Abstract
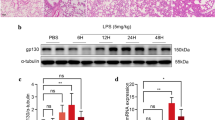
This study aimed to observe the expression of angiopoietin-2 (Ang-2) in the lung tissue of juvenile SD rats with lipopolysaccharide (LPS)-induced acute lung injury (ALI) and to clarify the role of ulinastatin (UTI). Ninety 18–21-day-old juvenile SD male rats were randomly divided into five groups (n = 18). ALI rat model was established by intraperitoneal injection of LPS (LPS 10 mg/kg), while the control group was given the same dose of normal saline. The UTI intervention group was given the injection of UTI (5000 U/mL) immediately after the injection of LPS, which was divided into UTI low-dose group (LPS + 5 ml/kg UTI), UTI medium-dose group (LPS + 10 ml/kg UTI), and UTI high-dose group (LPS + 20 ml/kg UTI).The respiratory status of each group of rats was observed, and six rats were randomly selected to be killed in each group at 6, 12, and 24 h, and the lung tissues were dissected and retained. The pathological changes of the lung tissues were observed by hematoxylin–eosin (HE) staining, the expression levels and locations of Ang-2 and vascular endothelial growth factor (VEGF) in lung tissue were observed by immunohistochemical staining, and the expressions of genes and proteins of Ang-2 and VEGF were detected by quantitative reverse transcription polymerase chain reaction (RT-PCR) and Western blot analysis. Three hours after intraperitoneal injection, rats in the model group developed shortness of breath and the developed respiratory distress progressed over time. The lung pathological changes in the model group were obvious compared with those in the control group, and gradually worsened with time, and the pathological changes of lung in the rats in the UTI intervention group were reduced compared with those in the model group. At different time points, the expressions of Ang-2 and VEGF in the lung tissue of rats in the model group were higher than those in the control group, and were lower in the UTI intervention group than those in the model group. The expressions of Ang-2 and VEGF protein were lower in the low-dose group of UTI group than those in the high-dose group of UTI group at different time points (P < 0.05), and the expressions of Ang-2 and VEGF protein in the low-dose group of UTI were significantly lower than those in the medium-dose group at 12 h and 24 h (P < 0.05). The expression of Ang-2 was increased in the lung tissue of juvenile SD rats with LPS-induced ALI, and was associated with the degree of lung injury. UTI might attenuate LPS-induced ALI by inhibiting the expression of Ang-2 in lung tissue, and the low dose was more obvious than the medium and high dose.








Similar content being viewed by others
Data availability
The data are free access to available upon request.
References
Ao L, Gao H, Jia L et al (2022) Matrine inhibits synovial angiogenesis in collagen-induced arthritis rats by regulating HIF-VEGF-Ang and inhibiting the PI3K/Akt signaling pathway. Mol Immunol 141:13–20. https://doi.org/10.1016/j.molimm.2021.11.002
Biel NM, Siemann DW (2016) Targeting the angiopoietin-2/Tie-2 axis in conjunction with VEGF signal interference. Cancer Lett 380:525–533. https://doi.org/10.1016/j.canlet.2014.09.035
Blázquez-Prieto J, Huidobro C, López-Alonso I et al (2021) Activation of p21 limits acute lung injury and induces early senescence after acid aspiration and mechanical ventilation. Transl Res 233:104–116. https://doi.org/10.1016/j.trsl.2021.01.008
Cao J, Kuang D, Luo M et al (2022) Targeting circNCLN/miR-291a-3p/TSLP signaling axis alleviates lipopolysaccharide-induced acute lung injury. Biochem Biophys Res Commun 617:60–67. https://doi.org/10.1016/j.bbrc.2022.05.095
Chen L, Li W, Qi D et al (2018) Honokiol protects pulmonary microvascular endothelial barrier against lipopolysaccharide-induced ARDS partially via the Sirt3/AMPK signaling axis. Life Sci 210:86–95. https://doi.org/10.1016/j.lfs.2018.08.064
Cui N, Liang Y, Wang J et al (2021) Minocycline attenuates oxidative and inflammatory injury in a intestinal perforation induced septic lung injury model via down-regulating lncRNA MALAT1 expression. Int Immunopharmacol 100:108–115. https://doi.org/10.1016/j.intimp.2021.108115
Deng J, Wang D, Tang J et al (2020) An increase in alveolar fluid clearance induced by hyperinsulinemia in obese rats with LPS-induced acute lung injury. Respir Physiol Neurobiol 279:103470. https://doi.org/10.1016/j.resp.2020.103470
Gottlieb DJ, Lederer DJ, Kim JS et al (2022) Effect of positive airway pressure therapy of obstructive sleep apnea on circulating angiopoietin-2. Sleep Med 96:119–121. https://doi.org/10.1016/j.sleep.2022.05.007
Guo Q, Jin J, Yuan JXJ et al (2011) VEGF, Bcl-2 and Bad regulated by angiopoietin-1 in oleic acid induced acute lung injury. Biochem Biophys Res Commun 413:630–636. https://doi.org/10.1016/j.bbrc.2011.09.015
Hsieh P, Wu Y, Yang M et al (2022) Deciphering the role of damage-associated molecular patterns and inflammatory responses in acute lung injury. Life Sci 2022:120782. https://doi.org/10.1016/j.lfs.2022.120782
Jin Z, Li M, Tang L et al (2022) Protective effect of Ulinastatin on acute lung injury in diabetic sepsis rats. Int Immunopharmacol 108:108908. https://doi.org/10.1016/j.intimp.2022.108908
Kong M, Zhu D, Dong J et al (2023) Iso-seco-tanapartholide from Artemisia argyi inhibits the PFKFB3-mediated glycolytic pathway to attenuate airway inflammation in lipopolysaccharide-induced acute lung injury mice. J Ethnopharmacol 301:115781. https://doi.org/10.1016/j.jep.2022.115781
Lange M, Hamahata A, Traber DL et al (2012) Pulmonary microvascular hyperpermeability and expression of vascular endothelial growth factor in smoke inhalation-and pneumonia-induced acute lung injury. Burns 38:1072–1078. https://doi.org/10.1016/j.burns.2012.02.019
Li S, Yang W, Zhou L et al (2019) Vascular permeability and hemodynamic effects of ulinastatin on organs affected by shock during early burn injury. Am J Emerg Med 37:249–253. https://doi.org/10.1016/j.ajem.2018.05.038
Li X, Wei Y, Li S et al (2022) Zanubrutinib ameliorates lipopolysaccharide-induced acute lung injury via regulating macrophage polarization. Int Immunopharmacol 111:109138. https://doi.org/10.1016/j.intimp.2022.109138
Liu W, Li Y, Bo L et al (2021) Positive regulation of TFEB and mitophagy by PGC-1α to alleviate LPS-induced acute lung injury in rats. Biochem Biophys Res Commun 577:1–5. https://doi.org/10.1016/j.bbrc.2021.08.064
Luo L, Huang F, Zhong S et al (2022) Astaxanthin attenuates ferroptosis via Keap1-Nrf2/HO-1 signaling pathways in LPS-induced acute lung injury. Life Sci 311:121091. https://doi.org/10.1016/j.lfs.2022.121091
Lymperopoulou K, Velissaris D, Kotsaki A et al (2015) Angiopoietin-2 associations with the underlying infection and sepsis severity. Cytokine 73:163–168. https://doi.org/10.1016/j.cyto.2015.01.022
Niemiec SM, Hilton SA, Wallbank A et al (2022) Lung function improves after delayed treatment with CNP-miR146a following acute lung injury. Nanomed Nanotechnol, Biol Med 40:102498. https://doi.org/10.1016/j.nano.2021.102498
Qiao J, Chen L, Huang X et al (2019) Effects of nebulized N-acetylcystein on the expression of HMGB1 and RAGE in rats with hyperoxia-nduced lung injury. J Cell Physiol 234:10547–10553. https://doi.org/10.1002/jcp.27724
Qiu Y, Chen C, Zhang J et al (2019) VEGF attenuates lung injury by inducing homing of CD133+ progenitors via VEGFR1. Biochem Biophys Res Commun 511:650–657. https://doi.org/10.1016/j.bbrc.2019.02.071
Song Y, Xu C, Wu J et al (2022) Palmatine alleviates LPS-induced acute lung injury via interfering the interaction of TAK1 and TAB1. Biochem Pharmacol 202:115120. https://doi.org/10.1016/j.bcp.2022.115120
Sun S, Yao Y, Huang C et al (2022) CD36 regulates LPS-induced acute lung injury by promoting macrophages M1 polarization. Cell Immunol 372:104475. https://doi.org/10.1016/j.cellimm.2021.104475
Tang B, Ma L, Yao X et al (2017) Hydrogen sulfide ameliorates acute lung injury induced by infrarenal aortic cross-clamping by inhibiting inflammation and angiopoietin 2 release. J Vasc Surg 65:501–508. https://doi.org/10.1016/j.jvs.2015.10.010
Wei F, Liu SY, Luo L et al (2017) Anti-inflammatory mechanism of ulinastatin: Inhibiting the hyperpermeability of vascular endothelial cells induced by TNF-α via the RhoA/ROCK signal pathway. Int Immunopharmacol 46:220–227. https://doi.org/10.1016/j.intimp.2017.03.007
Wu J, Yan X, Jin G (2019) Ulinastatin protects rats from sepsis-induced acute lung injury by suppressing the JAK-STAT3 pathway. J Cell Biochem 120:2554–2559. https://doi.org/10.1002/jcb.27550
Yang X, Song J, Hou S et al (2020) Ulinastatin ameliorates acute kidney injury induced by crush syndrome inflammation by modulating Th17/Treg cells. Int Immunopharmacol 81:106265. https://doi.org/10.1016/j.intimp.2020.106265
Zhang H, Sha J, Feng X et al (2019) Dexmedetomidine ameliorates LPS induced acute lung injury via GSK-3β/STAT3-NF-κB signaling pathway in rats. Int Immunopharmacol 74:105717. https://doi.org/10.1016/j.intimp.2019.105717
Zhong J, Lu W, Zhang J et al (2020) Notoginsenoside R1 activates the Ang2/Tie2 pathway to promote angiogenesis. Phytomedicine 78:153302. https://doi.org/10.1016/j.phymed.2020.153302
Funding
This work was supported by the Henan Province Science and Technology Research Project (222102310191).
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
About this article
Cite this article
Qiao, J., Guo, S., Huang, X. et al. Expression of Angiopoietin-2 in Lung Tissue of Juvenile SD Rats with Lipopolysaccharide-Induced Acute Lung Injury and the Role of Ulinastatin. Arch. Immunol. Ther. Exp. 71, 23 (2023). https://doi.org/10.1007/s00005-023-00688-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s00005-023-00688-7