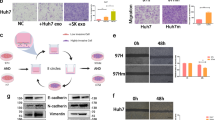
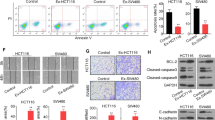
Abstract
Background
The current study probed into how tumor cell-derived exosomes (Exos) mediated hsa_circ_0001739/lncRNA AC159540.1 to manipulate microRNA (miR)-218-5p/FTO-N6-methyladenosine (m6A)/MYC signal axis in liver metastasis in colorectal cancer (CRC).
Methods
hsa_circ_0001739 and lncRNA AC159540.1 were identified as the upstream regulator of miR-218-5p using ENCORI and LncBase databases. Expression patterns of miR-218-5p, hsa_circ_0001739, lncRNA AC159540.1, FTO, and MYC were detected, accompanied by loss-and-gain-of function assays to examine their effects on CRC cell biological functions. SW480 cells-derived Exos were purified, followed by in vitro studies to uncover the effect of hsa_circ_0001739/lncRNA AC159540.
Results
miR-218-5p was downregulated while hsa_circ_0001739/lncRNA AC159540.1 was upregulated in CRC tissues and cells. Silencing of hsa_circ_0001739/lncRNA AC159540.1 restrained the malignant phenotypes of CRC cells. Exos-mediated hsa_circ_0001739/lncRNA AC159540.1 competitively inhibited miR-218-5p to elevate FTO and MYC. The inducing role of Exos-mediated hsa_circ_0001739/lncRNA AC159540.1 in CRC was also validated in vivo.
Conclusion
Conclusively, Exos-mediated circ_0001739/lncRNA AC159540.1 regulatory network is critical for CRC, offering a theoretical basis for CRC treatment.
Graphical abstract








Similar content being viewed by others
Data availability
The data and materials of the study can be obtained from the corresponding author upon request.
Code availability
Not applicable.
Abbreviations
- CRC :
-
Colorectal cancer
- FTO :
-
Fat mass and obesity-associated protein
- MYC :
-
MYC proto-oncogene, bHLH transcription factor
- m6A :
-
N6-methyladenosine
- PFN1 :
-
Profilin 1
- ncRNAs :
-
Non-coding RNAs
- cDNA :
-
Complementary DNA
- HIEC :
-
Human intestinal epithelial cell line
- DMEM :
-
Dulbecco’s modified Eagle’s medium
- FBS :
-
Fetal bovine serum
- PBS :
-
Phosphate buffer saline
- RT-qPCR :
-
Quantitative polymerase chain reaction
- SDS-PAGE :
-
Sodium dodecyl sulfate–polyacrylamide gel electrophoresis
- PVDF :
-
Polyvinylidene fluoride
- HRP :
-
Horseradish peroxidase
- CCK-8 :
-
Cell counting kit-8
- TUNEL :
-
Terminal deoxynucleotidyl transferase-mediated dUTP nick end-labeling
- DAB :
-
3,3′-Diaminobenzidine
- wt :
-
Wild type
- mut :
-
Mutant
- Me-RIP :
-
Methylated RNA immunoprecipitation
- HE :
-
Hematoxylin-eosin
- DLS :
-
Dynamic light scattering
- TEM :
-
Transmission electron microscopy
- RIPA :
-
Radioimmunoprecipitation assay
- BCA :
-
The bicinchoninic acid
- WHO :
-
World Health Organization
- HPA :
-
Human Protein Atlas
- GEO :
-
Gene Expression Omnibus
- FC :
-
Fold change
- OD :
-
Optical density
- ANOVA :
-
One-way analysis of variance
- NC :
-
Negative control
- oe-NC :
-
NC of overexpression plasmid
- oe-MYC :
-
Plasmid carrying MYC overexpression
- sh-NC :
-
NC shRNA
- sh-FTO-1 :
-
ShRNA targeting FTO-1
- sh-FTO-2 :
-
ShRNA targeting FTO-2
- sh-MYC :
-
ShRNA targeting MYC
- SPF :
-
Specified pathogen free
References
Agarwal V, Bell GW, Nam JW, Bartel DP. Predicting effective microRNA target sites in mammalian mRNAs. Elife. 2015;4:e05005. https://doi.org/10.7554/eLife.05005.
Bi J, Liu H, Cai Z, Dong W, Jiang N, Yang M, Huang J, Lin T. Circ-BPTF promotes bladder cancer progression and recurrence through the miR-31–5p/RAB27A axis. Aging (Albany NY). 2018;10:1964–76. https://doi.org/10.18632/aging.101520.
Ding Q, Chang CJ, Xie X, Xia W, Yang JY, Wang SC, Wang Y, Xia J, Chen L, Cai C, Li H, Yen CJ, Kuo HP, Lee DF, Lang J, Huo L, Cheng X, Chen YJ, Li CW, Jeng LB, Hsu JL, Li LY, Tan A, Curley SA, Ellis LM, Dubois RN, Hung MC. APOBEC3G promotes liver metastasis in an orthotopic mouse model of colorectal cancer and predicts human hepatic metastasis. J Clin Invest. 2011;121:4526–36. https://doi.org/10.1172/JCI45008.
Francavilla A, Turoczi S, Tarallo S, Vodicka P, Pardini B, Naccarati A. Exosomal microRNAs and other non-coding RNAs as colorectal cancer biomarkers: A review. Mutagenesis. 2020;35:243–60. https://doi.org/10.1093/mutage/gez038.
Fu Q, Cheng J, Zhang J, Zhang Y, Chen X, Xie J, Luo S. Downregulation of YEATS4 by miR-218 sensitizes colorectal cancer cells to L-OHP-induced cell apoptosis by inhibiting cytoprotective autophagy. Oncol Rep. 2016;36:3682–90. https://doi.org/10.3892/or.2016.5195.
Guo LL, Song CH, Wang P, Dai LP, Zhang JY, Wang KJ. Competing endogenous RNA networks and gastric cancer. World J Gastroenterol. 2015;21:11680–7. https://doi.org/10.3748/wjg.v21.i41.11680.
Haria PD, Baheti AD, Palsetia D, Ankathi SK, Choudhari A, Guha A, Saklani A, Sinha R. Follow-up of colorectal cancer and patterns of recurrence. Clin Radiol. 2021;76:908–15. https://doi.org/10.1016/j.crad.2021.07.016.
Hua J, Wang X, Ma L, Li J, Cao G, Zhang S, Lin W. CircVAPA promotes small cell lung cancer progression by modulating the miR-377-3p and miR-494-3p/IGF1R/AKT axis. Mol Cancer. 2022;21:123. https://doi.org/10.1186/s12943-022-01595-9.
Jiang X, Zhu Q, Wu P, Zhou F, Chen J. Upregulated long noncoding RNA LINC01234 predicts unfavorable prognosis for colorectal cancer and negatively correlates with KLF6 expression. Ann Lab Med. 2020;40:155–63. https://doi.org/10.3343/alm.2020.40.2.155.
Jin L, Han C, Zhai T, Zhang X, Chen C, Lian L. Circ_0030998 promotes tumor proliferation and angiogenesis by sponging miR-567 to regulate VEGFA in colorectal cancer. Cell Death Discov. 2021;7:160. https://doi.org/10.1038/s41420-021-00544-7.
Li JH, Liu S, Zhou H, Qu LH, Yang JH. starBase v2.0: Decoding miRNA-ceRNA, miRNA-ncRNA and protein-RNA interaction networks from large-scale CLIP-Seq data. Nucleic Acids Res. 2014;42:D92-7. https://doi.org/10.1093/nar/gkt1248.
Li L, Jiang Z, Zou X, Hao T. Exosomal circ_IFT80 enhances tumorigenesis and suppresses radiosensitivity in colorectal cancer by regulating miR-296-5p/MSI1 axis. Cancer Manag Res. 2021;13:1929–41. https://doi.org/10.2147/CMAR.S297123.
Li S, Li Y, Chen B, Zhao J, Yu S, Tang Y, Zheng Q, Li Y, Wang P, He X, Huang S. exoRBase: A database of circRNA, lncRNA and mRNA in human blood exosomes. Nucleic Acids Res. 2018;46:D106–12. https://doi.org/10.1093/nar/gkx891.
Liu M, Yin K, Guo X, Feng H, Yuan M, Liu Y, Zhang J, Guo B, Wang C, Zhou G, Zhou Z, Zhang CY, Chen X. Diphthamide biosynthesis 1 is a novel oncogene in colorectal cancer cells and is regulated by miR-218-5p. Cell Physiol Biochem. 2017;44:505–14. https://doi.org/10.1159/000485087.
Liu Y, Chen L, Liu T, Su X, Peng L, Chen J, Tan F, Xing P, Wang Z, Di J, Jiang B, Qu H. Genome-wide circular RNA (circRNA) and mRNA profiling identify a circMET-miR-410-3p regulatory motif for cell growth in colorectal cancer. Genomics. 2022;114:351–60. https://doi.org/10.1016/j.ygeno.2021.11.038.
Martin J, Petrillo A, Smyth EC, Shaida N, Khwaja S, Cheow HK, Duckworth A, Heister P, Praseedom R, Jah A, Balakrishnan A, Harper S, Liau S, Kosmoliaptsis V, Huguet E. Colorectal liver metastases: Current management and future perspectives. World J Clin Oncol. 2020;11:761–808. https://doi.org/10.5306/wjco.v11.i10.761.
Paraskevopoulou MD, Vlachos IS, Karagkouni D, Georgakilas G, Kanellos I, Vergoulis T, Zagganas K, Tsanakas P, Floros E, Dalamagas T, Hatzigeorgiou AG. DIANA-LncBase v2: Indexing microRNA targets on non-coding transcripts. Nucleic Acids Res. 2016;44:D231–8. https://doi.org/10.1093/nar/gkv1270.
Pegtel DM, Gould SJ. Exosomes. Annu Rev Biochem. 2019;88:487–514. https://doi.org/10.1146/annurev-biochem-013118-111902.
Peinado H, Zhang H, Matei IR, Costa-Silva B, Hoshino A, Rodrigues G, Psaila B, Kaplan RN, Bromberg JF, Kang Y, Bissell MJ, Cox TR, Giaccia AJ, Erler JT, Hiratsuka S, Ghajar CM, Lyden D. Pre-metastatic niches: Organ-specific homes for metastases. Nat Rev Cancer. 2017;17:302–17. https://doi.org/10.1038/nrc.2017.6.
Pu Y, Wei J, Wu Y, Zhao K, Wu Y, Wu S, Yang X, Xing C. THUMPD3-AS1 facilitates cell growth and aggressiveness by the miR-218-5p/SKAP1 axis in colorectal cancer. Cell Biochem Biophys. 2022;80:483–94. https://doi.org/10.1007/s12013-022-01074-4.
Ritchie ME, Phipson B, Wu D, Hu Y, Law CW, Shi W, Smyth GK. Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015;43:e47. https://doi.org/10.1093/nar/gkv007.
Roman-Canal B, Tarragona J, Moiola CP, Gatius S, Bonnin S, Ruiz-Miro M, Sierra JE, Rufas M, Gonzalez E, Porcel JM, Gil-Moreno A, Falcon-Perez JM, Ponomarenko J, Matias-Guiu X, Colas E. EV-associated miRNAs from peritoneal lavage as potential diagnostic biomarkers in colorectal cancer. J Transl Med. 2019;17:208. https://doi.org/10.1186/s12967-019-1954-8.
Ruan DY, Li T, Wang YN, Meng Q, Li Y, Yu K, Wang M, Lin JF, Luo LZ, Wang DS, Lin JZ, Bai L, Liu ZX, Zhao Q, Wu XY, Ju HQ, Xu RH. FTO downregulation mediated by hypoxia facilitates colorectal cancer metastasis. Oncogene. 2021;40:5168–81. https://doi.org/10.1038/s41388-021-01916-0.
Sarvizadeh M, Ghasemi F, Tavakoli F, Sadat Khatami S, Razi E, Sharifi H, Biouki NM, Taghizadeh M. Vaccines for colorectal cancer: An update. J Cell Biochem. 2019;120:8815–28. https://doi.org/10.1002/jcb.28179.
Satoh K, Yachida S, Sugimoto M, Oshima M, Nakagawa T, Akamoto S, Tabata S, Saitoh K, Kato K, Sato S, Igarashi K, Aizawa Y, Kajino-Sakamoto R, Kojima Y, Fujishita T, Enomoto A, Hirayama A, Ishikawa T, Taketo MM, Kushida Y, Haba R, Okano K, Tomita M, Suzuki Y, Fukuda S, Aoki M, Soga T. Global metabolic reprogramming of colorectal cancer occurs at adenoma stage and is induced by MYC. Proc Natl Acad Sci U S A. 2017;114:E7697-E706. https://doi.org/10.1073/pnas.1710366114
Schetter AJ, Okayama H, Harris CC. The role of microRNAs in colorectal cancer. Cancer J. 2012;18:244–52. https://doi.org/10.1097/PPO.0b013e318258b78f.
Shang A, Gu C, Wang W, Wang X, Sun J, Zeng B, Chen C, Chang W, Ping Y, Ji P, Wu J, Quan W, Yao Y, Zhou Y, Sun Z, Li D. Exosomal circPACRGL promotes progression of colorectal cancer via the miR-142-3p/miR-506-3p- TGF-beta1 axis. Mol Cancer. 2020;19:117. https://doi.org/10.1186/s12943-020-01235-0.
Siveen KS, Raza A, Ahmed EI, Khan AQ, Prabhu KS, Kuttikrishnan S, Mateo JM, Zayed H, Rasul K, Azizi F, Dermime S, Steinhoff M, Uddin S. The role of extracellular vesicles as modulators of the tumor microenvironment, metastasis and drug resistance in colorectal cancer. Cancers (Basel). 2019;11:746. https://doi.org/10.3390/cancers11060746.
Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2021;71:209–49. https://doi.org/10.3322/caac.21660.
Tang Z, Li C, Kang B, Gao G, Li C, Zhang Z. GEPIA: A web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res. 2017;45:W98–102. https://doi.org/10.1093/nar/gkx247.
Uhlen M, Zhang C, Lee S, Sjostedt E, Fagerberg L, Bidkhori G, Benfeitas R, Arif M, Liu Z, Edfors F, Sanli K, von Feilitzen K, Oksvold P, Lundberg E, Hober S, Nilsson P, Mattsson J, Schwenk JM, Brunnstrom H, Glimelius B, Sjoblom T, Edqvist PH, Djureinovic D, Micke P, Lindskog C, Mardinoglu A, Ponten F. A pathology atlas of the human cancer transcriptome. Science. 2017;357:eaan2507. https://doi.org/10.1126/science.aan2507.
Valcz G, Galamb O, Krenacs T, Spisak S, Kalmar A, Patai AV, Wichmann B, Dede K, Tulassay Z, Molnar B. Exosomes in colorectal carcinoma formation: ALIX under the magnifying glass. Mod Pathol. 2016;29:928–38. https://doi.org/10.1038/modpathol.2016.72.
Wang J, Zhou J, Jiang C, Zheng J, Namba H, Chi P, Asakawa T. LNRRIL6, a novel long noncoding RNA, protects colorectal cancer cells by activating the IL-6-STAT3 pathway. Mol Oncol. 2019a;13:2344–60. https://doi.org/10.1002/1878-0261.12538.
Wang L, Cho KB, Li Y, Tao G, Xie Z, Guo B. Long noncoding RNA (lncRNA)-mediated competing endogenous RNA networks provide novel potential biomarkers and therapeutic targets for colorectal cancer. Int J Mol Sci. 2019b;20:5758. https://doi.org/10.3390/ijms20225758.
Wang W, Lou W, Ding B, Yang B, Lu H, Kong Q, Fan W. A novel mRNA-miRNA-lncRNA competing endogenous RNA triple sub-network associated with prognosis of pancreatic cancer. Aging (Albany NY). 2019c;11:2610–27. https://doi.org/10.18632/aging.101933.
Wang X, Ren Y, Ma S, Wang S. Circular RNA 0060745, a novel circRNA, promotes colorectal cancer cell proliferation and metastasis through miR-4736 sponging. Onco Targets Ther. 2020;13:1941–51. https://doi.org/10.2147/OTT.S240642.
Wang X, Zhang Y, Huang L, Zhang J, Pan F, Li B, Yan Y, Jia B, Liu H, Li S, Zheng W. Decreased expression of hsa_circ_001988 in colorectal cancer and its clinical significances. Int J Clin Exp Pathol. 2015;8:16020–5.
Xie Y, Dang W, Zhang S, Yue W, Yang L, Zhai X, Yan Q, Lu J. The role of exosomal noncoding RNAs in cancer. Mol Cancer. 2019;18:37. https://doi.org/10.1186/s12943-019-0984-4.
Ye J, Liu J, Tang T, Xin L, Bao X, Yan Y. LINC00963 affects the development of colorectal cancer via miR-532-3p/HMGA2 axis. Cancer Cell Int. 2021;21:87. https://doi.org/10.1186/s12935-020-01706-w.
Yue C, Chen J, Li Z, Li L, Chen J, Guo Y. microRNA-96 promotes occurrence and progression of colorectal cancer via regulation of the AMPKalpha2-FTO-m6A/MYC axis. J Exp Clin Cancer Res. 2020;39:240. https://doi.org/10.1186/s13046-020-01731-7.
Zarour LR, Anand S, Billingsley KG, Bisson WH, Cercek A, Clarke MF, Coussens LM, Gast CE, Geltzeiler CB, Hansen L, Kelley KA, Lopez CD, Rana SR, Ruhl R, Tsikitis VL, Vaccaro GM, Wong MH, Mayo SC. Colorectal cancer liver metastasis: Evolving paradigms and future directions. Cell Mol Gastroenterol Hepatol. 2017;3:163–73. https://doi.org/10.1016/j.jcmgh.2017.01.006.
Zhang C, Wu R, Zhu H, Hu YZ, Jiang H, Lin NM, He QJ, Yang B. Enhanced anti-tumor activity by the combination of TRAIL/Apo-2L and combretastatin A-4 against human colon cancer cells via induction of apoptosis in vitro and in vivo. Cancer Lett. 2011;302:11–9. https://doi.org/10.1016/j.canlet.2010.12.001.
Zhang J, Li S, Zhang X, Li C, Zhang J, Zhou W. LncRNA HLA-F-AS1 promotes colorectal cancer metastasis by inducing PFN1 in colorectal cancer-derived extracellular vesicles and mediating macrophage polarization. Cancer Gene Ther. 2021a. https://doi.org/10.1038/s41417-020-00276-3.
Zhang L, Yu D. Exosomes in cancer development, metastasis, and immunity. Biochim Biophys Acta Rev Cancer. 2019;1871:455–68. https://doi.org/10.1016/j.bbcan.2019.04.004.
Zhang Z, Gao Q, Wang S. Kinase GSK3beta functions as a suppressor in colorectal carcinoma through the FTO-mediated MZF1/c-Myc axis. J Cell Mol Med. 2021b;25:2655–65. https://doi.org/10.1111/jcmm.16291.
Zhao J, Lin H, Huang K. Mesenchymal stem cell-derived extracellular vesicles transmitting microRNA-34a-5p suppress tumorigenesis of colorectal cancer through c-MYC/DNMT3a/PTEN axis. Mol Neurobiol. 2022;59:47–60. https://doi.org/10.1007/s12035-021-02431-9.
Zhou H, Liu Z, Wang Y, Wen X, Amador EH, Yuan L, Ran X, Xiong L, Ran Y, Chen W, Wen Y. Colorectal liver metastasis: Molecular mechanism and interventional therapy. Signal Transduct Target Ther. 2022;7:70. https://doi.org/10.1038/s41392-022-00922-2.
Zhou XG, Huang XL, Liang SY, Tang SM, Wu SK, Huang TT, Mo ZN, Wang QY. Identifying miRNA and gene modules of colon cancer associated with pathological stage by weighted gene co-expression network analysis. Onco Targets Ther. 2018;11:2815–30. https://doi.org/10.2147/OTT.S163891.
Zou D, Dong L, Li C, Yin Z, Rao S, Zhou Q. The m(6)A eraser FTO facilitates proliferation and migration of human cervical cancer cells. Cancer Cell Int. 2019;19:321. https://doi.org/10.1186/s12935-019-1045-1.
Funding
This work is supported by the National Natural Science Foundation of China (82002866, 82072384, 81602416, 81602691) and Guangdong Basic and Applied Basic Research Foundation (2023A1515030063).
Author information
Authors and Affiliations
Contributions
C.-F. Y. and Y.-M. G. designed and oversaw the study. C.-F. Y., J.-G. C., Z.-Y. L., L.-S. L., H.-X. X., and B. Z. initiated or participated in sample recruitment and sample preparation. J.-G. C., J.-R. C., and Y.-M. G. performed the bioinformatics analyses and interpreted the data. C.-F. Y., Z.-Y. L., L.-S. L., and H.-X. X. performed the experiment and analyzed the data. C.-F. Y., J.-G. C., Z.-Y. L., B. Z., and Y.-M. G. drafted the manuscript. All authors critically reviewed the manuscript and approved the final version.
Corresponding author
Ethics declarations
Ethics approval and consent to participate
The study procedure was ratified by the Ethics Committee of The First Affiliated Hospital of Sun Yat-Sen University and was implemented on the basis of the Declaration of Helsinki. All the patients provided their written informed consent. Animal experimentation was ratified by the Ethics Committee of The First Affiliated Hospital of Sun Yat-Sen University and performed in light of the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health.
Consent for publication
Not applicable.
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Bin Zhang and Yun-Miao Guo jointly supervised this work.
Graphical Headlights
1. Knockdown of hsa_circ_0001739 or lncRNA AC159540.1 can inhibit the proliferation, migration and invasion of CRC cells and promote their apoptosis.
2. EVs containing overexpressed hsa_circ_0001739/lncRNA AC159540.1 promote the proliferation, migration and invasion of CRC cells.
3. hsa_circ_0001739/lncRNA AC159540 dual pathway regulates miR-218-5p/FTO axis to promote CRC cell proliferation, migration and invasion.
4. EVs-mediated hsa_circ_0001739/lncRNA AC159540.1 promotes tumorigenesis and liver metastasis of CRC cells in nude mice.
5. This study provides a theoretical basis for understanding of the molecular mechanism of liver metastasis in CRC and search for potential therapeutic targets.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yue, CF., Chen, JG., Li, ZY. et al. Tumor cell-derived exosomes mediating hsa_circ_0001739/lncRNA AC159540.1 facilitate liver metastasis in colorectal cancer. Cell Biol Toxicol 39, 2551–2568 (2023). https://doi.org/10.1007/s10565-023-09837-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10565-023-09837-2