Abstract
Hypertension (HTN), a complex cardiovascular disease (CVD), significantly impacts global health, prompting a growing interest in complementary and alternative therapeutic approaches. This review article seeks to provide an up-to-date and thorough summary of modern therapeutic techniques for treating HTN, with an emphasis on the molecular mechanisms of action found in substances found in plants, herbs, and seafood. Bioactive molecules have been a significant source of novel therapeutics and are crucial in developing and testing new HTN remedies. Recent advances in science have made it possible to understand the complex molecular mechanisms underlying blood pressure (BP)-regulating effects of these natural substances better. Polyphenols, flavonoids, alkaloids, and peptides are examples of bioactive compounds that have demonstrated promise in influencing several pathways involved in regulating vascular tone, reducing oxidative stress (OS), reducing inflammation, and improving endothelial function. The article explains the vasodilatory, diuretic, and renin–angiotensin–aldosterone system (RAAS) modifying properties of vital plants such as garlic and olive leaf. Phytochemicals from plants are the primary in traditional drug development as models for novel antihypertensive drugs, providing diverse strategies to combat HTN due to their biological actions. The review also discusses the functions of calcium channel blockers originating from natural sources, angiotensin-converting enzyme (ACE) inhibitors, and nitric oxide (NO) donors. Including seafood components in this study demonstrates the increased interest in using bioactive chemicals originating from marine sources to treat HTN. Omega-3 fatty acids, peptides, and minerals obtained from seafood sources have anti-inflammatory, vasodilatory, and antioxidant properties that improve vascular health and control BP. Overall, we discussed the multiple functions of bioactive molecules and seafood components in the treatment of HTN.
Similar content being viewed by others
1 Introduction
Hypertension (HTN), a prevalent chronic disease associated with age, frequently leads to quite significant renal and cardiac issues. Along with blood pressure (BP), a variety of other cardiovascular risk factors are regularly reported [1]. It causes abnormally high BP in the arteries. There are two options: major [2] and secondary (less important) and optional. 90% to 95% of cases of high BP are classified as primary HTN, which means they have no known medical reason [3]. Secondary HTN is brought on by a variety of conditions that affect the arteries, kidneys, endocrine system, or heart in the remaining 4 to 9% of cases [4]. HTN is a major contributor to chronic kidney disease and a risk factor for aneurysms, heart attacks, strokes, and heart failure [5]. A small rise in arterial BP shortens life expectancy. BP can be controlled and the risk of health issues is reduced with the use of medications, dietary changes, and lifestyle adjustments [6]. One of the leading factors in death globally is HTN. Even though a variety of current therapeutics are used to treat clinical HTN, they all have negative consequences. BP is the force that the blood applies to the artery walls. SBP (systolic blood pressure under 120 mmHg) and DBP (diastolic blood pressure under 80 mmHg) are the two classifications. Patients with HTN experience an increase in both SBP and DBP that is greater than 90 mmHg. HTN affects 25.4 percent of the world's population now, with a 60 percent increase anticipated by 2025. HTN is divided into two types (Fig. 1) [7]. Many of today's medicines are obtained directly or indirectly from natural compounds, which have proven to be a great source of innovative medications. This is especially true in cancer and infectious disease research [8, 9]. Meanwhile, natural products have an impact in several circumstances [10]. Around the world, HTN has become one of the top preventable causes of disease and mortality. It is expected to kill 7.5 million people per year, or 12.8 percent of all deaths [11, 12]. The majority of currently used HTN medications cannot be administered as a single-pill therapy due to their low efficacy and side effects. Hence, new medicine development with a wide range of therapeutic effects is highly desirable. Recently advances in the investigation of natural lead compounds with antihypertensive activity, with an emphasis on the mechanisms underlying their antihypertensive effect [13]. Based on molecular mechanisms of action, we reviewed the multiple functions that plants, and seafood components may have in treating HTN. After that, we focused on the phytochemicals and seafood components that are most frequently used to treat and manage HTN. The use of bioactive molecules components in the treatment of HTN has potential. A comprehensive approach is provided by their various molecular modes of action, which include vasodilation, diuretic effects, and the decrease of oxidative stress (OS). For better cardiovascular health, this synergy may result in the successful and balanced control of HTN. Future developments appear promising as we explore deeper into the molecular interactions of bioactive molecules in the treatment of HTN. Individualized treatments focusing on particular processes might become available, improving efficacy and reducing negative consequences. Innovative and comprehensive approaches to managing HTN could be made potential through combined efforts between traditional and modern medications.
2 Methodology
A strategy was designed to identify literature published in prestigious journals. All the search results were from PubMed, Scopus, and WOS. The keywords hypertension, bioactive molecules, blood pressure, phytochemicals, and seafood compounds were used. We chose and analyzed original studies, review articles, and research reports published until 2023.
3 Causes of HTN
3.1 Essential HTN
"The treatment of the HTN itself is a difficult and nearly desperate task in the current state of our knowledge, and in fact, because knowing the HTN may be a significant compensatory mechanism that should not be tampered with even if it were certain that we could control it." This prevalent misunderstanding concerning the clinical meaning of essential HTN was first articulated by Paul Dudley White in 1931 [14]. The most common type of HTN is essential HTN, which affects 90 to 95 percent of hypertensive persons [3]. Sedentary lifestyles, potassium deficiency (hypokalemia), stress, [15], obesity [16], (those with a body mass index (BMI) higher than 25 account for more than 85 percent of instances) [17], alcohol intake [18], salt (sodium) sensitivity [19], and vitamin D deficiency have all been linked to an increased risk of developing HTN [20]. HTN risk is increased by aging [21], some inherited genetic alterations [22], and having a family history of HTN [23]. An increase in renin, a kidney-secreted enzyme, as well as sympathetic nervous system (SNS) over activity [24], are also risk factors [25]. HTN is hypothesized to be influenced by insulin resistance, which is a part of syndrome X, sometimes known as metabolic syndrome [26]. The rate of preterm birth is lower among children with essential HTN, who also present at an older age, have a greater family history of the condition, and present with essential HTN [27]. The complex character of primary HTN necessitates a comprehensive strategy. Dietary and exercise changes, for example, are essential. Early detection and continuous management are essential due to the interaction between genetic predisposition and environmental factors.
3.2 Secondary HTN
Secondary HTN is high BP that has an underlying, recognizable, and frequently reversible cause. Only 4 to 9 percent of instances of HTN are believed to be the consequence of secondary factors [28]. By addressing the major cause of high BP, this type of HTN differs from essential HTN [29]. HTN can also be influenced by other diseases that cause hormonal imbalances, such as excessive thyroid function, and a lack of thyroid and adrenal gland cancer. Secondary HTN can also be brought on by kidney disease, pre-eclampsia during pregnancy, and coarctation of the aorta [6]. A tiny but considerable portion of the hypertensive population suffers from secondary HTN, which, in contrast to initial HTN, may be treatable [30]. Secondary HTN is caused by recognized underlying conditions such as hormone imbalances [30]. To effectively treat the condition and reduce any potential health risks, it is essential to manage the underlying causes.
4 Pathophysiology of HTN
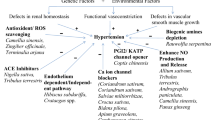
The etiology of HTN is still unknown . As the underlying cause of their high BP, between 2 and 5% of persons have kidney or adrenal insufficiency [4]. The majority of the processes that cause secondary HTN are known. On the other hand, those related to essential (main) HTN are much less well characterized. The cardiac output gradually recovers to normal as the total peripheral vascular resistance (TPR) increases. To explain this, the three following hypotheses have been put forth: (1) because the kidneys are unable to excrete sodium, natriuretic hormones including atrial natriuretic hormone are released, which speeds up salt excretion and increases TPR. (2) Vasoconstriction, salt and water retention, and hyperactivity of the renin-angiotensin system. HTN results from an increase in blood volume [31]. (3) A SNS that is hyperactive and causes an accelerated stress reaction [32]. (4) Furthermore, HTN is known to be highly genetic and polygenic (caused by numerous genes), and a few potential etiological factors have been proposed [33]. HTN researchers have recently become more interested in the relationship between vital HTN and long-term endothelial damage. However, it is unclear whether endothelial changes occur before HTN or as a result of long-term high BP. High BP is a major contributor to the risk of renal failure, coronary artery disease, and stroke on its own [6]. A lot of elements in the cardiovascular system influence BP, including cardiac output, blood volume, arterial tone balance, and so on. BP is kept in a healthy range by natriuretic peptides, the renin–angiotensin–aldosterone system (RAAS), endothelial cells, the immunological system, and the SNS. Any imbalance in the neurohumoral system's constituent parts might directly or indirectly lead to an increase or decrease in the average BP. A long-lasting imbalance also damages the cardiovascular system in addition to the target organ (such as CKD and left ventricular hypertrophy). Potassium channels [34] (Fig. 2), nitric oxide (NO) (Fig. 3), the renin-angiotensin system (Fig. 4), reactive oxygen species (ROS) (Fig. 5), and calcium ions (Fig. 6) are all physiological effectors that impact vascular tone, and any imbalance in these variables can lead to HTN. The bioavailability of NO is also decreased by superoxide, and superoxide generation is increased through uncoupled endothelial nitric oxide synthase (eNOS), which increases OS, a primary contributor to HTN [35]. Narrowed arteries, higher cardiac output, and inadequate kidney function all contribute to the pathophysiology of HTN, which results in raised BP. Targeted therapy and risk reduction are made easier with an understanding of these mechanisms.
As shown in (Tables 1 and 2), nature has developed or inspired a variety of lead compounds that can successfully alter these features. The information in the section that follows relates to popular traditional herbs that might have antihypertensive properties.
5 Medicinal plants and their bioactive compounds having hypotensive/antihypertensive effects
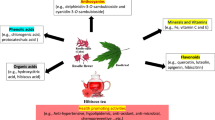
The medicinal plants, bioactive compounds used to treat HTN because of have antihypertensive activity (Fig. 7). Garlic, hibiscus, and omega-3 fatty acids are just a few examples of medicinal plants, herbs, and substances that have hypotensive properties. Utilizing these natural therapies can enhance existing medical procedures and improve cardiovascular health.
5.1 Allium sativum
5.1.1 Allium sativum: (Family: Alliaceae; Common name: Garlic)
Garlic known as Allium sativum is one of the first recognized medicinal plants, and it has been used for many years to cure a range of human diseases [93]. For many years, garlic has been used to treat many heart diseases, particularly hyperlipidemia. According to mythology, it possesses hypotensive properties as well. NO is thought to relax smooth muscles and dilate blood vessels when it is produced more frequently. The major chemical constituents of garlic is allicin (01) that provides its distinctive smell and many of its health benefits [94]. Formulations including garlic were observed to lower BP in HTN patients more effectively than a placebo [94]. Lower BP, a considerable reduction in lipid peroxidation, NO, and 8-hydroxy-2-deoxyguanosin, as well as an elevation in antioxidant vitamins, were all found (C and E). In this investigation, garlic was found to have a positive cardioprotective effect in essential HTN [95]. Alliums should only be used sparingly since they inhibit thiol group enzymes, which are inhibited by allyl and related sulfoxides [96]. Garlic has antioxidant characteristics that make it a promising treatment for HTN. Garlic consumption could be a healthy and supportive method of controlling BP.
5.2 Apium graveolens
5.2.1 Apium graveolens: (Family: Apiaceae; Common name: Celery)
The apiaceae family includes the annual or perennial plant Apium graveolens L., which grows in tropical and subtropical regions of Africa, Asia, and Europe [97]. Because celery affects the liver, which is associated to one type of HTN in Chinese belief, it is helpful for HTN [98]. Apium graveolens seeds can be utilized as a secure and efficient treatment for high BP, as shown by the significantly different BP readings in humans before and after treatment [99]. By combining fresh celery juice with vinegar, HTN-related headaches, shoulder pain, and dizziness can be relieved. Furthermore, it is used to treat climacteric HTN and HTN associated with pregnancy [100]. Plants are used to cure diseases because of their composition and therapeutic characteristics, which requires ongoing study into other beneficial and undiscovered aspects of them [97]. Apium graveolens used to decrease BP by relaxing blood vessels.
5.3 Coleus forskohlii
5.3.1 Coleus forskohlii: (Family: Lamiaceae; Common name: Karpurvali)
For a variety of conditions, Coleus forskohlii is frequently used internationally. The leaves have emmenagogue, diuretic, and expectorant properties in Egypt and Africa. In Brazil, it is used to treat intestinal issues and as a stomach benefit [101]. On isolated rabbit and cat hearts in vivo, it has a favorable inotropic effect at low dosages. Although it does not affect the guinea pigs' bronchial musculature, coleonol has a spasmolytic impact on the gastrointestinal smooth muscle in many other species. Coleonol has a depressant effect on the central nervous system when taken in large dosages [102]. In addition, coleonol has non-specific spasmolytic effects on the smooth muscles of the gastrointestinal tract in a variety of species, but not on the guinea pig's bronchial musculature [102]. Coleus forskohlii can be used to dilate blood vessels and improve heart function, perhaps assisting in the treatment of HTN. Combining it with medical advice can be advantageous.
5.4 Crataegus pinnatifida
5.4.1 Crataegus pinnatifida: (Family: Rosaceae; Common name: Chinese Hawthorn)
The Chinese hawkhorn known as Crataegus pinnatifida is readily available and has a long history of usage in traditional medicine [2]. It has been proven in pharmacological and clinical tests to reduce BP. The two main antioxidant compounds found in hawthorn, flavonoids (12), and oligomeric procyanidins, are what primarily contribute to the fruit's positive benefits on the heart. By lowering BP, boosting circulation, and preventing plaque buildup on arterial walls as well as blood clot formation in the brain, heart, and arteries. The alkaloid Rhynchophylline which is found in cat's claw, may help to prevent strokes and heart attacks [103]. Intravenous administration of the extract preparation decreased BP in rabbits that had been anesthetized for up to 3 h [104]. It was discovered that the principle causing hypotension. The medication of Crataegus pinnatifida has a broad impact on the cardiovascular system. One of the negative effects of nitrous oxide stimulation is hypotensive activity via vasorelaxation [105]. It exerts antioxidant and tonifying effects on cardiac myocytes [106]. The majority of experimental findings support the fact that Crataegus pinnatifida has potent pharmacological effects when used alone, but given current understandings of disease pathophysiology [2]. Hawthorn has advantages for the heart, including lowering BP. Along with medical counsel, including hawthorn into one's lifestyle may help with a complete approach to managing HTN.
5.5 Daucus carota
5.5.1 Daucus carota: (Family: Umbelliferae; Common name: Carrot)
The most significant root vegetable plant farmed worldwide is the edible carrot, Daucus carota, which primarily consists of two parts: the stem and the root. The majority of the root is made up of periderm, which has an inner core and an outside cortex that is pulpy [107]. It has been used to treat HTN in conventional medicine. According to in vitro tests, these substances reduced arterial BP in rats under anesthesia in a dose-dependent manner (1–10 mg/kg). Both substances showed a dose-dependent (10–200 g/mL) inhibitory impact on rabbit aorta contractions caused by K+ and spontaneously beating guinea pig atria at comparable doses [108]. Daucusol and daucuside, two new guaiane-type sesquiterpene terpenoids with an exciting epoxy unit, were found in the fruits of Daucus carota [109]. The investigations on carrot pomace stated above lead to the conclusion that more healthy products could be developed by utilizing carrots' functional, nutritional, and medicinal properties [110]. Carrots have nutrients that may help with BP control. While not a cure-all, including carrots in a balanced diet may improve cardiovascular health in general.
5.6 Desmodium styracifolium
5.6.1 Desmodium styracifolium: (Family: Leguminosae; Common name: Osbeck)
Traditional Chinese medicine (TCM) has used the Chinese herb Desmodium styracifolium [111] extensively to treat urolithiasis with little to no negative effect [112]. When administered intravenously to dogs under anesthesia, preparations produced from the plant's dry leaves and stem enhanced coronary circulation reduced arterial BP, slowed heart rate, and reduced cardiac oxygen consumption [104]. It is investigated that the in vivo and in vitro cardiovascular pharmacology of Desmodium styracifolium [111]. The extract assisted in loosening methoxamine-pre-constricted isolated helical tail artery strips. Desmodium styracifolium extracts had a favorable chronotropic effect with no visible impact on contractile force. The bioassay is based on the measurement of ACE inhibition from the enzymatic cleavage of the substrate dansyltriglycine into diglycine and dansylglycine, which is labeled with a chromophore and a fluorescent dye [111]. Further research is required before considering it a primary treatment.
5.7 Lycopersicon esculentum
5.7.1 Lycopersicon esculentum: (Family: Solanaceae; Common name: Tomato)
The tomato known as Lycopersicon esculentum L. is one of the most popular vegetables and the second-largest vegetable production in the world. It is an essential part of the "Mediterranean diet," which is closely linked to a lower risk of chronic degenerative diseases [113]. In people with mild, untreated HTN, a tomato extract called Lyc-O-Mato has been shown to reduce BP [114]. Treatment compliance was fairly high, and there were no documented negative effects of the medication [115]. As a result of tomato supplementation, OS is decreased and alterations in miRNA expression are induced. Additionally, the cross-sectional area decrease and improved diastolic function may be the result of these changes [116]. Tomatoes have elements that may help control BP. While not the only answer, including them in a balanced diet may improve cardiovascular health in general.
5.8 Pinus pinaster
5.8.1 Pinus pinaster: (Family: Pinaceae; Common name: Maritime Pine)
Pycnogenol is a French maritime pine bark extract. It has been utilized all over the world as a food supplement, herbal medicine, and sustenance for many different degenerative conditions [117]. The most frequent conditions it is used to treat include vascular diseases and venous insufficiency. However, several diseases, including HTN, are being researched about it. Taking 200 mg of pycnogenol per day can help people with mild HTN decrease their BP. Blocking ACEs is thought to be the mechanism of action [118]. After 8 weeks of using Pycnogenol supplements, mildly hypertensive patients who did not need normal pharmacological medication therapy experienced a significant drop in SBP and serum thromboxane B2 levels [119]. For better cardiovascular health, beneficial integration of it could supplement current approaches.
5.9 Punica granatum
5.9.1 Punica granatum: (Family: Lythraceae; Common name: Pomegranate)
Iran is the original home of the pomegranate that is known as Punica granatum L. The medical characteristics of this plant, in addition to its use as a fruit, have recently caught the attention of researchers from many different nations [120]. Pomegranate juice is gaining popularity as a fruit beverage. Studies show that pomegranate decreases ACE activity [121] by 37%. There have been a variety of outcomes from clinical studies. Following a year of daily consumption of 50 mL of pomegranate juice, an experiment found a lowering effect on SBP. Following 3 months of daily juice consumption of 240 mL, a different trial found no benefit [122]. Pomegranate can be used to help to regulate BP. Although it is not a sole treatment, using it in a healthy diet may help maintain cardiovascular health.
5.10 Raphanus sativus
5.10.1 Raphanus sativus: (Family: Cruciferae; Common name: Radish)
Radish known as Raphanus sativus is a root vegetable that is grown and consumed all over the world and is regarded to be a staple of the human diet [123]. Atropine's inhibitory impact was lessened in tissues that had been administered it, demonstrating that it had a stimulating effect on the heart that was resistant to blocking by serotonin and adrenergic receptors. In the rat aorta with intact endothelium, it stopped atropine-blocking phenylephrine-induced contractions. The extract's safety in mice was demonstrated at concentrations of up to 10 g/kg. The plant's circulatory inhibitory effects can be used to treat HTN since they are mediated by muscarinic receptor activation [124]. Inhibiting lipid peroxidation, promoting liver and RBC catalase, and inhibiting XOD activities in animal tissues are all possible effects of the flavonoids (12) and vitamin C in radish [125]. The radish provides minerals that could be useful for lowering BP. Although their benefits are probably minor, they should be taken into account as part of a complete strategy for heart health.
5.11 Rhaptopetalum coriaceum
5.11.1 Rhaptopetalum coriaceum Oliver: (Family: Scytopetalaceae)
An old method of treating HTN involves making a decoction of the plant's stem bark and soaking it in locally produced gin [126]. In addition to reducing noradrenaline and KCl-induced Ca2+ influx, this was achieved by inhibiting potential sensitive channels and receptor-operated channels. According to the in vitro data, nifedipine may not be as effective as the ethanol extract of Rhaptopetalum coriaceum as a calcium channel blocker [127]. It indicates that the endothelium is required for the relaxation response to the extract, which may be related to an intracellular rise of cyclic guanosine monophosphate (cGMP) [128]. The Coriaceous graptopetalum has unexplored possibilities for treating HTN. Traditional applications imply efficacy, although there is a lack of scientific support. Before considering its use, precaution and additional investigation are advised.
5.12 Solanum sisymbriifolium
5.12.1 Solanum sisymbriifolium: (Family: Solanaceae; Common Name: Sticky Nightshade)
In traditional Paraguayan medicine, the perennial herb Solanum sisymbriifolium Lam. is used as a diuretic and an antihypertensive [129]. Traditional medicine in Paraguay uses Solanum sisymbriifolium a perennial herb with diuretic and antihypertensive effects. Rats with HTN conditions were used to test the crude hydroalcoholic root extract's hypotensive effects. BP significantly decreased after the extract was injected intravenously into rats with HTN. A dose-dependent hypotensive response was observed after the extract was administered orally to conscious hypertensive rats [130]. Nuatigenosido was identified as one of the extract's potentially active components in another investigation. The BP of Rats was lowered by nuatigenosido at doses of 100 g/kg and 1 mg/kg intravenously, while frog atrial myocytes' exceed amplitude was raised at 107 M. The outcomes suggested that nuatigenosido is crucial for the medicinal effects of the herb [131]. It can be argued that the clinical efficacy characteristics of this plant, such as hypotension, appear to be partially mediated by the vasodilatation and cardiotonic action generated by nuatigenosido [132]. The role of Solanum sisymbriifolium in the management of HTN. For managing BP, it is wise to seek advice from medical experts and use treatments.
5.13 Theobroma cacao
5.13.1 Theobroma cacao: (Family: Malvaceae; Common names: Chocolate)
The rainforest regions of tropical America, from Mexico to Peru, are where cacao is native. The bark of cacao makes up around 84 percent of its composition, with the remaining percent consisting of seeds, pulp, and other ingredients [133]. Due to its high flavonoid (12) content, cocoa powder is used to help prevent CVD. Chocolate flavonoids (12) boost NO generation, improve vasodilation, and reduce endothelial dysfunction. An increasing amount of scientific evidence shows that eating 46 to 105 g of chocolate (Theobroma cacao) each day, with 214 to 400 mg of cocoa polyphenols, will reduce BP by roughly 5 mmHg and 3 mmHg, respectively [134]. The biological effects of cocoa polyphenols are garnering increasingly more attention. In fact, cocoa's high polyphenol concentration and extensive use in many foods make it particularly interesting from a nutritional and pharmacological perspective [135]. Flavonoids (12) found in Theobroma cacao, may have a beneficial effect on BP. However, due to its high-calorie content and other variables, it should only be used as a supplement, not as an individual treatment.
5.14 Uncaria rhynchophylla
5.14.1 Uncaria rhynchophylla: (Family: Rubiaceae; Common name: Cat's Claw herb)
Uncaria rhynchophylla is one of the original plants used to make the significant Chinese crude medication Gou-teng, which is primarily used to treat brain disorders, HTN, convulsions, eclampsia, and epilepsy [136]. Uncaria rhynchophylla has been used in TCM to decrease BP and treat a variety of neurological conditions. The indole alkaloid hirsutine, which affects Ca2+ channels, is responsible for the hypotensive effect [137]. By affecting both the voltage-dependent Ca2+ channel and the Ca2+ storage, hirsutine is expected to lower intracellular Ca2+ levels [138]. Different from Uncaria rhynchophylla and its equivalent, a methanol extract of hooks from an Uncaria species demonstrated a potent and pervasive hypotensive effect in rats [139]. The extraction of the 3-indole alkaloid, glycoside, cadambine, dihydrocadambine, and isodihydrocadambine from the extract spurred more research. While the cadambine was demonstrated to be inert, the two were found to be the hypotensive principles [140]. Due to its effects on blood vessel relaxation, Uncaria rhynchophylla has potential effects as a therapy for HTN. Before considering it to be a reliable alternative, further thorough research is required.
5.15 Viscum album
5.15.1 Viscum album: (Family: Santalaceae; Common name: Mistletoe)
A perennial, hemiparasitic shrub, epiphytic, called Viscum album that thrives on a variety of woody plant species, it is evergreen. It is a known pathogen, a medicinal plant, a native of Europe, and a mythological icon [141]. In the isolated and diffused heart model developed by Langendorff, aqueous leaves extracts of Viscum album significantly increased coronary vasodilator activity. The aqueous extract of Viscum album has some physiologically active elements that could operate as an inducer of the NO/soluble guanylate cyclase pathway [142]. The potential of extract to reduce BP was inhibited by propranolol. Atropine has no influence the extract's BP depression. The noradrenaline-blocking extract raised BP [118]. Mistletoe extract exerts an antihypertensive effect without changing HR, showing that sympathetic processes are involved [143]. Only one drawback is founded to Viscum album therapy, which needs to be confirmed by toxicology tests on cardiac muscle cell lines. This drawback is an elevated blood creatine phosphokinase myocardial band level [144]. Due to its vasodilatory effects, mistletoe has been studied for HTN. Before incorporating it into medical treatments, though, its effectiveness and safety must be thoroughly studied.
5.16 Zingiber officinale
5.16.1 Zingiber officinale: (Family: Zingiberaceae; Common name: Ginger)
The spice ginger, also known as Zingiber officinale, is used in food and medicine throughout the world. The plant has several compounds that provide it with medical activities, including those that are anti-arthritic, antidiabetic, anti-fungal, and anti-cancer [145]. It improves blood circulation by relaxing the muscles that surround blood vessels. Anesthetized rats' arterial BP dropped dose-dependently (0.2–3.0 mg/kg) after being given a crude extract of ginger. It was determined that, like verapamil, Zo. Cr blocked Ca2+ channels by skewing the Ca2+ dose–response curves to the right. The inhibition of voltage-dependent calcium channels is thought to be the cause of ginger's BP-lowering effect [146]. To deoxycorticosterone acetate salt-induced hypertensive rats, pet ether extract (50 mg/kg/day; po), lowered BP, while PE (10 mg/kg/day; po) and KGE (30 mg/kg/day; po) to fructose-induced hyper. The mechanism of action could be aided by the serotonergic antagonistic feature [147]. Only a few human trials of ginger's hypotensive impact have been conducted, and most of them utilized a modest dose with unclear findings [148]. Ginger has hypotensive effects. Although it is not a solo treatment, adding ginger to a healthy diet may help manage HTN naturally.
5.17 Andrographis paniculata
5.17.1 Andrographis paniculata: (Family: Acanthaceae; Common Name: Kalmegh)
Astragalus paniculatus in China, India, and other Southeast Asian nations, is a medicinal plant that has historically been used to cure colds, fevers, laryngitis, and several infectious disorders, from malaria to dysentery and diarrhea [149]. This is a traditional Chinese and Southeast Asian medicinal plant used to cure colds, fevers [150], upper respiratory and gastrointestinal tract infections, hepatitis, herpes, and CVD [76]. ACE, β-adrenoceptors, and autonomic ganglion receptors are all inhibited by andrographispaniculate [121, 151]. Its extracts contain a number of diterpenoid chemicals, including 14-deoxy-11,12-didehydroandrographolide, andrographolide (02), and 14-deoxyandrographolide (03) [152], which have anti-inflammatory, antibacterial [153], antioxidant, and hypotensive properties. Its chloroform extract can activate NO synthesis and, as a result, promote NO production in endothelial cells, causing smooth muscle relaxation by blocking Ach action [154]. Through lowering ROS and ACE activity, Astragalus paniculatus lowers BP in impulsively hypertensive rats [76]. According to Awang et al. [152] In isolated rat hearts, 14-deoxy-11,12-didehydroandrographolide lowered vascular resistance. The researchers found that a crude extract containing a significant amount of 14-deoxy-11,12-didehydroandrographolide significantly lowers BP because it increases NO release, which causes vasodilation. Additionally, 14-deoxy-11,12-didehydroandrographolide inhibits voltage-gated Ca2+ channels, lowering Ca2+ concentrations inside the cell. Endothelial protection and smooth muscle relaxation were achieved as a result of the L-type Ca2+ current and high K+ activation pathways being inhibited by the Andrographis paniculata chloroform extract. ACE inhibitory activity, β-adrenoceptors, and autonomic ganglion receptors are thought to be the mechanisms through which diterpenoid lactones as DDA from Andrographis paniculata exert their hypotensive effects [75, 155,156,157]. The ability of Andrographis paniculata to relax blood arteries makes it a potential treatment for treating HTN. Before deciding to use it as a primary treatment, additional reliable clinical data is required.
5.18 Bidens pilosa
5.18.1 Bidens pilosa L.: (Family Asteraceae; Common Names: Broom Stick)
An annual herb with a global reputation for being used to treat several diseases, Bidens pilosa L. has been extensively researched for the biological activity of its extracts, fractions, and isolated components [158]. The bioactive components have been used to treat bacterial, cancer, obesity, HTN, malaria, and CVD [43, 159], making it a popular plant today. There are at least 60 flavonoids (12) in Bidens pilosa, and they contains a range of chemical components [159]. As a result, preparations of this plant are extensively used as medicine to treat around 40 different diseases [159, 160] via a variety of anticipated [161] processes, including vasodilation, lipid profile improvement, free radical scavenging, insulin-sensibility, calcium channel blocker, and so on [73, 162,163,164]. Quercetin (08) improves endothelial function by boosting NO production and/or bioavailability. It has been confirmed quercetin's (08) potential to lower and prevent BP [159, 161, 162]. High doses of Bidens pilosaca leaf extracts lower plasma creatinine levels, which enhance plasma cholesterol levels. As a result, they theorized that Bidens pilosa's hypotensive action is independent of insulin sensitivity [165]. In multiple normotensive and hypertensive rat models (caused by fructose), the aqueous and CH3Cl leaf extracts of Bidens pilosa can reduce and prevent high BP during the course of a 3-week continuous therapy [161, 166]. Dimo et al. and Bartolome et al. [159, 162] have demonstrated the vasorelaxant effects of Bidens pilosaca, and Nguelefack et al. have seen increasing levels of a neutral to induce relaxation in noradrenaline and potassium chloride pre-constricted rat aortas [167]. Their theory was that calcium channel antagonism or a cyclooxygenase metabolite produced vasodilation [155]. They went on to assert that the vasodilation process had nothing to do with the ATP-dependent K+ channel [159, 167]. Due to its diuretic characteristics, the traditional treatment of Bidens pilosa may help with HTN. Before considering its use for BP control, it is imperative to consult with healthcare professionals.
5.19 Camellia sinensis
5.19.1 Camellia sinensis: (Family: Theaceae; Common Name: Tea)
One of the oldest and most widely favored medicinal drinks consumed worldwide is green tea. The leaf of the "Camellia sinensis" plant is used to make this product [168]. Catechins (09) are the most common flavonoids (12) found in tea (−)-epicatechin-3-gallate (ECG), (−)-epicatechin (EC), (−)-epigallocatechin-3-gallate (EGCG) (04), (−)-epigallocatechin (05) (EGC, primary component) [169]. As a result of an enzymatic process, the catechins (09) are transformed into flavins, and the origins are known to be effective vasodilators. Due to enhanced NO release and a decrease in OS and dimethylarginine levels, catechins (09) are also responsible for a considerable improvement in blood flow [170]. Anti-diabetic, anti-inflammatory, antibacterial, antihypertensive, and anti-cancer activities have been found in Camellia sinensis aqueous extract [171]. People who drank green and black tea regularly had a lower risk of HTN [169]. Based on a meta-analysis, the regular tea drinking had a substantial decreasing effect on DBP [172], however other studies found that it is concentration dependent [50]. Drinking concentrated, only green tea regularly can lessen the risk of CVD mortality [48]. Daily ingestion of black tea extract for seven days decreases SBP because the o-methylated EGCG content inhibits the ACE. After drinking lyophilized green tea extract, mildly hypertensive patients' SBP (4.9 mmHg) and DBP (4.7 mmHg) decreased significantly [173, 174]. Green tea can lower BP in a variety of ways, including maintaining a balance of vasoconstricting, vasodilating, and hyperpolarizing hormones [172]. It increases NO production by stimulating antioxidant enzymes and inhibiting pro-oxidant enzymes, which helps to improve heart function and regulate ROS production [175]. Studies have shown that medicinal plants can treat HTN using a variety of mechanisms, including vasodilation, NO production, inhibition of the renin-angiotensin system, activation of intracellular cGMP, and expansion of the vasodilator effect [176]. Tea made from Camellia sinensis contains substances that may only slightly help with BP control. Moderate tea consumption may support a healthy lifestyle for cardiovascular health.
5.20 Crataegus spp.
5.20.1 Crataegus spp.: (Family: Rose; Common Name: Hawthorns)
For many years, different cultures have used the medicinal benefits of hawthorn (Crataegus spp., a genus with about 300 species) for a range of therapeutic purposes [177]. Hawthorn plants have been used in traditional medicine to treat CVDs for a long time. Hawthorn medicine (500 mg daily for 10 weeks) can lower DBP in hypertensive patients [178]. A considerable BP decrease occurs only after the medication is given in higher doses over a longer period [179]. These plants also include oligomeric proanthocyanidins such as procyanidin in, procyanidin B-2, and flavonoids (12) like vitexin, rutin, and others. Endothelial cells and VSMCs respond well to hawthorn extracts [180]. The extract reduces the levels of VCAM-1, IL-6, NF-κB, and iNOS, all of which are pro-inflammatory [86, 181]. More in vivo, in vitro, and clinical experiments are needed to investigate the connection between the chemical composition of these plants, particularly hawthorn, and their modes of action in the treatment of different disorders [88, 182]. Hawthorn has potential in the treatment of HTN through promoting heart health and vasodilation. To improve standard treatments, it should be used carefully and combined with medical advice.
5.21 Cymbopogon citrates
5.21.1 Cymbopogon citrates: (Family: Gramineae; Common Name: Lemongrass)
Cymbopogon citratus, also known as lemongrass, is a plant that is frequently used in folk medicine for a variety of diseases. Yet further research is needed to fully understand its anti-hypertensive potential [63]. Researchers have found a variety of compounds in the herb's leaves and stems, including flavonoids (12), alkaloids, essential oil, tannins, phenols, saponins, and anthraquinones [183]. Citral, the major ingredient in Cymbopogon citratus, has antibacterial, antioxidant, chemoprotective, and antispasmodic properties when used alone or in combination with other ingredients. Citral prevents L-NAME from attenuating the generation and release of NO, resulting in vasorelaxation. Furthermore, the extract from the leaves may affect prostacyclin production, resulting in relaxation. [184]. In rats, Cymbopogon citratus fresh leaf extract, when combined with other herbal treatments like Citrus medica fruits extract and Persea americana fresh leaf extract, can reduce HTN caused by sugar and ethanol [63]. Ray discovered that twice-daily therapy with a lemongrass decoction reduced mean arterial pressure significantly [185]. Lemongrass oil can prevent ROS from forming. Citral suppresses iNOS and NF-κB activity, as well as having anti-inflammatory characteristics [72]. To increase the development of new herbal medicines and study their therapeutic potential, attention to medicinal plants has recently been focused on pharmacological research and phytochemical screening of secondary metabolites [186]. Cymbopogon citratus has a long history of supporting BP control. Because of its potential for minor effects, it can be used in addition to more well-known HTN control techniques.
5.22 Nigella sativa
5.22.1 Nigella sativa: (Family: Ranunculaceae; Common Name: Black Cumin)
Asthma, diarrhea, and dyslipidemia are just a few of the diseases that are treated and prevented with the seeds of Nigella sativa throughout the world in folk (herbal) medicine [187]. When Nigella sativa is ingested, BP is reduced [188, 189]. Most of the positive effects of seeds are due to thymoquinone, a major active component in black seed essential oil [190]. Another active component of Nigella sativa, thymol (16), has been demonstrated to reduce BP in endothelium cell membranes via an endothelial-independent mechanism (inhibition of calcium ion influx through calcium channels), followed by vasorelaxation [189]. The other bioactive elements include proteins, fatty acids, alkaloids, antioxidants, essential oils, flavonoids (12), and antioxidants. In people with mild HTN, Wong found that taking an Nigella sativa extract twice a day for 8 weeks dramatically lowered BP [191]. Nigella sativa may be effective in the treatment of HTN because it reduces ROS [192]. The oil of Nigella sativa can lower both DBP and SBP [193]. According to Ahmad et al. [71], TQ (Thymoquinone) induces vasodilation by lowering the production and release of COX-1 and COX-2 metabolites. Black cumin functions as an anti-inflammatory agent by reducing NF-κB [155, 194]. There is evidence to support including Nigella sativa and its bioactive ingredients in your diet regularly to improve your health [71]. The antioxidant and vasodilatory properties of the Nigella sativa indicate a potential for HTN. Its function should be considered supplemental, and more research is required before making clear suggestions.
5.23 Panax ginseng
5.23.1 Panax: (Panax ginseng, Panaxquinquefolius; family: Araliaceae; Common Name: Japanese Ginseng, Asian or Korean Ginseng)
Panax ginseng which is native to Korea and China and is used as an herbal treatment [195]. For centuries, the Panax ("all healer") was thought to be capable of treating all human diseases [196]. Panax roots, in both solid and liquid form, have long been used in folk medicine for a variety of therapeutic and pharmacological purposes [197]. The biological effects of this medicinal plant include a low BP antidiabetic, anticancer, antioxidation, vasorelaxation, anti-carcinogenic, anti-allergic, anti-inflammatory, and more [196, 198]. Although it is generally known that ginseng reduces BP, it is claimed that it can also raise BP to regulate hypotensive circumstances, most likely by altering vascular features, changing the autonomic nervous system, or adapting the baroreflex of arteries [199]. In both patients with moderate HTN and healthy people, it is discovered that ginsenoside from Panax ginseng has a substantially decreasing effect on SBP and DBP [200]. Ginsenoside Rg3 has a stronger effect on eNOS expression, which leads to more NO production and vasorelaxation [200]. Ginseng also inhibits the release of catecholamines from the adrenal glands, lowering BP [197]. Panax ginseng shows promise in the therapy of HTN. Although it is not a stand-alone treatment, its restricted use may support existing cardiovascular health treatments.
5.24 Salvia miltiorrhizae
5.24.1 Salvia miltiorrhizae: (Family: Labiatae; Common Name: Danshen)
TCM uses Salvia miltiorrhiza, also known as Danshen in Chinese. It is a Salvian perennial flowering plant that has long been valued for its use in TCM [90]. Root extracts have properties that are antimicrobial, and antioxidant, and that prevent CVD and inflammation [90, 91]. Danshen's roots extract reduces heart rate and SBP by boosting eNOS signaling synthesis and magnifying NO production to encourage vasodilation [201]. Tanshinone IIA causes vasodilation without the need for endothelial cells to be involved [201]. The metabolite of Danshen increases intracellular Ca2+ influx through altering receptor and voltage-dependent calcium channels [202]. Danshen also inhibits ACEs, lowering BP [203, 204] which has been connected to antihypertensive effects in the lab [205,206,207,208]. It is hypothesized that RA and RS can successfully suppress Ang II expression, stop RAS activation, inhibit TGF-1 expression, postpone the progression of the RIF, and protect against HTN and damage to cardiac function [209]. The cardiovascular benefits of Salvia miltiorrhizae suggest that it may be useful in the treatment of HTN. Consideration might be given to incorporating it with medical advice to improve overall BP control.
5.25 Rauvolfia serpentina
5.25.1 Rauvolfia serpentina: (Family: Apocynaceae; Common Name: Devil Pepper)
For many years, the root of a flowering plant belonging to the Apocynaceae family called Sarpgandha has been utilized in Ayurveda to cure several diseases that at first glance seem to have little in common with one another [210]. Rauwolfia Treatment of HTN is the most common application of serpentine (10). It slows down nervous system activity, slows down heartbeat, and opens up blood vessels. Rauwolfia also includes steroids, flavonoids (12), and tannins in addition to fatty acids, alcohols, sugars, and glycosides. All parts of the plant contain indole alkaloids, but the root bark contains higher concentration. Ajmalinine, ajmalidine, ajmaline, and raubasine, papaverine are a few of the indole derivatives that have been identified [211]. The most popular indole derivative, reserpine (11), has antihypertensive properties and lowers both SBP and DBP [210,211,212,213]. Storage vesicles in the cell become irreversibly attached to VMAT2 and 'leak' their contents into the cytosol, including monoamine, where MAO-A enzymes then contaminate the cytosol. Monoamine modification is age-independent [214]. The entire topic of Rauvolfia serpentine therapy for HTN has been studied up to this point, including discussions on the history of the plant, its various species and types, nomenclature, geographic distribution, chemistry, pharmacologic actions, and clinical studies, reported on the subject from all over the world [8, 215,216,217]. In the past, Rauvolfia serpentine was used to treat HTN. BP is lowered by its primary ingredient, reserpine (11), although side effects and current alternatives necessitate medical supervision and careful evaluation.
5.26 Stevioside
The leaves Stevia rebaudiana (Bertoni) are used to make the natural sweetener stevioside. The literature on Stevia is discussed, as well as the presence of its sweeteners, their biosynthetic process, and any toxicological implications [218]. In both animal and human experiments, it has been shown to reduce BP. Intravenous doses of 50, 100, and 200 mg/kg produced dose-dependent hypotensive effects on both SAP and DAP in awake SHRs. The most significant reductions in SAP and DAP were 31.4 to 4.2 percent and 40.8 to 5.6 percent, respectively [219]. Crude stevioside at doses up to 15.0 mgkg1 d1 had no antihypertensive effect in previously untreated mild HTN patients [220]. Stevioside inhibits extracellular Ca2+ influx and the generation of a vasodilator prostaglandin, which may alter vascular resistance. Stevioside also causes diuresis and natriuresis, resulting in a decrease in extracellular fluid volume [221]. This work demonstrated the efficacy of stevioside as a natural antihypertensive agent and suggested that its hypotensive action may be caused by a Ca2+ influx inhibition mechanism [221]. Due to its possible vasodilation, stevioside may have a minor effect on BP. However, its efficacy in managing HTN is still limited, necessitating additional study and medical advice.
5.27 Orientin
Water-soluble flavonoids (12) include orientin (15). With two ether groups and one ketone group, orientin (15)'s chemical structure reveals that phenol groups make up the majority of its composition [222]. Phyllostachys nigra bamboo leaves, which have been used in Chinese medicine, are the source of the substance known as "orientin (15)." Additionally, orientin (15) inhibits vasoconstriction caused by norepinephrine, CaCl2, and KCl in a concentration-dependent, non-competitive manner as well as contractions brought on by phenylephrine during both the initial fast release and sustained phases. Orientin (15) can encourage endothelial cells to generate NO [223, 224]. The polyphenolic chemicals found in BS were therefore the source of the ACE inhibitory actions of the methanol extracts of BS [225]. Through its antioxidant and vasodilatory properties, the flavonoid (12) orientin (15), which is present in many plants, has the potential to treat HTN. Even though it appears promising, additional research is required to determine its effectiveness and safety in the treatment of HTN. For advice, always seek the advice of a healthcare professional.
5.28 Ostruthol
Natural products have been utilized as part of herbal treatments for millennia to alleviate and treat a variety of diseases. Their unusual chemical diversity provides a wide source of drug-like compounds with a variety of biological actions, accounting for their substantial contributions to drug development [226]. Its activity against K+-spasms is much stronger than its activity against norepinephrine contractions [227]. However, more research is necessary before recommending it due to its clinical significance and safety. It might be an additional choice for managing HTN, but consulting with a healthcare professional is important.
5.29 Flavonoids
Numerous animal studies conducted over the past 10 years have demonstrated the flavonoids' (12) positive effects on the cardiovascular system. The anti-HTN actions of flavonoids (12) or extracts of plants rich in flavonoids (12), as well as their potential mechanisms, were therefore discussed. Flavonoids (12) have been identified to have a variety of potential effects on BP. The clinical use of flavonoids (12) as an adjuvant or core drug is not supported by the extensive evidence of their potential in the treatment of HTN, in addition. Therefore, the discussion also takes into account the synergistic effects of flavonoids (12) with other medications, pharmacokinetic research, clinical trials, and the safety of flavonoids (12) [228]. Among the many bioactive polyphenolic chemicals found in dietary plants and herbs, flavonoids (12) are a varied group. Flavonoids (12) have cardio-vasculoprotective properties that can delay or stop the progression of several CVDs, including HTN. Anthocyanin consumption and risk of myocardial infarction, flavanone intake and risk of ischemic stroke, and flavanols intake and risk of type 2 diabetes mellitus. Human randomized controlled trials (RCTs) demonstrate that quercetin (08) and catechins (09) significantly decrease BP. By enhancing NO bioavailability, lowering endothelial cell OS, or altering vascular ion channel function, flavonoids (12) exert their antihypertensive effects of antihypertensives and potential ways through which they control BP. The security of these substances as well as their prospective application in the control and treatment of HTN [229]. Due to their antioxidant and vasodilatory effects, flavonoids (12), which are widely present in fruits and vegetables, show potential in the control of HTN. Their inclusion in diets may improve cardiovascular health.
5.30 Polyphenol
Dietary polyphenols affect BP, HTN, and vascular health. There is proof that several foods high in polyphenols, such as berry fruits with high anthocyanin content, cocoa and green tea with high flavan-3-ol (17) content, almonds and pistachios with a high hydroxycinnamic acid content, and soy products with high isoflavone (13) content, can lower BP [230]. The NO-cGMP pathway and ACE inhibition are involved in the antihypertensive phytotherapy of polyphenol-rich diets for defense and enhancing endothelial function with arterial relaxation. OPCs trigger a laminar shear stress response in endothelial cells, induce endothelium-dependent vasodilation, reduce the synthesis of the vasoconstrictor ET-1, and also block the activities of metalloproteinases such as ACE, hence reducing BP [231]. Polyphenols, which are present in foods like berries and tea, show promise in the treatment of HTN. They may improve overall cardiovascular health when included in diets.
5.31 Alkaloids
Uncaria rhynchophylla alkaloid has been demonstrated to have both a substantial protective impact on human umbilical vein endothelial cells (HUVECs) and an antihypertensive effect. Further research has shown that Uncaria rhynchophylla alkaloid reduces the inflammatory response in artery walls, hence inhibiting the release of intercellular adhesion molecule-1 (ICAM-1) and vascular cell adhesion molecule-1 (VCAM-1) [232, 233]. The design of selective inhibitors may be studied using phytopharmaceuticals, such as alkaloids, which have a rich medicinal history and diverse clinical applications. The study came across the alkaloids that have already been put to the test and their significant outcomes, which prompted additional research on these and many other alkaloids for the identification of new efficient and selective ACE inhibitors [234]. It is used to treat HTN. The significance of medical guidance and modern therapies is highlighted by current alternatives and probable adverse effects.
5.32 Lycopene
Indirectly increasing NO production in the endothelium, lycopene (14) decreases OS, acts as an antioxidant, and lowers BP. In 54 patients with moderate HTN who were already taking ACE inhibitors or calcium channel blockers after 6 weeks of tomato extract supplementation, there was a significant decrease in both SBP and DBP, indicating a role for lycopene (14) in the treatment of HTN [235]. Lycopene (14) supplementation was observed to lower SBP in prehypertensive and hypertensive patients in a meta-analysis, but it did not affect DBP [236, 237]. Lycopene (14), which is rich in tomatoes and has antioxidant effects, may help with BP control. While not a cure-all, consuming lycopene (14)-rich foods may improve cardiovascular health.
5.33 Cinnamon
Cinnamon (Cinnamomum Zeylanicum) has promising antihypertensive activities and may even increase the effectiveness of amlodipine in lowering BP. SBP was significantly reduced to an average of 145.57 2.6 mmHg as a result of co-administration with amlodipine, a drop of 15.48% when compared to the hypertensive control group. Cinnamon consumption significantly lowers BP levels in hypertensive rats and improves the PK characteristics of amlodipine [238]. Although it is used in food preparation for flavor and taste, cinnamon may also play a part in the regulation of BP and glucose metabolism. Therefore, adding helpful dietary ingredients, including cinnamon, that have favorable effects, in addition to lowering the number of ones that have negative effects on BP and insulin function, may help regulate BP [239]. Cinnamon has a limited ability to reduce BP. While adding it to a healthy diet may have some advantages, established therapy and medical supervision are necessary for complete HTN management.
5.34 Garlic
Garlic (Allium sativum) and several of its bioactive components may have an antihypertensive effect. With a particular focus on S-allyl cysteine and allicin (01), the review's objective is to give an in-depth discussion about the molecular, biochemical, and cellular rationale underlying the antihypertensive characteristics of garlic and its bioactive ingredients. The function of S-allyl cysteine and allicin (01), two garlic bioactive, in modifying several parameters thought to be involved in the development of HTN. OS, NO bioavailability, hydrogen sulfide generation, angiotensin-converting enzyme activity, NF-κB expression, and vascular smooth muscle cell proliferation are some of these markers [240]. By boosting NO production [241], inducing vasodilatation with hydrogen sulfide [242], and inhibiting angiotensin-converting enzyme activity, garlic extracts have been found to have anti-hypertensive actions. Garlic extract is a vasorelaxant and may lessen the atherogenic potentials of cholesterol in rats [243]. The essential vasodilatory and antioxidant effects of garlic point to possible applications in BP control. Garlic in the diet could successfully support current HTN treatments even though it is not a cure-all.
5.35 Omega-3 fatty acids
The evidence for the benefits of omega-3 PUFA on BP is inconsistent, and the reduction shown in many trials is moderate. However, older people and hypertensive participants showed greater reductions when supplementing with doses of up to 3 g/day of fish oil or blue fish. However, in hypertensive people, consuming omega-3 can lower cardiovascular risk by decreasing the rise in BP. Therefore, more clinical research is required to determine how supplementing with -3 PUFA reduces cardiovascular risk in a homogeneous population [244]. Impaired vasodilation involving the malfunctioning of several vasodilatory pathways is a common feature of HTN. BP and vasodilation can be lowered by PUFAs with an omega-3 chain [245]. Fish and dietary supplements containing omega-3 fatty acids have been shown to have cardiovascular advantages, including the potential to lower BP. Along with medical advice, including them in diets can help manage HTN.
5.36 Linalool
A common remedy for HTN, linalool (06) is a monoterpene alcohol found in various aromatic medicinal plants native to Brazil [246]. The function of linalool (06) and its underlying mechanism in VSMC physiology. After treating VSMCs with angiotensin II (Ang II), MTT and western blotting tests were used to determine whether linalool (06) had any impact on the induced proliferation and migration of VSMCs [247]. A natural substance called linalool that is present in several plants may be useful in treating HTN. It may aid in relaxing blood vessels and lowering BP. Before recommending it as a primary therapeutic choice, more research is required.
5.37 NG-nitro-l-arginine methyl ester
Investigations were conducted to determine the impact of an aqueous Mentha cordifolia extract on the oxidative status in NG-nitro-l-arginine methyl ester (07)-induced HTN [248]. The antihypertensive impact of Phragmanthera incana leaf ethanol extract in rats with HTN brought on by NG-nitro-l-arginine methyl ester (07) [249]. In the treatment of HTN, NG-Nitro-l-Arginine Methyl Ester may be crucial. Because it inhibits NO synthase, it aids in BP control. However, due to possible side effects, its clinical use should be done with care.
6 Antihypertensive seafood compounds
The seafood compounds used to treat HTN because of having antihypertensive activity.
6.1 Phlorotannins
"Phlorotannin" (1,3,5-trihydroxy benzene) (18) is a class of polyphenolic chemicals found in brown algae, accounting for around 5–12 percent of the dry mass [250]. They are commonly found in brown seaweeds, particularly the Ecklonia species, and offer a variety of advantageous biological properties, including anticancer, antiallergic, and antihypertensive benefits [250,251,252,253]. Given their prior discoveries, it is not surprising that phlorotannins are connected to the ACE I inhibitory activity postulated by Olivares-Molina and Fernández [254]. According to reports, Dieckol is a non-competitive ACE I inhibitor that induces NO generation in EAhy926 cells without having any cytotoxic effects. However, its inhibitory action is not equivalent to captopril's [255]. Although the alleged low IC50 appears to support this, the related standard deviation shouldn't be far larger than the IC50 mean value [256]. Ecklonia cava was the source of the 6,6′-bieckol (19) (Fig. 8). With an IC50 Kjellman is less active than phlorofucofuroeckol A at inhibiting the ACE enzyme. In addition, Ko et al. [257]. 6,6′-bieckol (19) may be effective in the management of HTN. It appears that a dibenzo-1,4-dioxin moiety is necessary for ACE I inhibition, even though the structure–activity connections for phlorotannins are still being worked out [256]. Phlorotannins are hypothesized to inhibit ACE I because of their capacity to bind to proteins and the consequent reduction in ACE I binding effectiveness [255]. Phlorotannin's ability to bind to proteins is determined by its length and structure; phloroglucinol pentamers or hexamers are clearly superior inhibitors [258, 259]. These approaches have mostly been employed for separation monitoring and qualitative profiles, with few data on quantification or quality control development reported [260]. Phlorotannins from some seaweeds have the potential to lower BP by altering the activity of BP-regulating enzymes. To determine their clinical value and safety, more research is needed.
6.2 Polysaccharides
Polysaccharides are natural resources for vitamins and medications, which have gained popularity in recent years. Natural polysaccharides have been proven to have less adverse effects, but their bioactivities were challenging to compare to those of manufactured medicines due to their inherent physicochemical features [261]. Endothelin1 is the main constituent of the endothelin system, interconnects with two types of endothelin receptors inside cells. Major CVDs have been demonstrated to be candidates for endothelin receptor inhibition, particularly of the ETA subtype [262,263,264]. Pelvetia canaliculata (Linnaeus) Decaisne & Thuret, a brown alga, can be used to make d-polymannuronic sulphate, a chemical of the carbohydrate type that has been shown in vivo to have both acute and preventative hypotensive activity. d-Polymannuronic sulphate had a therapeutic potency of 50 mg/kg, which was on par with captopril's (14 mg/kg) level [265]. Polysaccharides are classified as specialty chemicals rather than commodity chemicals, including both natural gums and their derivatives. They are used in industrial applications as thickening and sizing agents [266]. Seafood, such as seaweed, contains antihypertensive polysaccharides that may lower BP in a number of different ways. Before making a clinical recommendation, more research must be done on their therapeutic potential.
6.3 ACE inhibitory peptides
ACE [121] is a major enzyme in the renin-angiotensin system (RAS) that converts angiotensin (Ang) I into angiotensin II (Ang II), a vasoconstrictor that causes high BP [267]. Patients with HTN around the world are treated with ACE inhibitors. Synthetic chemical drugs that are frequently used in the treatment and prevention of HTN include captopril, enalapril, alacepril, and lisinopril [121, 268]. Peptides made from partially hydrolyzed food proteins by enzymes have drawn a lot of attention recently. Bioactive peptides with a wide range of health advantages have been created by food protein hydrolysates and have all been categorized and reported. These peptides can be produced during enzymatic digestion or food processing even if they are inactive inside the original protein sequence [121]. Natural source ACE [121] inhibitors improve the chances of dietary control of HTN. The structure of some sequences substantiated the presence of peptides with ACE-inhibitory, antioxidant and immunomodulatory activities [269]. By blocking the enzymes that control BP, ACE inhibitory peptides found in seafood have the potential to reduce BP. Their effectiveness and safety call for further scientific investigation for clinical use.
6.4 Peptides from marine fish
Using the proper proteolytic enzymes, bioactive peptides can be isolated from the enzymatic hydrolysis of various marine fish sources. To develop bioactive peptides, marine fish products can be hydrolyzed using proteolytic enzymes obtained from plants, animals, and microorganisms [270]. Lineweaver–Burk plots have been used to kinetically assess the competitiveness of several antihypertensive peptides against ACE activity [271]. Antihypertensive peptides typically have a different mechanism of action than synthesized medicines. ACE inhibitory peptides interact very differently from synthesized medicines because they actively compete with ACE instead of generally blocking it. Angiotensin I is converted into angiotensin II by ACE by cleaving off a brief peptide. Instead of fighting angiotensin I, ACE interacts with antihypertensive peptides. Antihypertensive peptides work by suppressing the production of angiotensin II to relax artery walls and decrease fluid volume. Antihypertensive peptides hence boost blood and oxygen flow to the heart, liver, and kidneys while also improving cardiac function [272]. Because they block BP-regulating enzymes, peptides from marine fish have the potential to be used as natural antihypertensive medications. For safe and efficient integration, their role necessitates thorough scientific evaluation.
6.5 Seaweed polyphenols
Bioactive peptides and protein hydrolysates from seaweeds have been shown in numerous studies to have antihypertensive effects [273], as those from, among others, Undaria pinnatifida [274], Palmaria palmata [275], Porphyra columbina [276], and Porphyra yezoensis [277]. The antihypertensive impact associated with these algae may also be related to other bioactive substances found in seaweeds. This might apply to polyphenols. Similar to the antihypertensive action previously discovered for other phenolic derivatives from land plants, such as flavonoids (12), these polyphenolic chemicals' capacity to exert antioxidant effects can also be responsible for the indicated antihypertensive activity [278, 279]. In contrast, polyphenols have been reported to function as ACE inhibitors [251, 255]. Angiotensin II, a strong vasoconstrictor involved in the etiology of HTN, is produced when angiotensin I is converted to ACE, a zinc-containing protease. ACE also degrades the vasodilator bradykinin. This enzyme is essential for the regulation of BP. As a result, its inhibition has emerged as a primary focus for managing HTN. This explains the development of various ACE inhibitors, which are frequently regarded as one of the primary therapeutic approaches for CVDs in people [280]. Seaweed polyphenols could be showed potential in the treatment of HTN. Additional study is required to determine their therapeutic value and safety.
7 Conclusion and future direction
Traditional medicine refers to a broad range of centuries-old, culturally specific health care practices that were in use before the introduction of science to medical issues. In the end, throughout the past few decades, there has been a rise in the prevalence of HTN worldwide. The needs of the clinic are still obscure despite the use of numerous antihypertensive medications with various mechanisms of action [281]. The development of new lead compounds for the treatment of HTN has relied heavily on natural products and will continue to do so. To identify potential leads for structural alterations and optimization to produce future antihypertensive medications that are more effective and secure, numerous herbal treatments for HTN and plant extracts still need to be researched and identified [223]. The plants that are most frequently used to manage and treat HTN as well as their modes of action. Several factors, including endothelial function, ROS production, pro-inflammatory signaling, platelet activation, opening and closing of various ion channels, ACE inhibition, gene expression, and other pharmacological activities of natural plants and their isolates, influence the pathogenesis of HTN. According to the findings of relevant clinical and experimental research, herbal medicines will surely gather more attention in the future due to their wide spectrum of efficacy [7]. With a focus on the underlying molecular mechanisms of action within plants, herbs, phytochemicals, and marine components, this review has provided a thorough exploration of contemporary therapeutic approaches for treating HTN. Natural substances have significant potential as complementary therapies to established antihypertensive treatments, as they combine conventional knowledge with modern scientific findings. The complicated relationship of bioactive compounds from different sources indicates their ability to alter important pathways that control vascular function, OS, and BP. The evidence provided implies that these drugs have the potential for enhancing the available therapy options for HTN. Further research is required because of issues with standardization, bioavailability, and the transition from the bench to the bedside. Natural chemicals provide numerous benefits, including their potential synergistic effects, which can be utilized for future drug development and personalized therapy strategies. This research highlights the need for continuous multidisciplinary partnerships between traditional medicine practitioners, pharmacologists, and physicians to address the increasing global prevalence of HTN. We are in a position to begin a new era of comprehensive and efficient solutions for treating HTN and ultimately lowering the associated cardiovascular morbidity and mortality by utilizing the power of molecular pathways present in plants, and seafood components.
Availability of data and materials
No new data was generated for this review.
References
Staessen JA, Wang J, Bianchi G, Birkenhäger WH. Essential hypertension. The Lancet. 2003;361:1629–41.
Wu J, Peng W, Qin R, Zhou H. Crataegus pinnatifida: chemical constituents, pharmacology, and potential applications. Molecules. 2014;19:1685–712.
Carretero OA, Oparil S. Essential hypertension: part I: definition and etiology. Circulation. 2000;101:329–35.
Beevers G, Lip GY, O’Brien E. The pathophysiology of hypertension. BMJ. 2001;322:912–6.
Pierdomenico SD, Di Nicola M, Esposito AL, Di Mascio R, Ballone E, Lapenna D, Cuccurullo F. Prognostic value of different indices of blood pressure variability in hypertensive patients. Am J Hypertens. 2009;22:842–7.
Tabassum N, Ahmad F. Role of natural herbs in the treatment of hypertension. Pharmacogn Rev. 2011;5:30.
Verma T, Sinha M, Bansal N, Raj S, Kamal Y, Nagendra S, Chauhan S. Plants used as antihypertensive. Nat Prod Bioprospect. 2021;11(2):155–84.
Tirapelli CR, Ambrosio SR, de Oliveira AM, Tostes RC. Hypotensive action of naturally occurring diterpenes: a therapeutic promise for the treatment of hypertension. Fitoterapia. 2010;81:690–702.
Cragg GM, Newman DJ. Natural products: a continuing source of novel drug leads. Biochem Biophys Acta. 2013;1830:3670–95.
Newman DJ, Cragg GM. Natural products as sources of new drugs over the 30 years from 1981 to 2010. J Nat Prod. 2012;75:311–35.
Nakamura K, Naramoto K, Koyama M. Blood-pressure-lowering effect of fermented buckwheat sprouts in spontaneously hypertensive rats. J Funct Foods. 2013;5:406–15.
Jing P, Qian B, He Y, Zhao X, Zhang J, Zhao D, Lv Y, Deng Y. Screening milk-derived antihypertensive peptides using quantitative structure activity relationship (QSAR) modelling and in vitro/in vivo studies on their bioactivity. Int Dairy J. 2014;35:95–101.
Bai R, Wei Z, Liu J, Xie W, Yao H, Wu X, Jiang J, Wang Q, Xu J. Synthesis and biological evaluation of 4′-[(benzimidazole-1-yl) methyl] biphenyl-2-sulfonamide derivatives as dual angiotensin II/endothelin A receptor antagonists. Bioorg Med Chem. 2012;20:4661–7.
Messerli FH, Williams B, Ritz E. Essential hypertension. Lancet. 2007;370:591–603.
Kyrou I, Chrousos GP, Tsigos C. Stress, visceral obesity, and metabolic complications. Ann N Y Acad Sci. 2006;1083:77–110.
Wofford MR, Hall JE. Pathophysiology and treatment of obesity hypertension. Curr Pharm Des. 2004;10:3621–37.
Haslam DW. Obesity: gaining recognition in primary care. Trends Urol Gynaecol Sexual Health. 2007;12:12–4.
Lackland DT, Egan BM. Dietary salt restriction and blood pressure in clinical trials. Curr Hypertens Rep. 2007;9:314–9.
Djoussé L, Mukamal KJ. Alcohol consumption and risk of hypertension: does the type of beverage or drinking pattern matter? Rev Esp Cardiol. 2009;62:603–5.
Lee JH, O’Keefe JH, Bell D, Hensrud DD, Holick MF. Vitamin D deficiency: an important, common, and easily treatable cardiovascular risk factor? J Am Coll Cardiol. 2008;52:1949–56.
Stewart A, Wong K, Cachat J, Elegante M, Gilder T, Mohnot S, Wu N, Minasyan A, Tuohimaa P, Kalueff AV. Neurosteroid vitamin D system as a nontraditional drug target in neuropsychopharmacology. Behav Pharmacol. 2010;21:420–6.
Dickson ME, Sigmund CD. Genetic basis of hypertension: revisiting angiotensinogen. Hypertension. 2006;48:14–20.
Luma GB, Spiotta RT. Hypertension in children and adolescents. Am Fam Physician. 2006;73:1558–66.
Segura J, Ruilope LM. Obesity, essential hypertension and renin–angiotensin system. Public Health Nutr. 2007;10:1151–5.
Sorof J, Daniels S. Obesity hypertension in children: a problem of epidemic proportions. Hypertension. 2002;40:441–7.
Hwang IS, Ho H, Hoffman BB, Reaven GM. Fructose-induced insulin resistance and hypertension in rats. Hypertension. 1987;10:512–6.
Gupta-Malhotra M, Banker A, Shete S, Hashmi SS, Tyson JE, Barratt MS, Hecht JT, Milewicz DM, Boerwinkle E. Essential hypertension vs. secondary hypertension among children. Am J Hypertension. 2015;28:73–80.
Onusko EM. Diagnosing secondary hypertension. Am Fam Physician. 2003;67:67–74.
Dodt C, Wellhöner J, Schütt M, Sayk F. Glucocorticoids and hypertension. Der Internist. 2009;50:36–41.
Chiong JR, Aronow WS, Khan IA, Nair CK, Vijayaraghavan K, Dart RA, Behrenbeck TR, Geraci SA. Secondary hypertension: current diagnosis and treatment. Int J Cardiol. 2008;124:6–21.
Pimenta E, Oparil S. Role of aliskiren in cardio-renal protection and use in hypertensives with multiple risk factors. Vasc Health Risk Manag. 2009;5:453–63.
Takahashi H. Sympathetic hyperactivity in hypertension. Nihon Rinsho. 2008;66:1495–502.
Sagnella G, Swift P. The renal epithelial sodium channel: genetic heterogeneity and implications for the treatment of high blood pressure. Curr Pharm Des. 2006;12:2221–34.
Nelson MT, Quayle JM. Physiological roles and properties of potassium channels in arterial smooth muscle. Am J Physiol Cell Physiol. 1995;268:C799–822.
Mayet J, Hughes A. Cardiac and vascular pathophysiology in hypertension. Heart. 2003;89:1104–9.
Castro C, Lorenzo AG, González A, Cruzado M. Garlic components inhibit angiotensin II-induced cell-cycle progression and migration: Involvement of cell-cycle inhibitor p27Kip1 and mitogen-activated protein kinase. Mol Nutr Food Res. 2010;54:781–7.
Sendl A, Elbl G, Steinke B, Redl K, Breu W, Wagner H. Comparative pharmacological investigations of Allium ursinum and Allium sativum. Planta Med. 1992;58:1–7.
Banerjee S, Maulik M, Mancahanda S, Dinda A, Gupta S, Maulik S. Dose-dependent induction of endogenous antioxidants in rat heart by chronic administration of garlic. Life Sci. 2002;70:1509–18.
Vazquez-Prieto MA, Rodriguez Lanzi C, Lembo C, Galmarini CR, Miatello RM. Garlic and onion attenuates vascular inflammation and oxidative stress in fructose-fed rats. J Nutr Metab. 2011;2011: 475216.
Morihara N, Hayama M, Fujii H. Aged garlic extract scavenges superoxide radicals. Plant Foods Hum Nutr. 2011;66:17–21.
Padiya R, Chowdhury D, Borkar R, Srinivas R, Pal Bhadra M, Banerjee SK. Garlic attenuates cardiac oxidative stress via activation of PI3K/AKT/Nrf2-Keap1 pathway in fructose-fed diabetic rat. PLoS ONE. 2014;9: e94228.
Popović M, Kaurinović B, Trivić S, Mimica-Dukić N, Bursać M. Effect of celery (Apium graveolens) extracts on some biochemical parameters of oxidative stress in mice treated with carbon tetrachloride. Phytother Res. 2006;20:531–7.
Thiyagarajan R, Boobalan R. Antihypertensive, antihyperlipidemic and antioxidant influence of D-carvone in L-NAME induced hypertensive rats. Intern J Pharm Biol Arch. 2014;5:82–8.
Duffy SJ, Keaney JF Jr, Holbrook M, Gokce N, Swerdloff PL, Frei B, Vita JA. Short-and long-term black tea consumption reverses endothelial dysfunction in patients with coronary artery disease. Circulation. 2001;104:151–6.
Hodgson JM, Puddey IB, Burke V, Watts GF, Beilin LJ. Regular ingestion of black tea improves brachial artery vasodilator function. Clin Sci. 2002;102:195–201.
Nakagawa T, Yokozawa T. Direct scavenging of nitric oxide and superoxide by green tea. Food Chem Toxicol. 2002;40:1745–50.
Ludwig A, Lorenz M, Grimbo N, Steinle F, Meiners S, Bartsch C, Stangl K, Baumann G, Stangl V. The tea flavonoid epigallocatechin-3-gallate reduces cytokine-induced VCAM-1 expression and monocyte adhesion to endothelial cells. Biochem Biophys Res Commun. 2004;316:659–65.
Mineharu Y, Koizumi A, Wada Y, Iso H, Watanabe Y, Date C, Yamamoto A, Kikuchi S, Inaba Y, Toyoshima H. Coffee, green tea, black tea and oolong tea consumption and risk of mortality from cardiovascular disease in Japanese men and women. J Epidemiol Community Health. 2011;65:230–40.
Ribaldo PD, Souza DS, Biswas SK, Block K, Lopes de Faria JM, Lopes de Faria JB. Green tea (Camellia sinensis) attenuates nephropathy by downregulating Nox4 NADPH oxidase in diabetic spontaneously hypertensive rats. J Nutr. 2009;139:96–100.
Stepien M, Kujawska-Luczak M, Szulinska M, Kregielska-Narozna M, Skrypnik D, Suliburska J, Skrypnik K, Regula J, Bogdanski P. Beneficial dose-independent influence of Camellia sinensis supplementation on lipid profile, glycemia, and insulin resistance in an NaCl-induced hypertensive rat model. J Physiol Pharmacol. 2018;69:275–82.
Faria AM, Papadimitriou A, Silva KC, Lopes de Faria JM, Lopes de Faria JB. Uncoupling endothelial nitric oxide synthase is ameliorated by green tea in experimental diabetes by re-establishing tetrahydrobiopterin levels. Diabetes. 2012;61:1838–47.
Newsome BJ, Petriello MC, Han SG, Murphy MO, Eske KE, Sunkara M, Morris AJ, Hennig B. Green tea diet decreases PCB 126-induced oxidative stress in mice by up-regulating antioxidant enzymes. J Nutr Biochem. 2014;25:126–35.
Sulastri T, Sunyoto M, Suwitono MR, Levita J. The effect of red ginger bread consumption on the physiological parameters of healthy subjects. Jo Adv Pharm Educ Res. 2022;12:29.
Wang Y, Huang Y, Lam KS, Li Y, Wong WT, Ye H, Lau C-W, Vanhoutte PM, Xu A. Berberine prevents hyperglycemia-induced endothelial injury and enhances vasodilatation via adenosine monophosphate-activated protein kinase and endothelial nitric oxide synthase. Cardiovasc Res. 2009;82:484–92.
Zhang P, Song S-J, Liu W-L, Li L-L, Zhao W-L, Zhang Y. Eeffects of Coptis Chinensis on vasoconstrictive activity of isolated thoracic aorta of normoxic and chronic intermittent hypobaric hypoxic rats. Chin J Appl Physiol. 2011;27:420–5.
Gupta P, Bansal MP, Koul A. Spectroscopic characterization of lycopene extract from Lycopersicum esculentum (Tomato) and its evaluation as a chemopreventive agent against experimental hepatocarcinogenesis in mice. Phytother Res. 2013;27:448–56.
Sreelatha S, Padma P, Umadevi M. Protective effects of Coriandrum sativum extracts on carbon tetrachloride-induced hepatotoxicity in rats. Food Chem Toxicol. 2009;47:702–8.
Wu TT, Tsai CW, Yao HT, Lii CK, Chen HW, Wu YL, Chen PY, Liu KL. Suppressive effects of extracts from the aerial part of Coriandrum sativum L. on LPS-induced inflammatory responses in murine RAW 264.7 macrophages. J Sci Food Agric. 2010;90:1846–54.
Koçyõldõz ZÇ, Birman H, Olgaç V, Akgün-Dar K, Melikoğlu G, Meriçli A. Crataegus tanacetifolia leaf extract prevents L-NAME-induced hypertension in rats: a morphological study. Phytothe Res. 2006;20:66–70.
Cheng N, Wang Y, Gao H, Yuan J, Feng F, Cao W, Zheng J. Protective effect of extract of Crataegus pinnatifida pollen on DNA damage response to oxidative stress. Food Chem Toxicol. 2013;59:709–14.
Topal G, Koç E, Karaca Ç, Altuğ T, Ergin B, Demirci C, Melikoğlu G, Meriçli AH, Kucur M, Özdemir O. Effects of crataegus microphylla on vascular dysfunction in streptozotocin-induced diabetic rats. Phytother Res. 2013;27:330–7.
Premkumar K, Abraham SK, Santhiya S, Ramesh A. Protective effects of saffron (Crocus sativus Linn.) on genotoxins-induced oxidative stress in Swiss albino mice. Phytother Res. 2003;17:614–7.
Dzeufiet PDD, Mogueo A, Bilanda DC, Aboubakar B-FO, Tédong L, Dimo T, Kamtchouing P. Antihypertensive potential of the aqueous extract which combine leaf of Persea americana Mill. (Lauraceae), stems and leaf of Cymbopogon citratus (DC) Stapf. (Poaceae), fruits of Citrus medical L. (Rutaceae) as well as honey in ethanol and sucrose experimental model. BMC Complement Altern Med. 2014;14:1–12.
El-Beshbishy HA, Hassan MH, Aly HA, Doghish AS, Alghaithy AA. Crocin “saffron” protects against beryllium chloride toxicity in rats through diminution of oxidative stress and enhancing gene expression of antioxidant enzymes. Ecotoxicol Environ Saf. 2012;83:47–54.
Bharti S, Golechha M, Kumari S, Siddiqui KM, Arya DS. Akt/GSK-3β/eNOS phosphorylation arbitrates safranal-induced myocardial protection against ischemia–reperfusion injury in rats. Eur J Nutr. 2012;51:719–27.
Doh KC, Lim SW, Piao SG, Jin L, Heo SB, Zheng YF, Bae SK, Hwang GH, Min KI, Chung BH. Ginseng treatment attenuates chronic cyclosporine nephropathy via reducing oxidative stress in an experimental mouse model. Am J Nephrol. 2013;37:421–33.
Wang Y, Liu Y, Zhang X-Y, Xu L-H, Ouyang D-Y, Liu K-P, Pan H, He J, He X-H. Ginsenoside Rg1 regulates innate immune responses in macrophages through differentially modulating the NF-κB and PI3K/Akt/mTOR pathways. Int Immunopharmacol. 2014;23:77–84.
Qian Q, Qian S, Fan P, Huo D, Wang S. Effect of Salvia miltiorrhiza hydrophilic extract on antioxidant enzymes in diabetic patients with chronic heart disease: a randomized controlled trial. Phytother Res. 2012;26:60–6.
Hedhli N, Kalinowski A, Russell SK. Cardiovascular effects of neuregulin-1/ErbB signaling: role in vascular signaling and angiogenesis. Curr Pharm Design. 2014;20:4899–905.
Xagorari A, Papapetropoulos A, Mauromatis A, Economou M, Fotsis T, Roussos C. Luteolin inhibits an endotoxin-stimulated phosphorylation cascade and proinflammatory cytokine production in macrophages. J Pharmacol Exp Ther. 2001;296:181–7.
Ahmad A, Husain A, Mujeeb M, Khan SA, Najmi AK, Siddique NA, Damanhouri ZA, Simões DM, Malheiros J, Antunes PE, Figueirinha A, Cotrim MD, Fonseca DA. Vascular activity of infusion and fractions of Cymbopogon citratus (DC) Stapf. in human arteries. J Ethnopharmacol. 2020;258:112947.
Bastos JF, Moreira ÍJ, Ribeiro TP, Medeiros IA, Antoniolli AR, De Sousa DP, Santos MR. Hypotensive and vasorelaxant effects of citronellol, a monoterpene alcohol, in rats. Basic Clin Pharmacol Toxicol. 2010;106:331–7.
Liang Y-C, Yang M-T, Lin C-J, Chang CL-T, Yang W-C. Bidens pilosa and its active compound inhibit adipogenesis and lipid accumulation via down-modulation of the C/EBP and PPARγ pathways. Sci Rep. 2016;6:24285.
Chiang YM, Lo CP, Chen YP, Wang SY, Yang NS, Kuo YH, Shyur LF. Ethyl caffeate suppresses NF-κB activation and its downstream inflammatory mediators, iNOS, COX-2, and PGE2in vitro or in mouse skin. Br J Pharmacol. 2005;146:352–63.
Jayakumar T, Hsieh C, Lee JJ, Sheu JR. Experimental and clinical pharmacology of Andrographis paniculata and its major bioactive phytoconstituent andrographolide. Evid Based Complement Alrtern Med. 2013;2013:846740.
Zhang C, Tan B. Hypotensive activity of aqueous extract of Andrographis paniculata in rats. Clin Exp Pharmacol Physiol. 1996;23:675–8.
Das S, Periyasamy R, Pandey KN. Activation of IKK/NF-κB provokes renal inflammatory responses in guanylyl cyclase/natriuretic peptide receptor-A gene-knockout mice. Physiol Genomics. 2012;44:430–42.
Shin S-G, Kim JY, Chung HY, Jeong J-C. Zingerone as an antioxidant against peroxynitrite. J Agric Food Chem. 2005;53:7617–22.
Mao Q-Q, Xu X-Y, Cao S-Y, Gan R-Y, Corke H, Beta T, Li H-B. Bioactive compounds and bioactivities of ginger (Zingiber officinale Roscoe). Foods. 2019;8:185.
Ried K, Frank OR, Stocks NP. Aged garlic extract lowers blood pressure in patients with treated but uncontrolled hypertension: a randomised controlled trial. Maturitas. 2010;67:144–50.
Ried K, Frank O, Stocks N. Aged garlic extract reduces blood pressure in hypertensives: a dose–response trial. Eur J Clin Nutr. 2013;67:64–70.
Ashraf R, Khan RA, Ashraf I, Qureshi AA. Effects of Allium sativum (garlic) on systolic and diastolic blood pressure in patients with essential hypertension. Pak J Pharm Sci. 2013;26:859.
Hodgson JM, Puddey IB, Burke V, Beilin LJ, Jordan N. Effects on blood pressure of drinking green and black tea. J Hypertens. 1999;17:457–63.
Bogdanski P, Suliburska J, Szulinska M, Stepien M, Pupek-Musialik D, Jablecka A. Green tea extract reduces blood pressure, inflammatory biomarkers, and oxidative stress and improves parameters associated with insulin resistance in obese, hypertensive patients. Nutr Res. 2012;32:421–7.
Hodgson JM, Puddey IB, Woodman RJ, Mulder TP, Fuchs D, Scott K, Croft KD. Effects of black tea on blood pressure: a randomized controlled trial. Arch Intern Med. 2012;172:186–8.
Anselm E, Socorro VFM, Dal-Ros S, Schott C, Bronner C, Schini-Kerth VB. Crataegus special extract WS 1442 causes endothelium-dependent relaxation via a redox-sensitive Src-and Akt-dependent activation of endothelial NO synthase but not via activation of estrogen receptors. J Cardiovasc Pharmacol. 2009;53:253–60.
Imenshahidi M, Hosseinzadeh H, Javadpour Y. Hypotensive effect of aqueous saffron extract (Crocus sativus L.) and its constituents, safranal and crocin, in normotensive and hypertensive rats. Phytother Res. 2010;24:990–4.
Boskabady M, Shafei M, Shakiba A, Sefidi HS. Effect of aqueous-ethanol extract from Crocus sativus (saffron) on guinea-pig isolated heart. Phytother Res. 2008;22:330–4.
Dehkordi FR, Kamkhah AF. Antihypertensive effect of Nigella sativa seed extract in patients with mild hypertension. Fundam Clin Pharmacol. 2008;22:447–52.
Wang B-Q. Salvia miltiorrhiza: chemical and pharmacological review of a medicinal plant. J Med Plants Res. 2010;4:2813–20.
Woo K, Yip TW, Chook P, Kwong S, Szeto C, Li JK, Yu AW, Cheng WK, Chan TY, Fung K. Cardiovascular protective effects of adjunctive alternative medicine (Salvia miltiorrhiza and Pueraria lobata) in high-risk hypertension. Evid Based Complement Altern Med. 2013;2013:132912.
Jovanovski E, Bateman EA, Bhardwaj J, Fairgrieve C, Mucalo I, Jenkins AL, Vuksan V. Effect of Rg3-enriched Korean red ginseng (Panax ginseng) on arterial stiffness and blood pressure in healthy individuals: a randomized controlled trial. J Am Soc Hypertens. 2014;8:537–41.
Londhe V, Gavasane A, Nipate S, Bandawane D, Chaudhari P. Role of garlic (Allium sativum) in various diseases: an overview. Angiogenesis. 2011;12:129–34.
Ried K, Frank OR, Stocks NP, Fakler P, Sullivan T. Effect of garlic on blood pressure: a systematic review and meta-analysis. BMC Cardiovasc Disord. 2008;8:1–12.
Dhawan V, Jain S. Garlic supplementation prevents oxidative DNA damage in essential hypertension. Mol Cell Biochem. 2005;275:85–94.
Augusti K. Therapeutic values of onion (Allium cepa L.) and garlic (Allium sativum L.). Indian J Exp Biols. 1996;34:634–40.
Kooti W, Ali-Akbari S, Asadi-Samani M, Ghadery H, Ashtary-Larky D. A review on medicinal plant of Apium graveolens. Adv Herb Med. 2015;1:48–59.
Somanadhan B, Varughese G, Palpu P, Sreedharan R, Gudiksen L, Smitt UW, Nyman U. An ethnopharmacological survey for potential angiotensin converting enzyme inhibitors from Indian medicinal plants. J Ethnopharmacol. 1999;65:103–12.
Gharooni M, Sarkarati A. Application of Apium graveolens in treatment of hypertension. Tehran Univ Med J. 2000;58:67–9.
Simpson D. Buchu—South Africa’s amazing herbal remedy. Scott Med J. 1998;43:189–91.
Kavitha C, Rajamani K, Vadivel E. Coleus forskohlii: a comprehensive review on morphology, phytochemistry and pharmacological aspects. J Med Plants Res. 2010;4:278–85.
Dubey M, Srimal R, Nityanand S, Dhawan B. Pharmacological studies on coleonol, a hypotensive diterpene from Coleus forskohlii. J Ethnopharmacol. 1981;3:1–13.
Mashour NH, Lin GI, Frishman WH. Herbal medicine for the treatment of cardiovascular disease: clinical considerations. Arch Intern Med. 1998;158:2225–34.
Bensky D, Barolet R. Chinese herbal medicine: formulas and strategies. Seattle: Eastland Press; 1990.
Brixius K, Willms S, Napp A, Tossios P, Ladage D, Bloch W, Mehlhorn U, Schwinger RH. Crataegus special extract WS® 1442 induces an endothelium-dependent, NO-mediated vasorelaxation via eNOS-phosphorylation at serine 1177. Cardiovasc Drugs Ther. 2006;20:177–84.
Schüssler M, Hölzl J, Fricke U. Myocardial effects of flavonoids from Crataegus species. Arzneimittelforschung. 1995;45:842–5.
Fikselová M, Šilhár S, Mareček J, Frančáková H. Extraction of carrot (Daucus carota L.) carotenes under different conditions. Czech J Food Sci. 2008;26:268–74.
Gilani A, Shaheen F, Saeed S, Bibi S, Sadiq M, Faizi S. Hypotensive action of coumarin glycosides from Daucus carota. Phytomedicine. 2000;7:423–6.
Fu H-W, Zhang L, Yi T, Feng Y-L, Tian J-K. Two new guaiane-type sesquiterpenoids from the fruits of Daucus carota L. Fitoterapia. 2010;81:443–6.
Surbhi S, Verma R, Deepak R, Jain H, Yadav K. A review: Food, chemical composition and utilization of carrot (Daucus carota L.) pomace. Int J Chem Stud. 2018;6:2921–6.
Hansen K, Nyman U, Smitt UW, Adsersen A, Gudiksen L, Rajasekharan S, Pushpangadan P. In vitro screening of traditional medicines for anti-hypertensive effect based on inhibition of the angiotensin converting enzyme (ACE). J Ethnopharmacol. 1995;48:43–51.
Mi J, Duan J, Zhang J, Lu J, Wang H, Wang Z. Evaluation of antiurolithic effect and the possible mechanisms of Desmodium styracifolium and Pyrrosiae petiolosa in rats. Urol Res. 2012;40:151–61.
Pinela J, Barros L, Carvalho AM, Ferreira IC. Nutritional composition and antioxidant activity of four tomato (Lycopersicon esculentum L.) farmer’varieties in Northeastern Portugal homegardens. Food Chem Toxicol. 2012;50:829–34.
Engelhard YN, Gazer B, Paran E. Natural antioxidants from tomato extract reduce blood pressure in patients with grade-1 hypertension: a double-blind, placebo-controlled pilot study. Am Heart J. 2006;151:100.
Paran E, Novack V, Engelhard YN, Hazan-Halevy I. The effects of natural antioxidants from tomato extract in treated but uncontrolled hypertensive patients. Cardiovasc Drugs Ther. 2009;23:145–51.
Pereira BL, Arruda FC, Reis PP, Felix TF, Santos PP, Rafacho BP, Gonçalves AF, Claro RT, Azevedo PS, Polegato BF. Tomato (Lycopersicon esculentum) supplementation induces changes in cardiac miRNA expression, reduces oxidative stress and left ventricular mass, and improves diastolic function. Nutrients. 2015;7:9640–9.
Iravani S, Zolfaghari B. Pharmaceutical and nutraceutical effects of Pinus pinaster bark extract. Res Pharm Sci. 2011;6:1.
Liu X, Wei J, Tan F, Zhou S, Würthwein G, Rohdewald P. Pycnogenol®, French maritime pine bark extract, improves endothelial function of hypertensive patients. Life Sci. 2004;74:855–62.
Hosseini S, Lee J, Sepulveda RT, Rohdewald P, Watson RR. A randomized, double-blind, placebo-controlled, prospective, 16 week crossover study to determine the role of Pycnogenol in modifying blood pressure in mildly hypertensive patients. Nutr Res. 2001;21:1251–60.
Shaygannia E, Bahmani M, Zamanzad B, Rafieian-Kopaei M. A review study on Punica granatum L. J Evid Based Complement Altern Med. 2016;21:221–7.
Chalé FGH, Ruiz JCR, Fernández JJA, Ancona DAB, Campos MRS. ACE inhibitory, hypotensive and antioxidant peptide fractions from Mucuna pruriens proteins. Process Biochem. 2014;49:1691–8.
Aviram M, Dornfeld L. Pomegranate juice consumption inhibits serum angiotensin converting enzyme activity and reduces systolic blood pressure. Atherosclerosis. 2001;158:195–8.
Banihani SA. Radish (Raphanus sativus) and diabetes. Nutrients. 2017;9:1014.
Ghayur MN, Gilani AH. Radish seed extract mediates its cardiovascular inhibitory effects via muscarinic receptor activation. Fundam Clin Pharmacol. 2006;20:57–63.
Gutiérrez RMP, Perez RL. Raphanus sativus (Radish): their chemistry and biology. Sci World J. 2004;4:811.
Ajagbonna O, Adegunloye B, Sofola O. Relaxant effects of isolated rat aorta by Rhaptopetalum coriaceum bark extract. J Herbs Spices Med Plants. 1998;6:37–42.
Ajagbonna O, Oneyeyili P. Effects of ethanol extract of Rhaptopetalum coriaceum Oliv. stem bark on mean arterial pressure and heart rate in rats. Nig J Exp Clin Anal. 2002;2:30–3.
Nakano D, Itoh C, Takaoka M, Kiso Y, Tanaka T, Matsumura Y. Antihypertensive effect of sesamin. IV. Inhibition of vascular superoxide production by sesamin. Biol Pharm Bull. 2002;25:1247–9.
Dias MC, Conceição IL, Abrantes I, Cunha MJ. Solanum sisymbriifolium-a new approach for the management of plant-parasitic nematodes. Eur J Plant Pathol. 2012;133:171–9.
Ibarrola DA, Ibarrola M, Vera C, Montalbetti Y, Ferro E. Hypotensive effect of crude root extract of Solanum sisymbriifolium (Solanaceae) in normo-and hypertensive rats. J Ethnopharmacol. 1996;54:7–12.
Hellión-lbarrola M, Montalbetti Y, Heinichen O, Alvarenga N, Figueredo A, Ferro E. Isolation of hypotensive compounds from Solanum sisymbriifolium Lam. J Ethnopharmacol. 2000;70:301–7.
Bletsos F, Roupakias D, Tsaktsira M, Scaltsoyjannes A, Thanassoulopoulos C. Interspecific hybrids between three eggplant (Solanum melongena L.) cultivars and two wild species (Solanum torvum Sw. and Solanum sisymbriifolium Lam). Plant Breeding. 1998;117:159–64.
de Souza PA, Moreira LF, Sarmento DH, da Costa FB. Cacao—Theobroma cacao. In Exotic fruits; Elsevier: 2018; pp. 69-76
Taubert D, Berkels R, Roesen R, Klaus W. Chocolate and blood pressure in elderly individuals with isolated systolic hypertension. JAMA. 2003;290:1029–30.
Rusconi M, Conti A. Theobroma cacao L., the food of the gods: a scientific approach beyond myths and claims. Pharmacol Res. 2010;61:5–13.
Ndagijimana A, Wang X, Pan G, Zhang F, Feng H, Olaleye O. A review on indole alkaloids isolated from Uncaria rhynchophylla and their pharmacological studies. Fitoterapia. 2013;86:35–47.
Amaechina FC, Omogbai EK. Hypotensive effect of aqueous extract of the leaves of Phyllanthus amarus Schum and Thonn (Euphorbiaceae). Acta Pol Pharm. 2007;64:547–52.
Horie S, Koyama F, Takayama H, Ishikawa H, Aimi N, Ponglux D, Matsumoto K, Murayama T. Indole alkaloids of a Thai medicinal herb, Mitragyna speciosa, that has opioid agonistic effect in guinea-pig ileum. Planta Med. 2005;71:231–6.
Endo K, Oshima Y, Kikuchi H, Koshihara Y, Hikino H. Hypotensive principles of Uncaria hooks. Planta Med. 1983;49:188–90.
Yang W, Ip S-P, Liu L, Xian Y-F, Lin Z-X. Uncaria rhynchophylla and its major constituents on central nervous system: a review on their pharmacological actions. Curr Vasc Pharmacol. 2020;18:346–57.
Zuber D. Biological flora of central Europe: Viscum album L. Flora-Morphol Distribut Funct Ecol Plants. 2004;199:181–203.
Tenorio F, Del Valle L, González A, Pastelín G. Vasodilator activity of the aqueous extract of Viscum album. Fitoterapia. 2005;76:204–9.
Ben E, Eno A, Ofem O, Aidem U, Itam E. Increased plasma total cholesterol and high density lipoprotein levels produced by the crude extract from the leaves of Viscum album (mistletoe). Nigerian J Physiol Sci. 2006;21:55–60.
Poruthukaren KJ, Palatty PL, Baliga MS, Suresh S. Clinical evaluation of Viscum album mother tincture as an antihypertensive: a pilot study. J Evid Based Complement Altern Med. 2014;19:31–5.
Dhanik J, Arya N, Nand V. A review on Zingiber officinale. J Pharmacogn Phytochem. 2017;6:174–84.
Fugh-Berman A. Herbs and dietary supplements in the prevention and treatment of cardiovascular disease. Prev Cardiol. 2000;3:24–32.
Ghayur MN, Gilani AH. Ginger lowers blood pressure through blockade of voltage-dependent calcium channels. J Cardiovasc Pharmacol. 2005;45:74–80.
Nicoll R, Henein MY. Ginger (Zingiber officinale Roscoe): a hot remedy for cardiovascular disease? Int J Cardiol. 2009;131:408–9.
Mishra SK, Sangwan NS, Sangwan RS. Phcog rev: plant review Andrographis paniculata (Kalmegh): a review. Pharmacogn Rev. 2007;1:283–98.
Kunwar RM, Shrestha KP, Bussmann RW. Traditional herbal medicine in Far-west Nepal: a pharmacological appraisal. J Ethnobiol Ethnomed. 2010;6:1–18.
Karaki H, Nakagawa H, Urakawa N. Comparative effects of verapamil and sodium nitroprusside on contraction and 45Ca uptake in the smooth muscle of rabbit aorta, rat aorta and guinea-pig taenia coli. Br J Pharmacol. 1984;81:393–400.
Awang K, Abdullah NH, Hadi AHA, Su Fong Y. Cardiovascular activity of labdane diterpenes from Andrographis paniculata in isolated rat hearts. J Biomed Biotechnol. 2012;2012:876458.
Thisoda P, Rangkadilok N, Pholphana N, Worasuttayangkurn L, Ruchirawat S, Satayavivad J. Inhibitory effect of Andrographis paniculata extract and its active diterpenoids on platelet aggregation. Eur J Pharmacol. 2006;553:39–45.
Nyeem MAB, Mannan MA, Nuruzzaman M, Kamrujjaman K, Das SK. Indigenous king of bitter (Andrographis paniculata): a review. J Med Plants Stud. 2017;5:318–24.
Al Disi SS, Anwar MA, Eid AH. Anti-hypertensive herbs and their mechanisms of action: part I. Front Pharmacol. 2016;6:323.
Pereira A, Fraga-Corral M, García-Oliveira P, Jimenez-Lopez C, Lourenço-Lopes C, Carpena M, Otero P, Gullón P, Prieto M, Simal-Gandara J. Culinary and nutritional value of edible wild plants from northern Spain rich in phenolic compounds with potential health benefits. Food Funct. 2020;11:8493–515.
Tabassum N, Ahmad F. Department of Pharmaceutical Sciences, University of Kashmire, Hazratbal, Srinager, J&K-190 006, India. Role of natural herbs in the treatment of hypertension. Pharmacogn Rev. 2011;5:30–40.
Lima Silva F, Fischer DCH, Fechine Tavares J, Sobral Silva M, Filgueiras de Athayde-Filho P, Barbosa-Filho JM. Compilation of secondary metabolites from Bidens pilosa L. Molecules. 2011;16:1070–102.
Bartolome A, Villaseñor I, Yang WC. Bidens pilosa L. (Asteraceae): botanical properties, traditional uses, phytochemistry and pharmacology. Evid Based Complement Altern Med. 2013;2013:340215.
Kviecinski MR, Felipe KB, Correia JG, Ferreira EA, Rossi MH, de Moura Gatti F, Pedrosa RC. Brazilian Bidens pilosa Linné yields fraction containing quercetin-derived flavonoid with free radical scavenger activity and hepatoprotective effects. Libyan J Med. 2011;6:5651.
Bilanda D, Dzeufiet P, Kouakep L, Aboubakar BF, Tedong L, Kamtchouing P, Dimo T. Bidens Pilosa.
Dimo T, Rakotonirina SV, Tan PV, Azay J, Dongo E, Cros G. Leaf methanol extract of Bidens pilosa prevents and attenuates the hypertension induced by high-fructose diet in Wistar rats. J Ethnopharmacol. 2002;83:183–91.
Dimo T, Nguelefack T, Tan P, Yewah M, Dongo E, Rakotonirina S, Kamanyi A, Bopelet M. Possible mechanisms of action of the neutral extract from Bidens pilosa L. leaves on the cardiovascular system of anaesthetized rats. Phytother Res. 2003;17:1135–9.
Yang H-L, Chen S-C, Chang N-W, Chang J-M, Lee M-L, Tsai P-C, Fu H-H, Kao W-W, Chiang H-C, Wang H-H. Protection from oxidative damage using Bidens pilosa extracts in normal human erythrocytes. Food Chem Toxicol. 2006;44:1513–21.
Parveen G, Ali M. Extraction isolation and phytochemical screening of leaves and stems of Bidens pilosa and evaluation of antifungal potential of extracts. J Pharm Biol Sci. 2019;14:73–85.
Dimo T, Azay J, Tan PV, Pellecuer J, Cros G, Bopelet M, Serrano JJ. Effects of the aqueous and methylene chloride extracts of Bidens pilosa leaf on fructose-hypertensive rats. J Ethnopharmacol. 2001;76:215–21.
Nguelefack T, Dimo T, Mbuyo EN, Tan P, Rakotonirina S, Kamanyi A. Relaxant effects of the neutral extract of the leaves of Bidens pilosa Linn on isolated rat vascular smooth muscle. Phytother Res. 2005;19:207–10.
Namita P, Mukesh R, Vijay KJ. Camellia sinensis (green tea): a review. Glob J Pharmacol. 2012;6:52–9.
Deka A, Vita JA. Tea and cardiovascular disease. Pharmacol Res. 2011;64:136–45.
Oyama J-I, Maeda T, Kouzuma K, Ochiai R, Tokimitsu I, Higuchi Y, Sugano M, Makino N. Green tea catechins improve human forearm endothelial dysfunction and have antiatherosclerotic effects in smokers. Circ J. 2010;74:578–88.
Hong MH, Kim MH, Chang HJ, Kim NH, Shin BA, Ahn BW, Jung YD. (−)-Epigallocatechin-3-gallate inhibits monocyte chemotactic protein-1 expression in endothelial cells via blocking NF-κB signaling. Life Sci. 2007;80:1957–65.
Peng X, Zhou R, Wang B, Yu X, Yang X, Liu K, Mi M. Effect of green tea consumption on blood pressure: a meta-analysis of 13 randomized controlled trials. Sci Rep. 2014;4:6251.
Zhang L, Ho CT, Zhou J, Santos JS, Armstrong L, Granato D. Chemistry and biological activities of processed Camellia sinensis teas: a comprehensive review. Compr Rev Food Sci Food Saf. 2019;18:1474–95.
San Cheang W, Yuen Ngai C, Yen Tam Y, Yu Tian X, Tak Wong W, Zhang Y, Wai Lau C, Chen ZY, Bian Z-X, Huang Y. Black tea protects against hypertension-associated endothelial dysfunction through alleviation of endoplasmic reticulum stress. Sci Rep. 2015;5:10340.
Bagade A, Tumbigeremutt V, Pallavi G. Cardiovascular effects of berberine: a review of the literature. J Restorat Med. 2017;6:37.
Basati G, Mehrabi Rad F, Momeni Safarabadi A, Shakib P, Sedighi M, Cheraghi M. An overview of the most important medicinal plants that affect hypertension and their antihypertensive mechanism. J Med Plants and By-product. 2021;10:1–8.
Tassell MC, Kingston R, Gilroy D, Lehane M, Furey A. Hawthorn (Crataegus spp.) in the treatment of cardiovascular disease. Pharmacogn Rev. 2010;4:32.
Walker AF, Marakis G, Morris AP, Robinson PA. Promising hypotensive effect of hawthorn extract: a randomized double-blind pilot study of mild, essential hypertension. Phytother Res. 2002;16:48–54.
Bone K, Mills S. Principles and practice of phytotherapy, modern herbal medicine. Elsevier Health Sciences. Churchill Livingstone, Edinburgh. 2013.
Cui X, Liu X, Feng H, Zhao S, Gao H. Grape seed proanthocyanidin extracts enhance endothelial nitric oxide synthase expression through 5′-AMP activated protein kinase/Surtuin 1–Krüpple like factor 2 pathway and modulate blood pressure in ouabain induced hypertensive rats. Biol Pharm Bull. 2012;35:2192–7.
Negi P, Singh R, Dwivedi S. Evaluation of antihypertensive effect of fruit beverage of Crataegus crenulata Roxb.: a wild shrub of Himalayan hills. Defence Life Sci J. 2018;3:146–50.
Ghaffari S, Roshanravan N. Saffron; an updated review on biological properties with special focus on cardiovascular effects. Biomed Pharmacother. 2019;109:21–7.
Ekpenyong CE, Akpan EE, Daniel NE. Phytochemical constituents, therapeutic applications and toxicological profile of Cymbopogon citratus Stapf (DC) leaf extract. J Pharmacogn Phytochem. 2014;3:133–41.
Devi RC, Sim SM, Ismail R. Effect of Cymbopogon citratus and citral on vascular smooth muscle of the isolated thoracic rat aorta. Evid Based Complement Altern Med. 2012;2012:539475.
Nambiar VS, Matela H. Potential functions of lemon grass (Cymbopogon citratus) in health and disease. Int J Pharm Biol Arch. 2012;3:1035–43.
Carbajal D, Casaco A, Arruzazabala L, Gonzalez R, Tolon Z. Pharmacological study of Cymbopogon citratus leaves. J Ethnopharmacol. 1989;25:103–7.
Ali B, Blunden G. Pharmacological and toxicological properties of Nigella sativa. Phytother Res. 2003;17:299–305.
Amin B, Hosseinzadeh H. Black cumin (Nigella sativa) and its active constituent, thymoquinone: an overview on the analgesic and anti-inflammatory effects. Planta Med. 2015;82:8–16.
Khater HF, Ziam H, Abbas A, Abbas RZ, Raza MA, Hussain K, Younis E, Radwan I, Selim A. Avian coccidiosis: recent advances in alternative control strategies and vaccine development. Agrobiol Rec. 2020;1:11–25.
Jaarin K, Foong WD, Yeoh MH, Kamarul ZYN, Qodriyah HMS, Azman A, Zuhair JSF, Juliana AH, Kamisah Y. Mechanisms of the antihypertensive effects of Nigella sativa oil in L-NAME-induced hypertensive rats. Clinics. 2015;70:751–7.
Hussain DA, Hussain MM. Nigella sativa (black seed) is an effective herbal remedy for every disease except death-a Prophetic statement which modern scientists confirm unanimously: a review. Adv Med Plant Res. 2016;4:27–57.
Musharraf HM, Arman MSI. Prophetic medicine is the cheapest, safest and the best remedy in the prevention and treatment of hypertension (high blood pressure)–a mini review. Int J Mol Biol. 2018;3:245–50.
Fallah Huseini H, Amini M, Mohtashami R, Ghamarchehre M, Sadeqhi Z, Kianbakht S, Fallah Huseini A. Blood pressure lowering effect of Nigella sativa L. seed oil in healthy volunteers: a randomized, double-blind, placebo-controlled clinical trial. Phytother Res. 2013;27:1849–53.
Kundu JK, Liu L, Shin J-W, Surh Y-J. Thymoquinone inhibits phorbol ester-induced activation of NF-κB and expression of COX-2, and induces expression of cytoprotective enzymes in mouse skin in vivo. Biochem Biophys Res Commun. 2013;438:721–7.
Coon JT, Ernst E. Panax ginseng. Drug Saf. 2002;25:323–44.
Kim J-H. Pharmacological and medical applications of Panax ginseng and ginsenosides: a review for use in cardiovascular diseases. J Ginseng Res. 2018;42:264–9.
Kim JD, C. Cardiovascular diseases and Panax ginseng: a review on molecular mechanisms and medical applications. J Ginseng Res. 2012;36:16–26.
Choi KT. Botanical characteristics, pharmacological effects and medicinal components of Korean Panax ginseng CA Meyer. Acta Pharmacol Sin. 2008;29:1109–18.
Mucalo I, Jovanovski E, Rahelić D, Božikov V, Romić Ž, Vuksan V. Effect of American ginseng (Panax quinquefolius L.) on arterial stiffness in subjects with type-2 diabetes and concomitant hypertension. J Ethnopharmacol. 2013;150:148–53.
Lee KH, Bae IY, Park SI, Park J-D, Lee HG. Antihypertensive effect of Korean Red Ginseng by enrichment of ginsenoside Rg3 and arginine–fructose. J Ginseng Res. 2016;40:237–44.
Kim E-Y, Jho H-K, Kim D-I, Rhyu MR. Cirsium japonicum elicits endothelium-dependent relaxation via histamine H1-receptor in rat thoracic aorta. J Ethnopharmacol. 2008;116:223–7.
Wang S-P, Zang W-J, Kong S-S, Yu X-J, Sun L, Zhao X-F, Wang S-X, Zheng X-H. Vasorelaxant effect of isopropyl 3-(3, 4-dihydroxyphenyl)-2-hydroxypropanoate, a novel metabolite from Salvia miltiorrhiza, on isolated rat mesenteric artery. Eur J Pharmacol. 2008;579:283–8.
Yang L, Zou X, Liang Q, Chen H, Feng J, Yan L, Wang Z, Zhou D, Li S, Yao S. Sodium tanshinone IIA sulfonate depresses angiotensin II-induced cardiomyocyte hypertrophy through MEK/ERK pathway. Exp Mol Med. 2007;39:65–73.
Kwan YW, To KW, Lau WM, Tsang SH. Comparison of the vascular relaxant effects of ATP-dependent K+ channel openers on aorta and pulmonary artery isolated from spontaneously hypertensive and Wistar-Kyoto rats. Eur J Pharmacol. 1999;365:241–51.
Yang TY, Wei JCC, Lee MY, Chen CB, Ueng KC. A randomized, double-blind, placebo-controlled study to evaluate the efficacy and tolerability of Fufang Danshen (Salvia miltiorrhiza) as add-on antihypertensive therapy in Taiwanese patients with uncontrolled hypertension. Phytother Res. 2012;26:291–8.
Zhang J, An SJ, Fu JQ, Liu P, Shao TM, Li M, Li X, Jiao Z, Chai XQ. Mixed aqueous extract of Salvia miltiorrhiza reduces blood pressure through inhibition of vascular remodelling and oxidative stress in spontaneously hypertensive rats. Cell Physiol Biochem. 2016;40:347–60.
Lin T-H, Hsieh C-L. Pharmacological effects of Salvia miltiorrhiza (Danshen) on cerebral infarction. Chin Med. 2010;5:1–6.
Wang L, Ma R, Liu C, Liu H, Zhu R, Guo S, Tang M, Li Y, Niu J, Fu M. Salvia miltiorrhiza: a potential red light to the development of cardiovascular diseases. Curr Pharm Des. 2017;23:1077–97.
Hou G, Jiang Y, Zheng Y, Zhao M, Chen Y, Ren Y, Wang C, Li W. Mechanism of radix astragali and radix salviae miltiorrhizae ameliorates hypertensive renal damage. BioMed Res Int. 2021;2021:5598351.
Vakil RJ. Rauwolfia serpentina in the treatment of high blood pressure: a review of the literature. Circulation. 1955;12:220–9.
Lobay D. Rauwolfia in the treatment of hypertension. Integr Med. 2015;14:40.
Kiran K, Priya AJ, Devi RG. Medicinal and therapeutic uses of Rauwolfia serpentina-Indian snakeroot. Drug Invention Today 2018, 10.
Soni R, Jaiswal S, Bara JK, Saksena P. The use of Rauwolfia serpentina in hypertensive patients. J Biotechnol Biochem. 2016;2:28–32.
Hedgecock T, Phillips A, Ludrick B, Golden T, Wu N. Molecular mechanisms and applications of a reserpine-induced rodent model. SSR Inst Int J Life Sci. 2019;5:2160.
Schlepper M, Thormann J, Mitrovic V. Cardiovascular effects of forskolin and phosphodiesterase-III inhibitors. Inotropic Stimulation and myocardial energetics. 1989, 197–212.
Den Hertog A, Pielkenrood J, Van Den Akker J. The effect of forskolin on smooth muscle cells of guinea-pig taenia caeci. Eur J Pharmacol. 1984;106:181–4.
Lincoln TM, Fisher-Simpson V. A comparison of the effects of forskolin and nitroprusside on cyclic nucleotides and relaxation in the rat aorta. Eur J Pharmacol. 1984;101:17–27.
Geuns JM. Stevioside. Phytochemistry. 2003;64:913–21.
Chan P, Xu D-Y, Liu J-C, Chen Y-J, Tomlinson B, Huang W-P, Cheng J-T. The effect of stevioside on blood pressure and plasma catecholamines in spontaneously hypertensive rats. Life Sci. 1998;63:1679–84.
Ferri LA, Alves-Do-Prado W, Yamada SS, Gazola S, Batista MR, Bazotte RB. Investigation of the antihypertensive effect of oral crude stevioside in patients with mild essential hypertension. Phytother Res. 2006;20:732–6.
Liu J-C, Kao P-K, Chan P, Hsu Y-H, Hou C-C, Lien G-S, Hsieh M-H, Chen Y-J, Cheng J-T. Mechanism of the antihypertensive effect of stevioside in anesthetized dogs. Pharmacology. 2003;67:14–20.
Lam KY, Ling APK, Koh RY, Wong YP, Say YH. A review on medicinal properties of orientin. Adv Pharmacol Sci. 2016;2016:4104595.
Ren-Ren B, Xiao-Ming W, Jin-Yi X. Current natural products with antihypertensive activity. Chin J Nat Med. 2015;13:721–9.
Fu X-C, Wang M-W, Li S-P, Zhang Y, Wang H-L. Vasodilatation produced by orientin and its mechanism study. Biol Pharm Bull. 2005;28:37–41.
Mali VR, Mohan V, Bodhankar SL. Antihypertensive and cardioprotective effects of the Lagenaria siceraria fruit in N G-nitro-L-arginine methyl ester (L-NAME) induced hypertensive rats. Pharm Biol. 2012;50:1428–35.
Nikonov G, Manaeva ZA, Pek GY. Ostruthol from Xanthogallum purpurascens. Chem Nat Compd. 1966;2:295–6.
Patil AD, Freyer AJ, Breen A, Carte B, Johnson RK. Halistanol disulfate B, a novel sulfated sterol from the sponge Pachastrella sp.: inhibitor of endothelin converting enzyme. J Nat Prods. 1996;59:606–8.
Cao Y, Xie L, Liu K, Liang Y, Dai X, Wang X, Lu J, Zhang X, Li X. The antihypertensive potential of flavonoids from Chinese Herbal Medicine: a review. Pharmacol Res. 2021;174: 105919.
Maaliki D, Shaito AA, Pintus G, El-Yazbi A, Eid AH. Flavonoids in hypertension: a brief review of the underlying mechanisms. Curr Opin Pharmacol. 2019;45:57–65.
Grosso G, Godos J, Currenti W, Micek A, Falzone L, Libra M, Giampieri F, Forbes-Hernández TY, Quiles JL, Battino M. The effect of dietary polyphenols on vascular health and hypertension: current evidence and mechanisms of action. Nutrients. 2022;14:545.
Hügel HM, Jackson N, May B, Zhang AL, Xue CC. Polyphenol protection and treatment of hypertension. Phytomedicine. 2016;23:220–31.
Yang J, Li Y, Zhou H. Protective effect of total alkaloids from Uncariae Ramulus cum uncis combined with Raphani Semen on vascular endothelial cell injury by hypertension. Chin Tradit Patent Med. 2013;35:15–9.
Huachen J, Yunlun L, Yuehua J, Dongmei Q, Tao H, Haiqiang J. The effect and mechanism of component compatibility of rhynchophylla total alkaloids and sinapine cyanide sulfonate on change of vascular endothelial cells. Editor Off Chin J Arterioscler. 2013;21:397–403.
Khan H, Amin S. ACE inhibition of plant alkaloids targeted approach for selective inhibition. Mini-Rev Org Chem. 2017;14:85–9.
Santos PCJL, Krieger JE, Pereira AC. Renin–angiotensin system, hypertension, and chronic kidney disease: pharmacogenetic implications. J Pharmacol Sci. 2012;120:77–88.
Wong ZW, Thanikachalam PV, Ramamurthy S. Molecular understanding of the protective role of natural products on isoproterenol-induced myocardial infarction: a review. Biomed Pharmacother. 2017;94:1145–66.
Yeo HY, Kim OY, Lim HH, Kim JY, Lee JH. Association of serum lycopene and brachial-ankle pulse wave velocity with metabolic syndrome. Metabolism. 2011;60:537–43.
Abdelrahman IA, Ahad A, Raish M, Jardan YAB, Alam MA, Al-Jenoobi FI. Cinnamon modulates the pharmacodynamic & pharmacokinetic of amlodipine in hypertensive rats. Saudi Pharm J. 2023;31: 101737.
Preuss HG, Echard B, Polansky MM, Anderson R. Whole cinnamon and aqueous extracts ameliorate sucrose-induced blood pressure elevations in spontaneously hypertensive rats. J Am Coll Nutr. 2006;25:144–50.
Shouk R, Abdou A, Shetty K, Sarkar D, Eid AH. Mechanisms underlying the antihypertensive effects of garlic bioactives. Nutr Res. 2014;34:106–15.
Al-Qattan KK, Thomson M, Al-Mutawa’a S, Al-Hajeri D, Drobiova H, Ali M. Nitric oxide mediates the blood-pressure lowering effect of garlic in the rat two-kidney, one-clip model of hypertension. J Nutr. 2006;136:774S-776S.
Ginter E, Simko V. Garlic (Allium sativum L.) and cardiovascular diseases. Bratisl Lek Listy. 2010;111:452–6.
Ashraf MZ, Hussain M, Fahim M. Antiatherosclerotic effects of dietary supplementations of garlic and turmeric: restoration of endothelial function in rats. Life Sci. 2005;77:837–57.
Cabo J, Alonso R, Mata P. Omega-3 fatty acids and blood pressure. Br J Nutr. 2012;107:S195–200.
Bercea CI, Cottrell GS, Tamagnini F, McNeish AJ. Omega-3 polyunsaturated fatty acids and hypertension: a review of vasodilatory mechanisms of docosahexaenoic acid and eicosapentaenoic acid. Br J Pharmacol. 2021;178:860–77.
Anjos PJ, Lima AO, Cunha PS, De Sousa DP, Onofre AS, Ribeiro TP, Medeiros IA, Antoniolli ÂR, Quintans-Júnior LJ, Santos MR. Cardiovascular effects induced by linalool in normotensive and hypertensive rats. Zeitschrift für Naturforschung C. 2013;68:181–90.
Liang Y, Zhong Y, Li X, Xiao Y, Wu Y, Xie P. Biological evaluation of linalool on the function of blood vessels. Mol Med Rep. 2021;24:1–9.
Pakdeechote P, Prachaney P, Berkban W, Kukongviriyapan U, Kukongviriyapan V, Khrisanapant W, Phirawatthakul Y. Vascular and antioxidant effects of an aqueous mentha cordifolia extract in experimental NG-nitro-L-arginine methyl ester-induced hypertension. Zeitschrift für Naturforschung C. 2014;69:35–45.
Adedapo AD, Ajayi AM, Ekwunife NL, Falayi OO, Oyagbemi A, Omobowale TO, Adedapo AA. Antihypertensive effect of Phragmanthera incana (Schum) Balle on NG-nitro-L-Arginine methyl ester (L-NAME) induced hypertensive rats. J Ethnopharmacol. 2020;257: 112888.
Li Y-X, Wijesekara I, Li Y, Kim S-K. Phlorotannins as bioactive agents from brown algae. Process Biochem. 2011;46:2219–24.
Wijesekara I, Kim S-K. Angiotensin-I-converting enzyme (ACE) inhibitors from marine resources: prospects in the pharmaceutical industry. Mar Drugs. 2010;8:1080–93.
Wijesekara I, Yoon NY, Kim SK. Phlorotannins from Ecklonia cava (Phaeophyceae): biological activities and potential health benefits. BioFactors. 2010;36:408–14.
Sanjeewa KKA, Kim E-A, Son K-T, Jeon Y-J. Bioactive properties and potentials cosmeceutical applications of phlorotannins isolated from brown seaweeds: a review. J Photochem Photobiol, B. 2016;162:100–5.
Olivares-Molina A, Fernández K. Comparison of different extraction techniques for obtaining extracts from brown seaweeds and their potential effects as angiotensin I-converting enzyme (ACE) inhibitors. J Appl Phycol. 2016;28:1295–302.
Wijesinghe W, Ko S-C, Jeon Y-J. Effect of phlorotannins isolated from Ecklonia cava on angiotensin I-converting enzyme (ACE) inhibitory activity. Nurs Res Pract. 2011;5:93–100.
Jung HA, Hyun SK, Kim HR, Choi JS. Angiotensin-converting enzyme I inhibitory activity of phlorotannins from Ecklonia stolonifera. Fish Sci. 2006;72:1292–9.
Ko S-C, Kang MC, Kang N, Kim H-S, Lee S-H, Ahn G, Jung W-K, Jeon Y-J. Effect of angiotensin I-converting enzyme (ACE) inhibition and nitric oxide (NO) production of 6, 6′-bieckol, a marine algal polyphenol and its anti-hypertensive effect in spontaneously hypertensive rats. Process Biochem. 2017;58:326–32.
Shibata T, Yamaguchi K, Nagayama K, Kawaguchi S, Nakamura T. Inhibitory activity of brown algal phlorotannins against glycosidases from the viscera of the turban shell Turbo cornutus. Eur J Phycol. 2002;37:493–500.
Seca AM, Pinto DC. Overview on the antihypertensive and anti-obesity effects of secondary metabolites from seaweeds. Mar Drugs. 2018;16:237.
Venkatesan J, Keekan KK, Anil S, Bhatnagar I, Kim S-K. Phlorotannins. Encyclopedia of food chemistry; 2019, 515.
Mizrahy S, Peer D. Polysaccharides as building blocks for nanotherapeutics. Chem Soc Rev. 2012;41:2623–40.
Habib GB, Basra SS. Are there any new pharmacologic therapies on the horizon to better treat hypertension? A state-of-the-art paper. J Cardiovasc Pharmacol Ther. 2014;19:516–25.
Planes N, Caballero-George C. Marine and soil derived natural products: a new source of novel cardiovascular protective agents targeting the endothelin system. Planta Med. 2015;81:630–6.
Nasser SA, El-Mas MM. Endothelin ETA receptor antagonism in cardiovascular disease. Eur J Pharmacol. 2014;737:210–3.
Zhu H-B, Geng M-Y, Guan H-S, Zhang J-T. Antihypertensive effects of D-polymannuronic sulfate and its related mechanisms in renovascular hypertensive rats. Acta Pharmacol Sin. 2000;21:727–32.
Sandford PA, Baird J. Industrial utilization of polysaccharides. In: The polysaccharides. Amsterdam: Elsevier; 1983. p. 411–90.
Meisel H, Walsh D, Murray B, FitzGerald R. ACE inhibitory peptides. Nutraceutical proteins and peptides in health and disease 2006, 269–315.
Dicpinigaitis PV. Angiotensin-converting enzyme inhibitor-induced cough: ACCP evidence-based clinical practice guidelines. Chest. 2006;129:169S-173S.
Wu J, Liao W, Udenigwe CC. Revisiting the mechanisms of ACE inhibitory peptides from food proteins. Trends Food Sci Technol. 2017;69:214–9.
Šližytė R, Mozuraitytė R, Martínez-Alvarez O, Falch E, Fouchereau-Peron M, Rustad T. Functional, bioactive and antioxidative properties of hydrolysates obtained from cod (Gadus morhua) backbones. Process Biochem. 2009;44:668–77.
Zhao Y, Li B, Dong S, Liu Z, Zhao X, Wang J, Zeng M. A novel ACE inhibitory peptide isolated from Acaudina molpadioidea hydrolysate. Peptides. 2009;30:1028–33.
Ahhmed AM, Muguruma M. A review of meat protein hydrolysates and hypertension. Meat Sci. 2010;86:110–8.
Cardoso SM, Pereira OR, Seca AM, Pinto DC, Silva AM. Seaweeds as preventive agents for cardiovascular diseases: from nutrients to functional foods. Mar Drugs. 2015;13:6838–65.
Sato M, Hosokawa T, Yamaguchi T, Nakano T, Muramoto K, Kahara T, Funayama K, Kobayashi A, Nakano T. Angiotensin I-converting enzyme inhibitory peptides derived from wakame (Undaria pinnatifida) and their antihypertensive effect in spontaneously hypertensive rats. J Agric Food Chem. 2002;50:6245–52.
Beaulieu L, Sirois M, Tamigneaux É. Evaluation of the in vitro biological activity of protein hydrolysates of the edible red alga, Palmaria palmata (dulse) harvested from the Gaspe coast and cultivated in tanks. J Appl Phycol. 2016;28:3101–15.
Cian RE, López-Posadas R, Drago SR, De Medina FS, Martínez-Augustin O. A Porphyra columbina hydrolysate upregulates IL-10 production in rat macrophages and lymphocytes through an NF-κB, and p38 and JNK dependent mechanism. Food Chem. 2012;134:1982–90.
Qu W, Ma H, Li W, Pan Z, Owusu J, Venkitasamy C. Performance of coupled enzymatic hydrolysis and membrane separation bioreactor for antihypertensive peptides production from Porphyra yezoensis protein. Process Biochem. 2015;50:245–52.
Jiménez R, Duarte J, Perez-Vizcaino F. Epicatechin: endothelial function and blood pressure. J Agric Food Chem. 2012;60:8823–30.
Perez-Vizcaino F, Duarte J, Jimenez R, Santos-Buelga C, Osuna A. Antihypertensive effects of the flavonoid quercetin. Pharmacol Rep. 2009;61:67–75.
Laurent S. Antihypertensive drugs. Pharmacol Res. 2017;124:116–25.
Rates SMK. Plants as source of drugs. Toxicon. 2001;39:603–13.
Funding
This research received no external funding.
Author information
Authors and Affiliations
Contributions
All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Islam, M.R., Dhar, P.S., Akash, S. et al. Bioactive molecules from terrestrial and seafood resources in hypertension treatment: focus on molecular mechanisms and targeted therapies. Nat. Prod. Bioprospect. 13, 45 (2023). https://doi.org/10.1007/s13659-023-00411-1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13659-023-00411-1