Abstract
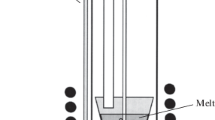
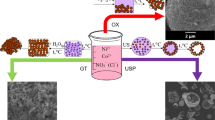
A new and highly efficient way of obtaining finely dispersed LiCoPO4 powder is developed with a given morphology from ammonium substituted precursor NH4CoPO4⋅H2O in a lithium nitrate melt. It is shown that the morphology of the obtained product is determined by the morphology of the used precursor and depends on the physicochemical conditions of its preparation. The obtained LiCoPO4 and its precursors are characterized by means of XRD, SEM, and BET. Electrochemical tests show the resulting powder is electrochemically active. Cathode material based on the obtained LiCoPO4 shows a high specific discharge capacity of 110 mA h/g at a current density corresponding to a charge/discharge rate of 1C, due to the high dispersion and lamellar morphology of the particles of the synthesized powder. The proposed procedure is characterized by the speed of obtaining the target product. It does not require the use of expensive equipment or additional stages of high-temperature crystallization and grinding, and can be scaled up to industrial use.








Similar content being viewed by others
REFERENCES
A. Dixit, SMC Bull. 10, 151 (2019).
L. Junxiang, W. Jiaqi, N. Youxuan, et al., Mater. Today, No. 43, 132 (2021).
H. Jiangtao, H. Weiyuan, X. Luyi, et al., Nanoscale 12, 15036 (2020).
V. V. Klyuev, V. V. Volynskii, V. N. Tyugaev, et al., RF Patent No. 2453950, Byull. Izobret., No. 17, 9 (2012).
N. Tolganbek, Y. Yerkinbekova, S. Kalybekkyzy, et al., J. Alloys Compd. 882, 160 (2021).
S. Kanungo, A. Bhattacharjee, N. Bahadursha, et al., Nanomaterials, No. 12, 32 (2022).
A. Kraytsberg and Y. Ein-Eli, Adv. Energy Mater., No. 2, 922 (2012).
S.-P. Chen, D. Lv, J. Chen, et al., Energy Fuels 36, 1232 (2022).
D. Jugović and D. Uskoković, J. Power Sources 190, 538 (2009).
S. Kirillov, I. V. Romanova, T. V. Lisnycha, et al., Electrochim. Acta 286, 163 (2018).
S. Karafiludis, A. Buzanich, C. Heinekamp, et al., Nanoscale, 1 (2023).
S. Karafiludis, A. G. Buzanich, Z. Kochovski, et al., Cryst. Growth Des. 22, 4305 (2022).
D. Choi, X. Li, W. A. Henderson, et al., Heliyon 2, e00081 (2016). https://doi.org/10.1016/j.heliyon.2016.e00081
M. B. Pinson and M. Z. Bazant, J. Electrochem. Soc. 160, 243 (2013).
E. Markevich, R. Sharabi, H. Gottlieb, et al., Electrochem. Commun., No. 15, 22 (2012).
Q. Truong, M. R. Devaraju, Y. Ganbe, et al., Sci. Rep., No. 4, 39 (2014).
X. Wu, M. Meledina, H. Tempel, et al., J. Power Sources 450, 227 (2020).
Funding
This work was performed as part of State Task no. FMEZ-2022-0015.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Zharov, N.V., Maslova, M.V., Ivanenko, V.I. et al. New Approaches to Synthesizing Nanostructured Electrode Materials Based on Double Lithium and Cobalt Phosphates in Salt Melts. Russ. J. Phys. Chem. 97, 2529–2535 (2023). https://doi.org/10.1134/S0036024423110365
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1134/S0036024423110365