147:e93–621. https://doi.org/10.1161/CIR.0000000000001123.
Centers for Disease Control Obesity Data. 2023. https://www.cdc.gov/obesity/data/adult.html. Accessed 14 Aug.
Lin X, Xu Y, Xu J, Pan X, Song X, Shan L, et al. Global burden of noncommunicable disease attributable to high body mass index in 195 countries and territories, 1990–2017. Endocrine. 2020;69:310–20. https://doi.org/10.1007/s12020-020-02352-y.
GBD 2015 Obesity Collaborators, Afshin A, Forouzanfar MH, Reitsma MB, Sur P, Estep K, et al. Health effects of overweight and obesity in 195 countries over 25 years. N Engl J Med. 2017;377:13–27. https://doi.org/10.1056/NEJMoa1614362.
Hales CM, Carroll MD, Fryar CD, Ogden CL. Prevalence of obesity and severe obesity among adults: United States, 2017–2018. NCHS Data Brief. 2020:1–8.
Ogden CL, Fryar CD, Martin CB, Freedman DS, Carroll MD, Gu Q, et al. Trends in obesity prevalence by race and Hispanic origin-1999-2000 to 2017–2018. JAMA. 2020;324:1208–10. https://doi.org/10.1001/jama.2020.14590.
Hales CM, Fryar CD, Carroll MD, Freedman DS, Aoki Y, Ogden CL. Differences in obesity prevalence by demographic characteristics and urbanization level among adults in the United States, 2013–2016. JAMA. 2018;319:2419–29. https://doi.org/10.1001/jama.2018.7270.
Wu H, Ballantyne CM. Metabolic inflammation and insulin resistance in obesity. Circ Res. 2020;126:1549–64. https://doi.org/10.1161/CIRCRESAHA.119.315896.
• Javed Z, Valero-Elizondo J, Maqsood MH, Mahajan S, Taha MB, Patel KV, et al. Social determinants of health and obesity: findings from a national study of US adults. Obesity (Silver Spring). 2022;30:491–502. https://doi.org/10.1002/oby.23336. This paper demonstrates the graded associated of social determinants with overweight and obesity across multiple racial groups in the United States highlighting the intersection of sex, race, and structural disadvantage in the obesity epidemic.
Cuevas AG, Chen R, Slopen N, Thurber KA, Wilson N, Economos C, et al. Assessing the role of health behaviors, socioeconomic status, and cumulative stress for racial/ethnic disparities in obesity. Obesity (Silver Spring). 2020;28:161–70. https://doi.org/10.1002/oby.22648.
Roth GA, Mensah GA, Johnson CO, Addolorato G, Ammirati E, Baddour LM, et al. Global burden of cardiovascular diseases and risk factors, 1990–2019: Update From the GBD 2019 Study. J Am Coll Cardiol. 2020;76:2982–3021. https://doi.org/10.1016/j.jacc.2020.11.010.
Hales CM, Fryar CD, Carroll MD, Freedman DS, Ogden CL. Trends in obesity and severe obesity prevalence in US youth and adults by sex and age, 2007–2008 to 2015–2016. JAMA. 2018;319:1723–5. https://doi.org/10.1001/jama.2018.3060.
Driscoll AK, Gregory E. Increases in prepregnancy obesity: United States 2016–2019. NCHS Data Brief. 2020.
Faulkner JL. Obesity-associated cardiovascular risk in women: hypertension and heart failure. Clin Sci (Lond). 2021;135:1523–44. https://doi.org/10.1042/CS20210384.
Cooper AJ, Gupta SR, Moustafa AF, Chao AM. Sex/gender differences in obesity prevalence, comorbidities, and treatment. Curr Obes Rep. 2021;10:458–66. https://doi.org/10.1007/s13679-021-00453-x.
Mouton AJ, Li X, Hall ME, Hall JE. Obesity, hypertension, and cardiac dysfunction: novel roles of immunometabolism in macrophage activation and inflammation. Circ Res. 2020;126:789–806. https://doi.org/10.1161/CIRCRESAHA.119.312321.
Szabo L, McCracken C, Cooper J, Rider OJ, Vago H, Merkely B, et al. The role of obesity-related cardiovascular remodelling in mediating incident cardiovascular outcomes: a population-based observational study. Eur Heart J Cardiovasc Imaging. 2023;24:921–9. https://doi.org/10.1093/ehjci/jeac270.
• Kammerlander AA, Lyass A, Mahoney TF, Massaro JM, Long MT, Vasan RS, et al. Sex differences in the associations of visceral adipose tissue and cardiometabolic and cardiovascular disease risk: the Framingham Heart Study. J Am Heart Assoc. 2021;10:e019968. https://doi.org/10.1161/JAHA.120.019968. A longitudinal analysis demonstrating that in women visceral adiposity was a stronger predictor than BMI of both incident cardiometabolic risk factors and cardiovascular disease suggesting that body composition is important to obesity and CVD in women.
Khera AV, Chaffin M, Wade KH, Zahid S, Brancale J, Xia R, et al. Polygenic prediction of weight and obesity trajectories from birth to adulthood. Cell. 2019;177(587–596):e589. https://doi.org/10.1016/j.cell.2019.03.028.
Sakaue S, Kanai M, Karjalainen J, Akiyama M, Kurki M, Matoba N, et al. Trans-biobank analysis with 676,000 individuals elucidates the association of polygenic risk scores of complex traits with human lifespan. Nat Med. 2020;26:542–8. https://doi.org/10.1038/s41591-020-0785-8.
Kaur G, Lau E. Sex differences in heart failure with preserved ejection fraction: from traditional risk factors to sex-specific risk factors. Womens Health (Lond). 2022;18:17455057221140208. https://doi.org/10.1177/17455057221140209.
Regensteiner JG, Golden S, Huebschmann AG, Barrett-Connor E, Chang AY, Chyun D, et al. Sex differences in the cardiovascular consequences of diabetes mellitus: a scientific statement from the American Heart Association. Circulation. 2015;132:2424–47. https://doi.org/10.1161/CIR.0000000000000343.
Ohkuma T, Komorita Y, Peters SAE, Woodward M. Diabetes as a risk factor for heart failure in women and men: a systematic review and meta-analysis of 47 cohorts including 12 million individuals. Diabetologia. 2019;62:1550–60. https://doi.org/10.1007/s00125-019-4926-x.
• Du T, Fernandez C, Barshop R, Guo Y, Krousel-Wood M, Chen W, et al. Sex differences in cardiovascular risk profile from childhood to midlife between individuals who did and did not develop diabetes at follow-up: the Bogalusa Heart Study. Diabetes Care. 2019;42:635–43. https://doi.org/10.2337/dc18-2029. Longitudinal study showing that higher cumulative exposure to cardiometabolic risk factors including high BMI in women predicts risk of diabetes mellitus.
de Jong M, Woodward M, Peters SAE. Diabetes, glycated hemoglobin, and the risk of myocardial infarction in women and men: a prospective cohort study of the UK Biobank. Diabetes Care. 2020;43:2050–9. https://doi.org/10.2337/dc19-2363.
Forman JP, Stampfer MJ, Curhan GC. Diet and lifestyle risk factors associated with incident hypertension in women. JAMA. 2009;302:401–11. https://doi.org/10.1001/jama.2009.1060.
Krotkiewski M, Bjorntorp P, Sjostrom L, Smith U. Impact of obesity on metabolism in men and women. Importance of regional adipose tissue distribution. J Clin Invest. 1983;72:1150–62. https://doi.org/10.1172/JCI111040.
White UA, Tchoukalova YD. Sex dimorphism and depot differences in adipose tissue function. Biochim Biophys Acta. 2014;1842:377–92. https://doi.org/10.1016/j.bbadis.2013.05.006.
• Chen GC, Arthur R, Iyengar NM, Kamensky V, Xue X, Wassertheil-Smoller S, et al. Association between regional body fat and cardiovascular disease risk among postmenopausal women with normal body mass index. Eur Heart J. 2019;40:2849–55. https://doi.org/10.1093/eurheartj/ehz391. Substudy of Womens Health Initiative with body composition by imaging showing that higher trunk fat associated with CVD risk in postmenopausal women.
Porter JW, Barnas JL, Welly R, Spencer N, Pitt J, Vieira-Potter VJ, et al. Age, sex, and depot-specific differences in adipose-tissue estrogen receptors in individuals with obesity. Obesity (Silver Spring). 2020;28:1698–707. https://doi.org/10.1002/oby.22888.
Pou KM, Massaro JM, Hoffmann U, Vasan RS, Maurovich-Horvat P, Larson MG, et al. Visceral and subcutaneous adipose tissue volumes are cross-sectionally related to markers of inflammation and oxidative stress: the Framingham Heart Study. Circulation. 2007;116:1234–41. https://doi.org/10.1161/CIRCULATIONAHA.107.710509.
Chait A, den Hartigh LJ. Adipose tissue distribution, inflammation and its metabolic consequences, including diabetes and cardiovascular disease. Front Cardiovasc Med. 2020;7:22. https://doi.org/10.3389/fcvm.2020.00022.
Hetemaki N, Mikkola TS, Tikkanen MJ, Wang F, Hamalainen E, Turpeinen U, et al. Adipose tissue estrogen production and metabolism in premenopausal women. J Steroid Biochem Mol Biol. 2021;209:105849. https://doi.org/10.1016/j.jsbmb.2021.105849.
Vasan RS, Pan S, Xanthakis V, Beiser A, Larson MG, Seshadri S, et al. Arterial stiffness and long-term risk of health outcomes: the Framingham Heart Study. Hypertension. 2022;79:1045–56. https://doi.org/10.1161/HYPERTENSIONAHA.121.18776.
• Mitchell GF, Rong J, Larson MG, Cooper LL, Xanthakis V, Benjamin EJ, et al. Longitudinal hemodynamic correlates of and sex differences in the evolution of blood pressure across the adult lifespan: the Framingham Heart Study. J Am Heart Assoc. 2023;12:e027329. https://doi.org/10.1161/JAHA.122.027329. Provides evidence that proximal aortic stiffening predicts rises in pulse pressure more strongly in women.
• Haider N, Lebastchi J, Jayavelu AK, Batista TM, Pan H, Dreyfuss JM, et al. Signaling defects associated with insulin resistance in nondiabetic and diabetic individuals and modification by sex. J Clin Invest. 2021;131. https://doi.org/10.1172/JCI151818. Comprehensive phophorylation atlas in iPSC-derived myoblasts demonstrating marked differences in the phosphoproteome in men and women that related to cardiometabolic pathways including insulin resistance.
Tabit CE, Shenouda SM, Holbrook M, Fetterman JL, Kiani S, Frame AA, et al. Protein kinase C-beta contributes to impaired endothelial insulin signaling in humans with diabetes mellitus. Circulation. 2013;127:86–95. https://doi.org/10.1161/CIRCULATIONAHA.112.127514.
Cooper LL, Wang N, Beiser AS, Romero JR, Aparicio HJ, Lioutas VA, et al. Digital peripheral arterial tonometry and cardiovascular disease events: the Framingham Heart Study. Stroke. 2021;52:2866–73. https://doi.org/10.1161/STROKEAHA.120.031102.
Suboc TM, Dharmashankar K, Wang J, Ying R, Couillard A, Tanner MJ, et al. Moderate obesity and endothelial dysfunction in humans: influence of gender and systemic inflammation. Physiol Rep. 2013. https://doi.org/10.1002/phy2.58.
Moreau KL, Hildreth KL, Meditz AL, Deane KD, Kohrt WM. Endothelial function is impaired across the stages of the menopause transition in healthy women. J Clin Endocrinol Metab. 2012;97:4692–700. https://doi.org/10.1210/jc.2012-2244.
Minissian MB, Kilpatrick S, Eastwood JA, Robbins WA, Accortt EE, Wei J, et al. Association of spontaneous preterm delivery and future maternal cardiovascular disease. Circulation. 2018;137:865–71. https://doi.org/10.1161/CIRCULATIONAHA.117.031403.
Parikh NI, Gonzalez JM, Anderson CAM, Judd SE, Rexrode KM, Hlatky MA, et al. Adverse pregnancy outcomes and cardiovascular disease risk: unique opportunities for cardiovascular disease prevention in women: a scientific statement from the American Heart Association. Circulation. 2021;143:e902–16. https://doi.org/10.1161/CIR.0000000000000961.
Wilson PW, D’Agostino RB, Sullivan L, Parise H, Kannel WB. Overweight and obesity as determinants of cardiovascular risk: the Framingham experience. Arch Intern Med. 2002;162:1867–72. https://doi.org/10.1001/archinte.162.16.1867.
Ji H, Kim A, Ebinger JE, Niiranen TJ, Claggett BL, Bairey Merz CN, et al. Sex differences in blood pressure trajectories over the life course. JAMA Cardiol. 2020;5:19–26. https://doi.org/10.1001/jamacardio.2019.5306.
Vasan RS, Larson MG, Leip EP, Kannel WB, Levy D. Assessment of frequency of progression to hypertension in non-hypertensive participants in the Framingham Heart Study: a cohort study. Lancet. 2001;358:1682–6. https://doi.org/10.1016/S0140-6736(01)06710-1.
Liu J, Fox CS, Hickson DA, May WD, Hairston KG, Carr JJ, et al. Impact of abdominal visceral and subcutaneous adipose tissue on cardiometabolic risk factors: the Jackson Heart Study. J Clin Endocrinol Metab. 2010;95:5419–26. https://doi.org/10.1210/jc.2010-1378.
•• Samargandy S, Matthews KA, Brooks MM, Barinas-Mitchell E, Magnani JW, Thurston RC, et al. Trajectories of blood pressure in midlife women: does menopause matter? Circ Res. 2022;130:312–22. https://doi.org/10.1161/CIRCRESAHA.121.319424. Identification of distinct patterns of blood pressure elevation in women across the menopausal period with clear association of obesity with both higher premenopausal and higher rise at menopause.
Wolter NL, Jaffe IZ. Emerging vascular cell-specific roles for mineralocorticoid receptor: implications for understanding sex differences in cardiovascular disease. Am J Physiol Cell Physiol. 2023;324:C193–204. https://doi.org/10.1152/ajpcell.00372.2022.
Faulkner JL, Kennard S, Huby AC, Antonova G, Lu Q, Jaffe IZ, et al. Progesterone predisposes females to obesity-associated leptin-mediated endothelial dysfunction via upregulating endothelial MR (mineralocorticoid receptor) expression. Hypertension. 2019;74:678–86. https://doi.org/10.1161/HYPERTENSIONAHA.119.12802.
Wang L, Lin M, Yu J, Fan Z, Zhang S, Lin Y, et al. The impact of bariatric surgery versus non-surgical treatment on blood pressure: systematic review and meta-analysis. Obes Surg. 2021;31:4970–84. https://doi.org/10.1007/s11695-021-05671-9.
•• Kim MS, Kim WJ, Khera AV, Kim JY, Yon DK, Lee SW, et al. Association between adiposity and cardiovascular outcomes: an umbrella review and meta-analysis of observational and Mendelian randomization studies. Eur Heart J. 2021;42:3388–403. https://doi.org/10.1093/eurheartj/ehab454. Key comprehensive systematic review that provides sex-specific metrics associating measures of obesity with incidence cardiovacular diseases and delineates the role of genetics.
Bonaca MP, Hamburg NM, Creager MA. Contemporary medical management of peripheral artery disease. Circ Res. 2021;128:1868–84. https://doi.org/10.1161/CIRCRESAHA.121.318258.
Heffron SP, Dwivedi A, Rockman CB, Xia Y, Guo Y, Zhong J, et al. Body mass index and peripheral artery disease. Atherosclerosis. 2020;292:31–6. https://doi.org/10.1016/j.atherosclerosis.2019.10.017.
Horton WB, Barrett EJ. Microvascular dysfunction in diabetes mellitus and cardiometabolic disease. Endocr Rev. 2021;42:29–55. https://doi.org/10.1210/endrev/bnaa025.
Gill D, Zuber V, Dawson J, Pearson-Stuttard J, Carter AR, Sanderson E, et al. Risk factors mediating the effect of body mass index and waist-to-hip ratio on cardiovascular outcomes: Mendelian randomization analysis. Int J Obes (Lond). 2021;45:1428–38. https://doi.org/10.1038/s41366-021-00807-4.
• Lau ES, Paniagua SM, Guseh JS, Bhambhani V, Zanni MV, Courchesne P, et al. Sex differences in circulating biomarkers of cardiovascular disease. J Am Coll Cardiol. 2019;74:1543–53. https://doi.org/10.1016/j.jacc.2019.06.077. Longitudinal study demonstrating sex-based differences in the association of systemic biomarkers with incidence cardiovascular disease. Findings suggest that markers of inflammation and adipokines are more important in women.
Joseph JJ, Deedwania P, Acharya T, Aguilar D, Bhatt DL, Chyun DA, et al. Comprehensive management of cardiovascular risk factors for adults with type 2 diabetes: a scientific statement from the American Heart Association. Circulation. 2022;145:e722–59. https://doi.org/10.1161/CIR.0000000000001040.
Emerging Risk Factors Collaboration, Di Angelantonio E, Kaptoge S, Wormser D, Willeit P, Butterworth AS, et al. Association of cardiometabolic multimorbidity with mortality. JAMA. 2015;314:52–60. https://doi.org/10.1001/jama.2015.7008.
Wang L, Li X, Wang Z, Bancks MP, Carnethon MR, Greenland P, et al. Trends in prevalence of diabetes and control of risk factors in diabetes among US adults, 1999–2018. JAMA. 2021;326:1–13. https://doi.org/10.1001/jama.2021.9883.
Bangalore S, Fayyad R, Laskey R, DeMicco DA, Messerli FH, Waters DD. Body-weight fluctuations and outcomes in coronary disease. N Engl J Med. 2017;376:1332–40. https://doi.org/10.1056/NEJMoa1606148.
Ma C, Avenell A, Bolland M, Hudson J, Stewart F, Robertson C, et al. Effects of weight loss interventions for adults who are obese on mortality, cardiovascular disease, and cancer: systematic review and meta-analysis. BMJ. 2017;359:j4849. https://doi.org/10.1136/bmj.j4849.
Arterburn DE, Telem DA, Kushner RF, Courcoulas AP. Benefits and risks of bariatric surgery in adults: a review. JAMA. 2020;324:879–87. https://doi.org/10.1001/jama.2020.12567.
Sjostrom L, Peltonen M, Jacobson P, Sjostrom CD, Karason K, Wedel H, et al. Bariatric surgery and long-term cardiovascular events. JAMA. 2012;307:56–65. https://doi.org/10.1001/jama.2011.1914.
Sundstrom J, Bruze G, Ottosson J, Marcus C, Naslund I, Neovius M. Weight loss and heart failure: a nationwide study of gastric bypass surgery versus intensive lifestyle treatment. Circulation. 2017;135:1577–85. https://doi.org/10.1161/CIRCULATIONAHA.116.025629.
Kenchaiah S, Evans JC, Levy D, Wilson PW, Benjamin EJ, Larson MG, et al. Obesity and the risk of heart failure. N Engl J Med. 2002;347:305–13. https://doi.org/10.1056/NEJMoa020245.
Mahajan R, Stokes M, Elliott A, Munawar DA, Khokhar KB, Thiyagarajah A, et al. Complex interaction of obesity, intentional weight loss and heart failure: a systematic review and meta-analysis. Heart. 2020;106:58–68. https://doi.org/10.1136/heartjnl-2019-314770.
• Suthahar N, Lau ES, Blaha MJ, Paniagua SM, Larson MG, Psaty BM, et al. Sex-specific associations of cardiovascular risk factors and biomarkers with incident heart failure. J Am Coll Cardiol. 2020;76:1455–65. https://doi.org/10.1016/j.jacc.2020.07.044. Study demonstrating that comprehensive cardiovascular risk factor panels relate to heart failure risk in both men and women and suggesting selecting biomarkers have greater predictive value in women.
Savji N, Meijers WC, Bartz TM, Bhambhani V, Cushman M, Nayor M, et al. The association of obesity and cardiometabolic traits with incident HFpEF and HFrEF. JACC Heart Fail. 2018;6:701–9. https://doi.org/10.1016/j.jchf.2018.05.018.
Sotomi Y, Hikoso S, Nakatani D, Mizuno H, Okada K, Dohi T, et al. Sex differences in heart failure with preserved ejection fraction. J Am Heart Assoc. 2021;10:e018574. https://doi.org/10.1161/JAHA.120.018574.
DeFilippis EM, Beale A, Martyn T, Agarwal A, Elkayam U, Lam CSP, et al. Heart failure subtypes and cardiomyopathies in women. Circ Res. 2022;130:436–54. https://doi.org/10.1161/CIRCRESAHA.121.319900.
Eaton CB, Pettinger M, Rossouw J, Martin LW, Foraker R, Quddus A, et al. Risk factors for incident hospitalized heart failure with preserved versus reduced ejection fraction in a multiracial cohort of postmenopausal women. Circ Heart Fail. 2016. https://doi.org/10.1161/CIRCHEARTFAILURE.115.002883.
Paulus WJ, Tschope C. A novel paradigm for heart failure with preserved ejection fraction: comorbidities drive myocardial dysfunction and remodeling through coronary microvascular endothelial inflammation. J Am Coll Cardiol. 2013;62:263–71. https://doi.org/10.1016/j.jacc.2013.02.092.
Hamburg NM, Palmisano J, Larson MG, Sullivan LM, Lehman BT, Vasan RS, et al. Relation of brachial and digital measures of vascular function in the community: the Framingham heart study. Hypertension. 2011;57:390–6. https://doi.org/10.1161/HYPERTENSIONAHA.110.160812.
Crea F, Bairey Merz CN, Beltrame JF, Kaski JC, Ogawa H, Ong P, et al. The parallel tales of microvascular angina and heart failure with preserved ejection fraction: a paradigm shift. Eur Heart J. 2017;38:473–7. https://doi.org/10.1093/eurheartj/ehw461.
• Lau ES, Cunningham T, Hardin KM, Liu E, Malhotra R, Nayor M, et al. Sex differences in cardiometabolic traits and determinants of exercise capacity in heart failure with preserved ejection fraction. JAMA Cardiol. 2020;5:30–7. https://doi.org/10.1001/jamacardio.2019.4150. Comprehensive physiology assessment showing that even with lower burden of cardiometabolic traits, women have poorer cardiac and skeletal muscle responses to exercise.
Nguyen ITN, Brandt MM, van de Wouw J, van Drie RWA, Wesseling M, Cramer MJ, et al. Both male and female obese ZSF1 rats develop cardiac dysfunction in obesity-induced heart failure with preserved ejection fraction. PLoS ONE. 2020;15:e0232399. https://doi.org/10.1371/journal.pone.0232399.
InanlooRahatloo K, Liang G, Vo D, Ebert A, Nguyen I, Nguyen PK. Sex-based differences in myocardial gene expression in recently deceased organ donors with no prior cardiovascular disease. PLoS ONE. 2017;12:e0183874. https://doi.org/10.1371/journal.pone.0183874.
Peterson LR, Soto PF, Herrero P, Mohammed BS, Avidan MS, Schechtman KB, et al. Impact of gender on the myocardial metabolic response to obesity. JACC Cardiovasc Imaging. 2008;1:424–33. https://doi.org/10.1016/j.jcmg.2008.05.004.
• Sorimachi H, Obokata M, Takahashi N, Reddy YNV, Jain CC, Verbrugge FH, et al. Pathophysiologic importance of visceral adipose tissue in women with heart failure and preserved ejection fraction. Eur Heart J. 2021;42:1595–605. https://doi.org/10.1093/eurheartj/ehaa823. Study showing that greater degrees of abdominal adiposity on imaging relate to exercise induced elevations in left heart pressures in women.
Oguntade AS, Islam N, Malouf R, Taylor H, Jin D, Lewington S, et al. Body composition and risk of incident heart failure in 1 million adults: a systematic review and dose-response meta-analysis of prospective cohort studies. J Am Heart Assoc. 2023;12:e029062. https://doi.org/10.1161/JAHA.122.029062.
Cordola Hsu AR, Xie B, Peterson DV, LaMonte MJ, Garcia L, Eaton CB, et al. Metabolically healthy/unhealthy overweight/obesity associations with incident heart failure in postmenopausal women: the Women’s Health Initiative. Circ Heart Fail. 2021;14:e007297. https://doi.org/10.1161/CIRCHEARTFAILURE.120.007297.
Doumouras AG, Wong JA, Paterson JM, Lee Y, Sivapathasundaram B, Tarride JE, et al. Bariatric surgery and cardiovascular outcomes in patients with obesity and cardiovascular disease: a population-based retrospective cohort study. Circulation. 2021;143:1468–80. https://doi.org/10.1161/CIRCULATIONAHA.120.052386.
Chatterjee NA, Giulianini F, Geelhoed B, Lunetta KL, Misialek JR, Niemeijer MN, et al. Genetic obesity and the risk of atrial fibrillation: causal estimates from Mendelian randomization. Circulation. 2017;135:741–54. https://doi.org/10.1161/CIRCULATIONAHA.116.024921.
Schnabel RB, Yin X, Gona P, Larson MG, Beiser AS, McManus DD, et al. 50 year trends in atrial fibrillation prevalence, incidence, risk factors, and mortality in the Framingham Heart Study: a cohort study. Lancet. 2015;386:154–62. https://doi.org/10.1016/S0140-6736(14)61774-8.
Tedrow UB, Conen D, Ridker PM, Cook NR, Koplan BA, Manson JE, et al. The long- and short-term impact of elevated body mass index on the risk of new atrial fibrillation the WHS (women’s health study). J Am Coll Cardiol. 2010;55:2319–27. https://doi.org/10.1016/j.jacc.2010.02.029.
Mahajan R, Nelson A, Pathak RK, Middeldorp ME, Wong CX, Twomey DJ, et al. Electroanatomical remodeling of the atria in obesity: impact of adjacent epicardial fat. JACC Clin Electrophysiol. 2018;4:1529–40. https://doi.org/10.1016/j.jacep.2018.08.014.
• Ball J, Lochen ML, Wilsgaard T, Schirmer H, Hopstock LA, Morseth B, et al. Sex differences in the impact of body mass index on the risk of future atrial fibrillation: insights from the longitudinal population-based Tromso Study. J Am Heart Assoc. 2018;7. https://doi.org/10.1161/JAHA.117.008414. Longitudinal study demonstrating association of BMI with risk of atrial fibrillation in both men and women and a decrease in BMI over time reduced atrial fibrillation risk in women.
Abed HS, Wittert GA, Leong DP, Shirazi MG, Bahrami B, Middeldorp ME, et al. Effect of weight reduction and cardiometabolic risk factor management on symptom burden and severity in patients with atrial fibrillation: a randomized clinical trial. JAMA. 2013;310:2050–60. https://doi.org/10.1001/jama.2013.280521.
Chung MK, Eckhardt LL, Chen LY, Ahmed HM, Gopinathannair R, Joglar JA, et al. Lifestyle and risk factor modification for reduction of atrial fibrillation: a scientific statement from the American Heart Association. Circulation. 2020;141:e750–72. https://doi.org/10.1161/CIR.0000000000000748.
Pathak RK, Middeldorp ME, Meredith M, Mehta AB, Mahajan R, Wong CX, et al. Long-term effect of goal-directed weight management in an atrial fibrillation cohort: a long-term follow-up study (LEGACY). J Am Coll Cardiol. 2015;65:2159–69. https://doi.org/10.1016/j.jacc.2015.03.002.
Park DY, An S, Murthi M, Kattoor AJ, Kaur A, Ravi V, et al. Effect of weight loss on recurrence of atrial fibrillation after ablative therapy: a systematic review and meta-analysis. J Interv Card Electrophysiol. 2022;64:763–71. https://doi.org/10.1007/s10840-022-01168-2.
Rahmani J, Haghighian Roudsari A, Bawadi H, Thompson J, Khalooei Fard R, Clark C, et al. Relationship between body mass index, risk of venous thromboembolism and pulmonary embolism: a systematic review and dose-response meta-analysis of cohort studies among four million participants. Thromb Res. 2020;192:64–72. https://doi.org/10.1016/j.thromres.2020.05.014.
•• Brink A, Elf J, Svensson PJ, Engstrom G, Melander O, Zoller B. Sex-specific risk factors for deep venous thrombosis and pulmonary embolism in a population-based historical cohort study of middle-aged and older individuals. J Am Heart Assoc. 2023;12:e027502. https://doi.org/10.1161/JAHA.122.027502. Cohort study showing that adiposity metrics predict venous thrombosis risk in women but not in men.
Frischmuth T, Tondel BG, Braekkan SK, Hansen JB, Morelli VM. The risk of incident venous thromboembolism attributed to overweight and obesity: the Tromso Study. Thromb Haemost. 2023. https://doi.org/10.1055/s-0043-1772212.
Rodger MA, Scarvelis D, Kahn SR, Wells PS, Anderson DA, Chagnon I, et al. Long-term risk of venous thrombosis after stopping anticoagulants for a first unprovoked event: a multi-national cohort. Thromb Res. 2016;143:152–8. https://doi.org/10.1016/j.thromres.2016.03.028.
LaVasseur C, Neukam S, Kartika T, Samuelson Bannow B, Shatzel J, DeLoughery TG. Hormonal therapies and venous thrombosis: considerations for prevention and management. Res Pract Thromb Haemost. 2022;6:e12763. https://doi.org/10.1002/rth2.12763.
James AH, Jamison MG, Brancazio LR, Myers ER. Venous thromboembolism during pregnancy and the postpartum period: incidence, risk factors, and mortality. Am J Obstet Gynecol. 2006;194:1311–5. https://doi.org/10.1016/j.ajog.2005.11.008.
Creanga AA, Syverson C, Seed K, Callaghan WM. Pregnancy-related mortality in the United States, 2011–2013. Obstet Gynecol. 2017;130:366–73. https://doi.org/10.1097/AOG.0000000000002114.
Jacobsen AF, Skjeldestad FE, Sandset PM. Ante- and postnatal risk factors of venous thrombosis: a hospital-based case-control study. J Thromb Haemost. 2008;6:905–12. https://doi.org/10.1111/j.1538-7836.2008.02961.x.
Singh GK, DiBari JN. Marked Disparities in pre-pregnancy obesity and overweight prevalence among US women by race/ethnicity, nativity/immigrant status, and sociodemographic characteristics, 2012–2014. J Obes. 2019;2019:2419263. https://doi.org/10.1155/2019/2419263.
Villamor E, Cnattingius S. Interpregnancy weight change and risk of adverse pregnancy outcomes: a population-based study. Lancet. 2006;368:1164–70. https://doi.org/10.1016/S0140-6736(06)69473-7.
Santos S, Voerman E, Amiano P, Barros H, Beilin LJ, Bergstrom A, et al. Impact of maternal body mass index and gestational weight gain on pregnancy complications: an individual participant data meta-analysis of European, North American and Australian cohorts. BJOG. 2019;126:984–95. https://doi.org/10.1111/1471-0528.15661.
• Heslehurst N, Ngongalah L, Bigirumurame T, Nguyen G, Odeniyi A, Flynn A, et al. Association between maternal adiposity measures and adverse maternal outcomes of pregnancy: systematic review and meta-analysis. Obes Rev. 2022;23:e13449. https://doi.org/10.1111/obr.13449. A contemporary meta-analysis linking higher adiposity metrics to several adverse pregnancy outcomes including hypertensive disorders of pregnancy and gestational diabetes that are known to be associated with future cardiovascular risk.
Honigberg MC, Zekavat SM, Aragam K, Klarin D, Bhatt DL, Scott NS, et al. Long-term cardiovascular risk in women with hypertension during pregnancy. J Am Coll Cardiol. 2019;74:2743–54. https://doi.org/10.1016/j.jacc.2019.09.052.
Parikh NI, Jeppson RP, Berger JS, Eaton CB, Kroenke CH, LeBlanc ES, et al. Reproductive risk factors and coronary heart disease in the Women’s Health Initiative observational study. Circulation. 2016;133:2149–58. https://doi.org/10.1161/CIRCULATIONAHA.115.017854.
Ogunwole SM, Zera CA, Stanford FC. Obesity management in women of reproductive age. JAMA. 2021;325:433–4. https://doi.org/10.1001/jama.2020.21096.
Mehta LS, Warnes CA, Bradley E, Burton T, Economy K, Mehran R, et al. Cardiovascular considerations in caring for pregnant patients: a scientific statement from the American Heart Association. Circulation. 2020;141:e884–903. https://doi.org/10.1161/CIR.0000000000000772.
Powell-Wiley TM, Poirier P, Burke LE, Despres JP, Gordon-Larsen P, Lavie CJ, et al. Obesity and cardiovascular disease: a scientific statement from the American Heart Association. Circulation. 2021;143:e984–1010. https://doi.org/10.1161/CIR.0000000000000973.
Jensen MD, Ryan DH, Apovian CM, Ard JD, Comuzzie AG, Donato KA, et al. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society. Circulation. 2014;129:S102-138. https://doi.org/10.1161/01.cir.0000437739.71477.ee.
Look ARG, Gregg EW, Jakicic JM, Blackburn G, Bloomquist P, Bray GA, et al. Association of the magnitude of weight loss and changes in physical fitness with long-term cardiovascular disease outcomes in overweight or obese people with type 2 diabetes: a post-hoc analysis of the Look AHEAD randomised clinical trial. Lancet Diabetes Endocrinol. 2016;4:913–21. https://doi.org/10.1016/S2213-8587(16)30162-0.
Look ARG, Wing RR, Bolin P, Brancati FL, Bray GA, Clark JM, et al. Cardiovascular effects of intensive lifestyle intervention in type 2 diabetes. N Engl J Med. 2013;369:145–54. https://doi.org/10.1056/NEJMoa1212914.
Iwamoto SJ, Abushamat LA, Zaman A, Millard AJ, Cornier MA. Obesity management in cardiometabolic disease: state of the art. Curr Atheroscler Rep. 2021;23:59. https://doi.org/10.1007/s11883-021-00953-0.
Andrew CA, Saunders KH, Shukla AP, Aronne LJ. Treating obesity in patients with cardiovascular disease: the pharmacotherapeutic options. Expert Opin Pharmacother. 2019;20:585–93. https://doi.org/10.1080/14656566.2018.1561867.
Marx N, Husain M, Lehrke M, Verma S, Sattar N. GLP-1 Receptor agonists for the reduction of atherosclerotic cardiovascular risk in patients with type 2 diabetes. Circulation. 2022;146:1882–94. https://doi.org/10.1161/CIRCULATIONAHA.122.059595.
•• Wadden TA, Bailey TS, Billings LK, Davies M, Frias JP, Koroleva A, et al. Effect of subcutaneous semaglutide vs placebo as an adjunct to intensive behavioral therapy on body weight in adults with overweight or obesity: the STEP 3 randomized clinical trial. JAMA. 2021;325:1403–13. https://doi.org/10.1001/jama.2021.1831. Randomized clinical trial showing benefit of GLP-1 RA, semaglutide, combined with lifestyle intervention on weight loss in a study with a large percentage of women.
Lundgren JR, Janus C, Jensen SBK, Juhl CR, Olsen LM, Christensen RM, et al. Healthy weight loss maintenance with exercise, liraglutide, or both combined. N Engl J Med. 2021;384:1719–30. https://doi.org/10.1056/NEJMoa2028198.
•• Jastreboff AM, Aronne LJ, Ahmad NN, Wharton S, Connery L, Alves B, et al. Tirzepatide once weekly for the treatment of obesity. N Engl J Med. 2022;387:205–16. https://doi.org/10.1056/NEJMoa2206038. Randomized clinical trial showing that combined GLP-1 RA/GIP agent, tirzepatide, resulted in marked weight loss at highest dose.
Pfeffer MA, Claggett B, Diaz R, Dickstein K, Gerstein HC, Kober LV, et al. Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N Engl J Med. 2015;373:2247–57. https://doi.org/10.1056/NEJMoa1509225.
Marso SP, Daniels GH, Brown-Frandsen K, Kristensen P, Mann JF, Nauck MA, et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2016;375:311–22. https://doi.org/10.1056/NEJMoa1603827.
Marso SP, Bain SC, Consoli A, Eliaschewitz FG, Jodar E, Leiter LA, et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375:1834–44. https://doi.org/10.1056/NEJMoa1607141.
Hernandez AF, Green JB, Janmohamed S, D’Agostino RB Sr, Granger CB, Jones NP, et al. Albiglutide and cardiovascular outcomes in patients with type 2 diabetes and cardiovascular disease (harmony outcomes): a double-blind, randomised placebo-controlled trial. Lancet. 2018;392:1519–29. https://doi.org/10.1016/S0140-6736(18)32261-X.
Gerstein HC, Colhoun HM, Dagenais GR, Diaz R, Lakshmanan M, Pais P, et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. Lancet. 2019;394:121–30. https://doi.org/10.1016/S0140-6736(19)31149-3.
Husain M, Birkenfeld AL, Donsmark M, Dungan K, Eliaschewitz FG, Franco DR, et al. Oral semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2019;381:841–51. https://doi.org/10.1056/NEJMoa1901118.
Gerstein HC, Sattar N, Rosenstock J, Ramasundarahettige C, Pratley R, Lopes RD, et al. Cardiovascular and renal outcomes with efpeglenatide in type 2 diabetes. N Engl J Med. 2021;385:896–907. https://doi.org/10.1056/NEJMoa2108269.
Ryan DH, Lingvay I, Colhoun HM, Deanfield J, Emerson SS, Kahn SE, et al. Semaglutide effects on cardiovascular outcomes in people with overweight or obesity (SELECT) rationale and design. Am Heart J. 2020;229:61–9. https://doi.org/10.1016/j.ahj.2020.07.008.
Wilding JPH, Batterham RL, Calanna S, Davies M, Van Gaal LF, Lingvay I, et al. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384:989–1002. https://doi.org/10.1056/NEJMoa2032183.
Pi-Sunyer X, Astrup A, Fujioka K, Greenway F, Halpern A, Krempf M, et al. A randomized, controlled trial of 3.0 mg of liraglutide in weight management. N Engl J Med. 2015;373:11–22. https://doi.org/10.1056/NEJMoa1411892.
Zinman B, Wanner C, Lachin JM, Fitchett D, Bluhmki E, Hantel S, et al. Empagliflozin, cardiovascular outcomes, and mortality in type 2 diabetes. N Engl J Med. 2015;373:2117–28. https://doi.org/10.1056/NEJMoa1504720.
Neal B, Perkovic V, Mahaffey KW, de Zeeuw D, Fulcher G, Erondu N, et al. Canagliflozin and cardiovascular and renal events in type 2 diabetes. N Engl J Med. 2017;377:644–57. https://doi.org/10.1056/NEJMoa1611925.
Wiviott SD, Raz I, Bonaca MP, Mosenzon O, Kato ET, Cahn A, et al. Dapagliflozin and cardiovascular outcomes in type 2 diabetes. N Engl J Med. 2019;380:347–57. https://doi.org/10.1056/NEJMoa1812389.
Cannon CP, Pratley R, Dagogo-Jack S, Mancuso J, Huyck S, Masiukiewicz U, et al. Cardiovascular outcomes with ertugliflozin in type 2 diabetes. N Engl J Med. 2020;383:1425–35. https://doi.org/10.1056/NEJMoa2004967.
Bhatt DL, Szarek M, Pitt B, Cannon CP, Leiter LA, McGuire DK, et al. Sotagliflozin in patients with diabetes and chronic kidney disease. N Engl J Med. 2021;384:129–39. https://doi.org/10.1056/NEJMoa2030186.
McMurray JJV, Solomon SD, Inzucchi SE, Kober L, Kosiborod MN, Martinez FA, et al. Dapagliflozin in patients with heart failure and reduced ejection fraction. N Engl J Med. 2019;381:1995–2008. https://doi.org/10.1056/NEJMoa1911303.
Packer M, Anker SD, Butler J, Filippatos G, Pocock SJ, Carson P, et al. Cardiovascular and renal outcomes with empagliflozin in heart failure. N Engl J Med. 2020;383:1413–24. https://doi.org/10.1056/NEJMoa2022190.
Anker SD, Butler J, Filippatos G, Ferreira JP, Bocchi E, Bohm M, et al. Empagliflozin in heart failure with a preserved ejection fraction. N Engl J Med. 2021;385:1451–61. https://doi.org/10.1056/NEJMoa2107038.
Bhatt DL, Szarek M, Steg PG, Cannon CP, Leiter LA, McGuire DK, et al. Sotagliflozin in patients with diabetes and recent worsening heart failure. N Engl J Med. 2021;384:117–28. https://doi.org/10.1056/NEJMoa2030183.
Solomon SD, McMurray JJV, Claggett B, de Boer RA, DeMets D, Hernandez AF, et al. Dapagliflozin in heart failure with mildly reduced or preserved ejection fraction. N Engl J Med. 2022;387:1089–98. https://doi.org/10.1056/NEJMoa2206286.
Ferro EG, Michos ED, Bhatt DL, Lincoff AM, Elshazly MB. New decade, new FDA guidance for diabetes drug development: lessons learned and future directions. J Am Coll Cardiol. 2020;76:2522–6. https://doi.org/10.1016/j.jacc.2020.09.590.
Sattar N, Lee MMY, Kristensen SL, Branch KRH, Del Prato S, Khurmi NS, et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of randomised trials. Lancet Diabetes Endocrinol. 2021;9:653–62. https://doi.org/10.1016/S2213-8587(21)00203-5.
Kristensen SL, Rorth R, Jhund PS, Docherty KF, Sattar N, Preiss D, et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet Diabetes Endocrinol. 2019;7:776–85. https://doi.org/10.1016/S2213-8587(19)30249-9.
Gerstein HC, Li Z, Ramasundarahettige C, Baek S, Branch KRH, Del Prato S, et al. Exploring the relationship between efpeglenatide dose and cardiovascular outcomes in type 2 diabetes: insights from the AMPLITUDE-O trial. Circulation. 2023;147:1004–13. https://doi.org/10.1161/CIRCULATIONAHA.122.063716.
Breton-Romero R, Weisbrod RM, Feng B, Holbrook M, Ko D, Stathos MM, et al. Liraglutide treatment reduces endothelial endoplasmic reticulum stress and insulin resistance in patients with diabetes mellitus. J Am Heart Assoc. 2018;7:e009379. https://doi.org/10.1161/JAHA.118.009379.
Rakipovski G, Rolin B, Nohr J, Klewe I, Frederiksen KS, Augustin R, et al. The GLP-1 analogs liraglutide and semaglutide reduce atherosclerosis in ApoE(-/-) and LDLr(-/-) mice by a mechanism that includes inflammatory pathways. JACC Basic Transl Sci. 2018;3:844–57. https://doi.org/10.1016/j.jacbts.2018.09.004.
Sharma A, Verma S. Mechanisms by which glucagon-like-peptide-1 receptor agonists and sodium-glucose cotransporter-2 inhibitors reduce cardiovascular risk in adults with type 2 diabetes mellitus. Can J Diabetes. 2020;44:93–102. https://doi.org/10.1016/j.jcjd.2019.09.003.
•• Singh AK, Singh R. Gender difference in cardiovascular outcomes with SGLT-2 inhibitors and GLP-1 receptor agonist in type 2 diabetes: a systematic review and meta-analysis of cardio-vascular outcome trials. Diabetes Metab Syndr. 2020;14:181–7. https://doi.org/10.1016/j.dsx.2020.02.012. A meta-analysis consistent with benefit of SGLT2 inhibitors and GLP-1 RA on cardiovascular outcomes in both women and men with T2DM.
Raparelli V, Elharram M, Moura CS, Abrahamowicz M, Bernatsky S, Behlouli H, et al. Sex differences in cardiovascular effectiveness of newer glucose-lowering drugs added to metformin in type 2 diabetes mellitus. J Am Heart Assoc. 2020;9:e012940. https://doi.org/10.1161/JAHA.119.012940.
Brown E, Heerspink HJL, Cuthbertson DJ, Wilding JPH. SGLT2 inhibitors and GLP-1 receptor agonists: established and emerging indications. Lancet. 2021;398:262–76. https://doi.org/10.1016/S0140-6736(21)00536-5.
Pereira MJ, Eriksson JW. Emerging role of SGLT-2 inhibitors for the treatment of obesity. Drugs. 2019;79:219–30. https://doi.org/10.1007/s40265-019-1057-0.
Cai X, Yang W, Gao X, Chen Y, Zhou L, Zhang S, et al. The association Between the dosage of SGLT2 inhibitor and weight reduction in type 2 diabetes patients: a meta-analysis. Obesity (Silver Spring). 2018;26:70–80. https://doi.org/10.1002/oby.22066.
• Cheong AJY, Teo YN, Teo YH, Syn NL, Ong HT, Ting AZH, et al. SGLT inhibitors on weight and body mass: a meta-analysis of 116 randomized-controlled trials. Obesity (Silver Spring). 2022;30:117–28. https://doi.org/10.1002/oby.23331. Quantification of the degree of weight loss across multiple trials of SGLT2 inhibitors.
Teo YH, Teo YN, Syn NL, Kow CS, Yoong CSY, Tan BYQ, et al. Effects of sodium/glucose cotransporter 2 (SGLT2) inhibitors on cardiovascular and metabolic outcomes in patients without diabetes mellitus: a systematic review and meta-analysis of randomized-controlled trials. J Am Heart Assoc. 2021;10:e019463. https://doi.org/10.1161/JAHA.120.019463.
Pereira MJ, Lundkvist P, Kamble PG, Lau J, Martins JG, Sjostrom CD, et al. A randomized controlled trial of dapagliflozin plus once-weekly exenatide versus placebo in individuals with obesity and without diabetes: metabolic effects and markers associated with bodyweight loss. Diabetes Ther. 2018;9:1511–32. https://doi.org/10.1007/s13300-018-0449-6.
Deol H, Lekkakou L, Viswanath AK, Pappachan JM. Combination therapy with GLP-1 analogues and SGLT-2 inhibitors in the management of diabesity: the real world experience. Endocrine. 2017;55:173–8. https://doi.org/10.1007/s12020-016-1125-0.
• Zelniker TA, Wiviott SD, Raz I, Im K, Goodrich EL, Bonaca MP, et al. SGLT2 inhibitors for primary and secondary prevention of cardiovascular and renal outcomes in type 2 diabetes: a systematic review and meta-analysis of cardiovascular outcome trials. Lancet. 2019;393:31–9. https://doi.org/10.1016/S0140-6736(18)32590-X. A patient-level meta-analysis showing consistent cardiovascular benefit of SGLT2 inhibitors in women with T2DM.
Rahman H, Khan SU, Lone AN, Ghosh P, Kunduru M, Sharma S, et al. Sodium-glucose cotransporter-2 inhibitors and primary prevention of atherosclerotic cardiovascular disease: a meta-analysis of randomized trials and systematic review. J Am Heart Assoc. 2023;12:e030578. https://doi.org/10.1161/JAHA.123.030578.
Nassif ME, Windsor SL, Borlaug BA, Kitzman DW, Shah SJ, Tang F, et al. The SGLT2 inhibitor dapagliflozin in heart failure with preserved ejection fraction: a multicenter randomized trial. Nat Med. 2021;27:1954–60. https://doi.org/10.1038/s41591-021-01536-x.
Vaduganathan M, Docherty KF, Claggett BL, Jhund PS, de Boer RA, Hernandez AF, et al. SGLT-2 inhibitors in patients with heart failure: a comprehensive meta-analysis of five randomised controlled trials. Lancet. 2022;400:757–67. https://doi.org/10.1016/S0140-6736(22)01429-5.
Heidenreich PA, Bozkurt B, Aguilar D, Allen LA, Byun JJ, Colvin MM, et al. 2022 AHA/ACC/HFSA guideline for the management of heart failure: a report of the American College of Cardiology/American Heart Association Joint Committee on Clinical Practice Guidelines. Circulation. 2022;145:e895–1032. https://doi.org/10.1161/CIR.0000000000001063.
Paul SK, Bhatt DL, Montvida O. The association of amputations and peripheral artery disease in patients with type 2 diabetes mellitus receiving sodium-glucose cotransporter type-2 inhibitors: real-world study. Eur Heart J. 2021;42:1728–38. https://doi.org/10.1093/eurheartj/ehaa956.
Sharma A, Aziz H, Verma S, Abramson BL, Choi R, Chua GL, et al. Permission to prescribe: do cardiologists need permission to prescribe diabetes medications that afford cardiovascular benefit? Curr Opin Cardiol. 2021;36:672–81. https://doi.org/10.1097/HCO.0000000000000892.
• Gunawan F, Nassif ME, Partridge C, Ahmad T, Kosiborod M, Inzucchi SE. Relative frequency of cardiology vs. endocrinology visits by type 2 diabetes patients with cardiovascular disease in the USA: implications for implementing evidence-based use of glucose-lowering medications. Cardiovasc Endocrinol Metab. 2020;9:56–9. https://doi.org/10.1097/XCE.0000000000000195. Study showing higher rate of visits with cardiologists in patients with T2DM emphasizing multidisclinary care for implementation of new diabetes-treatment guidelines.
American Diabetes Association Professional Practice Committee. 10 Cardiovascular disease and risk management: standards of medical care in diabetes-2022. Diabetes Care. 2022;45:S144–74. https://doi.org/10.2337/dc22-S010.
Virani SS, Newby LK, Arnold SV, Bittner V, Brewer LC, Demeter SH, et al. 2023 AHA/ACC/ACCP/ASPC/NLA/PCNA guideline for the management of patients with chronic coronary disease: a report of the American Heart Association/American College of Cardiology Joint Committee on Clinical Practice Guidelines. Circulation. 2023. https://doi.org/10.1161/CIR.0000000000001168.
Dave CV, Schneeweiss S, Wexler DJ, Brill G, Patorno E. Trends in clinical characteristics and prescribing preferences for SGLT2 inhibitors and GLP-1 receptor agonists, 2013–2018. Diabetes Care. 2020;43:921–4. https://doi.org/10.2337/dc19-1943.
Lingvay I, Catarig AM, Frias JP, Kumar H, Lausvig NL, le Roux CW, et al. Efficacy and safety of once-weekly semaglutide versus daily canagliflozin as add-on to metformin in patients with type 2 diabetes (SUSTAIN 8): a double-blind, phase 3b, randomised controlled trial. Lancet Diabetes Endocrinol. 2019;7:834–44. https://doi.org/10.1016/S2213-8587(19)30311-0.
Zelniker TA, Wiviott SD, Raz I, Im K, Goodrich EL, Furtado RHM, et al. Comparison of the effects of glucagon-like peptide receptor agonists and sodium-glucose cotransporter 2 inhibitors for prevention of major adverse cardiovascular and renal outcomes in type 2 diabetes mellitus. Circulation. 2019;139:2022–31. https://doi.org/10.1161/CIRCULATIONAHA.118.038868.
Ludvik B, Frias JP, Tinahones FJ, Wainstein J, Jiang H, Robertson KE, et al. Dulaglutide as add-on therapy to SGLT2 inhibitors in patients with inadequately controlled type 2 diabetes (AWARD-10): a 24-week, randomised, double-blind, placebo-controlled trial. Lancet Diabetes Endocrinol. 2018;6:370–81. https://doi.org/10.1016/S2213-8587(18)30023-8.
Zinman B, Bhosekar V, Busch R, Holst I, Ludvik B, Thielke D, et al. Semaglutide once weekly as add-on to SGLT-2 inhibitor therapy in type 2 diabetes (SUSTAIN 9): a randomised, placebo-controlled trial. Lancet Diabetes Endocrinol. 2019;7:356–67. https://doi.org/10.1016/S2213-8587(19)30066-X.
Adhikari R, Jha K, Dardari Z, Heyward J, Blumenthal RS, Eckel RH, et al. National trends in use of sodium-glucose cotransporter-2 inhibitors and glucagon-like peptide-1 receptor agonists by cardiologists and other specialties, 2015 to 2020. J Am Heart Assoc. 2022;11:e023811. https://doi.org/10.1161/JAHA.121.023811.
Essien UR, Singh B, Swabe G, Johnson AE, Eberly LA, Wadhera RK, et al. Association of prescription co-payment with adherence to glucagon-like peptide-1 receptor agonist and sodium-glucose cotransporter-2 inhibitor therapies in patients with heart failure and diabetes. JAMA Netw Open. 2023;6:e2316290. https://doi.org/10.1001/jamanetworkopen.2023.16290.
Funding
Dr. Hamburg is supported by R01HL134893, R01HL160003, R01HL168889, and AHASFRN35120118.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of Interest
Dr. Hamburg has done consulting for Novo Nordisk. The other authors have nothing to disclose.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Behrooz, L., Lenneman, C.G. & Hamburg, N.M. Emerging Medical Therapies for the Treatment of Obesity in Women with Cardiovascular Diseases. Curr Cardiol Rep 25, 1475–1488 (2023). https://doi.org/10.1007/s11886-023-01961-z
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11886-023-01961-z