Abstract
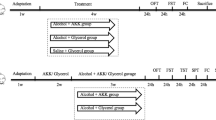
Brain damage caused by ethanol abuse may lead to permanent damage, including severe dementia. The aim of this study was to investigate the effects of ginger powder on ethanol-induced cognitive disorders by examining oxidative damage and inflammation status, and the gene expression of N-methyl-D-aspartate (NMDA) and γ-Aminobutyric acid (GABA)-A receptors in the hippocampus of male rats. 24 adult male Sprague–Dawley rats were allocated randomly to four groups as follows control, ethanol (4g/kg/day, by gavage), ginger (1g/kg/day, by gavage), and ginger-ethanol. At the end of the study, memory and learning were evaluated by the shuttle box test. Moreover, to explore mechanisms involved in ethanol-induced cognitive impairment and the protective effect of ginger, the expression of Nuclear factor kappa B (NF-κB), nuclear factor erythroid 2–related factor 2 (Nrf2), NMDA receptor, and GABA-A receptor was measured along with inflammatory and oxidative biomarkers in the hippocampus tissue. The results showed that ethanol could induce cognitive impairment in the ethanol group, while pretreatment with ginger could reverse it. The gene expression of the NF-κB/ Tumor necrosis factor (TNF)-α/Interleukin (IL)-1β pathway and NMDA and GABA-A receptors significantly increased in the ethanol group compared to the control group. While pretreatment with ginger could significantly improve ethanol-induced cognitive impairment through these pathways in the ginger-ethanol group compared to the ethanol group (P < 0.05). It can be concluded that ginger powder could ameliorate ethanol-induced cognitive impairment by modulating the expression of NMDA and GABA-A receptors and inhibiting oxidative damage and the NF-κB/TNF-α/IL-1β pathway in the rat hippocampus.







Similar content being viewed by others
Data availability
The data used to support the findings of this study is available from the corresponding author upon request.
Code availability
Not applicable.
References
Aebi H (1984) [13] Catalase in vitro. Methods Enzymol 105:121–126
Ahmed Mustafa Z, Hamed Ali R, Rostum Ali D, Abdulkarimi R, Abdulkareem NK, Akbari A (2021) The combination of ginger powder and zinc supplement improves the fructose-induced metabolic syndrome in rats by modulating the hepatic expression of NF-κB, mTORC1, PPAR-α SREBP-1c, and Nrf2. J Food Biochem 45:e13546
Akbari A, Jelodar G (2013) The effect of oxidative stress and antioxidants on men fertility. Zahedan J Res Med Sci 15:1–7
Akbari A, Jelodar G, Nazifi S (2016) The proposed mechanisms of radio frequency waves (RFWs) on nervous system functions impairment. Comp Clin Pathol 25:1289–1301
Akbari A, Nasiri K, Heydari M, Mosavat SH, Iraji A (2017) The protective effect of hydroalcoholic extract of Zingiber officinale roscoe (Ginger) on ethanol-induced reproductive toxicity in male rats. J Evid Based Complementary Altern Med 22:609–617
Al Hroob AM, Abukhalil MH, Alghonmeen RD, Mahmoud AM (2018) Ginger alleviates hyperglycemia-induced oxidative stress, inflammation and apoptosis and protects rats against diabetic nephropathy. Biomed Pharmacother 106:381–389
Al-Amin ZM, Thomson M, Al-Qattan KK, Peltonen-Shalaby R, Ali M (2006) Anti-diabetic and hypolipidaemic properties of ginger (Zingiber officinale) in streptozotocin-induced diabetic rats. Br J Nutr 96:660–666
Alirezei M, Jelodar G, Niknam P, Al E (2011) Betaine prevents ethanol-induced oxidative stress and reduces total homocysteine in the rat cerebellum. J Physiol Biochem 67:605–612
Atta A, Elkoly T, Mouneir S, Kamel G, Alwabel N, Zaher S (2010) Hepatoprotective effect of methanol extracts of Zingiber officinale and Cichoriumintybus. Indian J Pharm Sci 72:564–570
Azam F, Amer AM, Abulifa AR, Elzwawi MM (2014) Ginger components as new leads for the design and development of novel multi-targeted anti-Alzheimer’s drugs: a computational investigation. Drug Des Dev Ther 8:2045–2059
Cao J, Hou J, Ping J, Cai D (2018) Advances in developing novel therapeutic strategies for Alzheimer’s disease. Mol Neurodegener 13:64–64
Chandrasekar R (2013) Alcohol and NMDA receptor: current research and future direction. Front Mol Neurosci 6:14–21
Cushman JD, Moore MD, Jacobs NS, Olsen RW, Fanselow MS (2011) Behavioral pharmacogenetic analysis on the role of the α4 GABAA receptor subunit in the ethanol-mediated impairment of hippocampus-dependent contextual learning. Alcoholism: Clin Exp Res 35:1948–1959
Davies M (2003) The role of GABAA receptors in mediating the effects of alcohol in the central nervous system. J Psychiatry Neurosci: JPN 28:263–274
Dröge W (2002) Free radicals in the physiological control of cell function. Physiol Rev 82:47–95
El-Akabawy G, El-Kholy W (2014) Neuroprotective effect of ginger in the brain of streptozotocin-induced diabetic rats. Ann Anat - Anatomischer Anzeiger 196:119–128
Farooqui T, Farooqui AA (2017) Neuroprotective effects of phytochemicals in neurological disorders. The Ohio State University, Columbus, OH, USA
Fathi R, Akbari A, Nasiri K, Chardahcherik M (2021) Ginger (Zingiber officinale roscoe) extract could upregulate the renal expression of NRF2 and TNFα and prevents ethanol-induced toxicity in rat kidney. Avicenna J Phytomed 11:134–139
Fukushima W, Miyake Y, Tanaka K, Sasaki S, Kiyohara C, Tsuboi Y, Yamada T, Oeda T, Miki T, Kawamura N, Sakae N, Fukuyama H, Hirota Y, Nagai M, Kinki F (2010) Alcohol drinking and risk of Parkinson’s disease: a case-control study in Japan. BMC Neurol 10:111–119
Gawel K, Kukula KW, Banono NS, Nieoczym D, Targowska-Duda KM, Czernicka L, Parada-Turska J, Esguerra CV (2021) 6-Gingerol, a major constituent of Zingiber officinale Rhizoma, exerts anticonvulsant activity in the pentylenetetrazole-induced seizure model in Larval Zebrafish. Int J Mol Sci 22:7745–7751
Ghasemzadeh A, Jaafar HZ, Rahmat A (2010) Antioxidant activities, total phenolics and flavonoids content in two varieties of Malaysia young ginger (Zingiber officinale Roscoe). Molecules 15:4324–4333
Glass CK, Saijo K, Winner B, Marchetto MC, Gage FH (2010) Mechanisms underlying inflammation in neurodegeneration. Cell 140:918–934
Gohlke J, Griffith W, Faustman E (2008) Computational models of ethanol-induced neurodevelopmental toxicity across species: Implications for risk assessment. Birth Defects Res B Dev Reprod Toxicol 83:1–11
Gomar A, Hosseini A, Mirazi N, Gomar M (2015) Effect of Zingiber Officinale (ginger rhizomes) hydroethanolic extract on hyoscine-induced memory impairment in adult male rats. Int Clin Neurosci J 2:105–110
Haorah J, Ramirez SH, Floreani N, Gorantla S, Morsey B, Persidsky Y (2008) Mechanism of alcohol-induced oxidative stress and neuronal injury. Free Radic Biol Med 45:1542–1550
Harrison NL, Skelly MJ, Grosserode EK, Lowes DC, Zeric T, Phister S, Salling MC (2017) Effects of acute alcohol on excitability in the CNS. Neuropharmacology 122:36–45
Hokenson RE, Short AK, Chen Y, Pham AL, Adams ET, Bolton JL, Swarup V, Gall CM, Baram TZ (2021) Unexpected role of physiological estrogen in acute stress-induced memory deficits. J Neurosci 41:648–662
Hussein UK, Hassan NEHY, Elhalwagy MEA, Zaki AR, Abubakr HO, Nagulapalli Venkata KC, Jang KY, Bishayee A (2017) Ginger and propolis exert neuroprotective effects against monosodium glutamate-induced neurotoxicity in rats. Molecules (Basel, Switzerland) 22:1928–1934
Jafarpour M, Yousefi G, Hamedi A, Shariat A, Salehi A, Heydari M (2016) Effect of a traditional syrup from Citrus medica L. fruit juice on migraine headache: A randomized double blind placebo controlled clinical trial. J Ethnopharmacol 179:170–176
Jin Z, Bhandage AK, Bazov I, Kononenko O, Bakalkin G, Korpi ER, Birnir B (2014) Selective increases of AMPA, NMDA, and kainate receptor subunit mRNAs in the hippocampus and orbitofrontal cortex but not in prefrontal cortex of human alcoholics. Front Cell Neurosci 8:11–21
Kim CY, Seo Y, Lee C, Park GH, Jang JH (2018) Neuroprotective effect and molecular mechanism of [6]-gingerol against scopolamine-induced amnesia in C57BL/6 Mice. Evid Based Complement Altern Med 2018:8941564–8941571
Kumar S, Fleming RL, Morrow AL (2004) Ethanol regulation of gamma-aminobutyric acid A receptors: genomic and nongenomic mechanisms. Pharmacol Ther 101:211–226
Kumar S, Porcu P, Werner DF, Matthews DB, Diaz-Granados JL, Helfand RS, Morrow AL (2009) The role of GABA(A) receptors in the acute and chronic effects of ethanol: a decade of progress. Psychopharmacology 205:529–564
Lantz RC, Chen G, Sarihan M, Solyom A, Jolad S, Timmermann B (2007) The effect of extracts from ginger rhizome on inflammatory mediator production. Phytomedicine 14:123–128
Lim S, Moon M, Oh H, Kim HG, Kim SY, Oh MS (2014) Ginger improves cognitive function via NGF-induced ERK/CREB activation in the hippocampus of the mouse. J Nutr Biochem 25:1058–1065
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2− ΔΔCT method. Methods 25:402–408
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Ma H, Li J (2022) The ginger extract could improve diabetic retinopathy by inhibiting the expression of e/iNOS and G6PDH, apoptosis, inflammation, and angiogenesis. J Food Biochem 46:e14084
Marefati N, Abdi T, Beheshti F, Vafaee F, Mahmoudabady M, Hosseini M (2021) Zingiber officinale (Ginger) hydroalcoholic extract improved avoidance memory in rat model of streptozotocin-induced diabetes by regulating brain oxidative stress. Horm Mol Biol Clin Investig 43:15–26
Moon M, Kim HG, Choi JG, Oh H, Lee PK, Ha SK, Kim SY, Park Y, Huh Y, Oh MS (2014) 6-Shogaol, an active constituent of ginger, attenuates neuroinflammation and cognitive deficits in animal models of dementia. Biochem Biophys Res Commun 449(1):8–13. https://doi.org/10.1016/j.bbrc.2014.04.121. (Erratum in: Biochem Biophys Res Commun. 2020 Jan 8;521(2):545. PMID: 24796668)
Mosavat SH, Ghahramani L, Sobhani Z, Haghighi ER, Heydari M (2015) Topical Allium ampeloprasum Subsp Iranicum (Leek) extract cream in patients with symptomatic hemorrhoids a pilot randomized and controlled clinical trial. J Evid Based Complement Altern Med 20:132–136
Nagy J (2008) Alcohol related changes in regulation of NMDA receptor functions. Curr Neuropharmacol 6:39–54
Niazi M, Hashempu MH, Taghizadeh M, Heydari M, Shariat A (2017) Efficacy of topical Rose (Rosa damascena Mill.) oil for migraine headache: A randomized double-blinded placebo-controlled cross-over trial. Complement Ther Med 34:35–41
Nicoll R, Henein M (2009) Ginger (Zingiber officinale Roscoe): a hot remedy for cardiovascular disease? Int J Cardiol 131:408–409
Perra S, Pillolla G, Luchicchi A, Pistis M (2008) Alcohol inhibits spontaneous activity of basolateral amigdala proyection neurons in the rat: Involvement of the endocannabinoid system. Alcohol Clin Exp Res 32:443–449
Pervin Z, Stephen JM (2021) Effect of alcohol on the central nervous system to develop neurological disorder: pathophysiological and lifestyle modulation can be potential therapeutic options for alcohol-induced neurotoxication. AIMS Neurosci 8:390–399
Prosser R, Mangrum C, Glass J (2008) Acute ethanol modulates glutamatergic and serotonergic phase shifts of the mouse circadian clock in vitro. Neurosci Lett 152:837–848
Puglia MP, Valenzuela CF (2010) Ethanol acutely inhibits ionotropic glutamate receptor-mediated responses and long-term potentiation in the developing CA1 hippocampus. Alcoholism: Clin Exp Res 34:594–606
Qasemzadeh MJ, Sharifi H, Hamedanian M, Gharehbeglou M, Heydari M, Sardari M, Akhlaghdoust M, Minae MB (2015) The effect of Viola odorata flower syrup on the cough of children with asthma a double-blind, randomized controlled trial. J Evid Based Complementary Altern Med 20:287–291
Rafieian-Kopaei M (2013) Medicinal plants for renal injury prevention. J Renal Inj Prev 2:63–71
Rauca C, Pohle W, Grunenberg K, Franze S (2000) Hypothermia inhibits pentylenetetrazol kindling and prevents kindling-induced deficit in shuttle-box avoidance. Pharmacol Biochem Behav 65:23–30
Roberto M, Treistman S, Pietrzykowsk A, Weiner J, Galindo R, Mameli M, Al E (2006) Actions of acute and chronic ethanol on presynaptic terminals. Alcohol Clin Exp Res 30:222–232
Shanley MR, Miura Y, Guevara CA, Onoichenco A, Kore R, Ustundag E, Darwish R, Renzoni L, Urbaez A, Blicker E, Seidenberg A, Milner TA, Friedman AK (2023) Estrous cycle mediates midbrain neuron excitability altering social behavior upon stress. J Neurosci 43:736–748
Sharifi H, Minaie MB, Qasemzadeh MJ, Ataei N, Gharehbeglou M, Heydari M (2015) Topical use of Matricaria recutita L (Chamomile) oil in the treatment of monosymptomatic enuresis in children a double-blind randomized controlled trial. J Evid Based Complementary Altern Med. https://doi.org/10.1177/2156587215608989
Shukla Y, Singh M (2007) Cancer preventive properties of ginger: a brief review. Food Chem Toxicol 45:683–690
Taherianfard M, Farhangdareshuri F, Shomali T (2018) Interactive effect of aqueous-alcoholic extract of ginger as well as GABAA receptor agonist and antagonist on pain sensitivity in male rats. Physiol Pharmacol 22:247–253
Wallner M, Hanchar H, Olsen R (2006) Low-dose alcohol actions on alpha4beta3delta GABAA receptors are reversed by the behavioral alcohol antagonist Ro15-4513. Proc Natl Acad Sci USA 103:8540–8545
Wang P, Luo Q, Qiao H, Ding H, Cao Y, Yu J, Liu R, Zhang Q, Zhu H, Qu L (2017) The Neuroprotective effects of carvacrol on ethanol-induced hippocampal neurons impairment via the antioxidative and antiapoptotic pathways. Oxid Med Cell Longev 2017:4079425
Wilkie M, Besheer J, Kelley S, Kumar S, O’buckley T, Morrow A, Hodge C (2007) Acute ethanol administration rapidly increases phosphorylation of conventional protein kinase C in specific mammalian brain regions in vivo. Alcohol Clin Exp Res 31:1259–1267
Xiang Y, Kim KY, Gelernter J, Park IH, Zhang H (2015) Ethanol upregulates NMDA receptor subunit gene expression in human embryonic stem cell-derived cortical neurons. PLoS ONE 10:e0134907–e0134907
Zhou M, Baudry M (2006) Developmental changes in NMDA neurotoxicity reflect developmental changes in subunit composition of NMDA receptors. J Neurosci 26:2956–2963
Zhou Y, Zheng J, Li S, Zhou T, Zhang P, Li HB (2016) Alcoholic beverage consumption and chronic diseases. Int J Environ Res Public Health 13:522–531
Acknowledgements
This research project was performed at the Department of Neurology, General Hospital of Ningxia Medical University, and was also supported by the scientific and research group affiliated with Dr. Abolfazl Akbari, General Hospital of Ningxia Medical University, and Xi’an Gaoxin Hospital.
Funding
None.
Author information
Authors and Affiliations
Contributions
JW: Resources, Methodology, Investigation. AA: Conceptualization, Project administration, Resources, Writing review & editing. MC: Methodology, Analysis, writing original draft, Writing review & editing. JW: Supervision, Resources, Writing review & editing. All the authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the General Hospital of Ningxia Medical University animal ethical committee, Approved No. GHNMU2021-0518.
Consent to participate
Not applicable.
Consent for publication
All the authors have read this version of the manuscript and agreed to publish it.
Conflicts of interest/competing interests
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Wang, J., Akbari, A., Chardahcherik, M. et al. Ginger (Zingiber Officinale Roscoe) ameliorates ethanol-induced cognitive impairment by modulating NMDA and GABA-A receptors in rat hippocampus. Metab Brain Dis 39, 67–76 (2024). https://doi.org/10.1007/s11011-023-01301-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11011-023-01301-8