Abstract
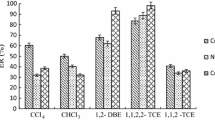
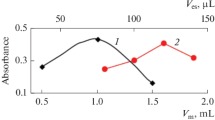
In this research, a dispersive solid phase extraction procedure based on changing the solubility of octadecylamine with pH was proposed to determine Ag+ ions in different water samples. For this purpose, first, the pH of sample solution containing the analyte was adjusted to 10.5. Then desired volume of the octadecylamine dissolved in acidic solution was injected into the solution. Because of the low solubility of octadecylamine in alkaline solution, a cloudy state was formed. The produced octadecylamine particles acted as a complexing agent for Ag+ ions and adsorbent for the formed complex. The obtained cloudy solution was centrifuged and the sedimented particles were removed and dissolved in a diluted nitric acid solution. It was injected into a flame atomic absorption spectrometry to determine the extracted amounts of the analyte. The effect of important parameters such as the amount of octadecylamine, volume of nitric acid, and centrifugation and vortexing conditions on the extraction efficiency of the procedure was studied and optimized. In optimal conditions, the developed method showed a linear range of 0.50–200 µg L−1. The limits of detection and quantification were 0.18 and 0.50 µg L−1, respectively. Extraction recovery was 93.6%. The relative standard deviations were less than 4%. The effectiveness of the method was investigated by determination of Ag+ ions in water and wastewater samples.
Graphical abstract





Similar content being viewed by others
Data availability
Not applicable.
References
A.S. Kholmogorova, M.L. Chernysh, L.K. Neudachina, I.S. Puzyrev, React. Funct. Polym. 152, 104596 (2020)
M.T. Doménech-Carbó, F.D. Turo, N. Montoya, F. Catalli, A. Doménech-Carbó, C.D. Vito, Sci. Rep. 8, 10676 (2018)
E.Y. Frag, N.A. El-ZaherSally, E.A. Elashery, Microchem. J. 155, 104750 (2020)
N. Duran, P.D. Marcato, G.I.H. De Souza, O.L. Alves, E. Esposito, J. Biomed. Nanotech. 3, 203–208 (2007)
S.P. Deshmukh, S.M. Patil, S.B. Mullani, S.D. Delekar, Mater. Sci. Eng. 97, 954–965 (2019)
P.D. Howe, S. Dobson, Silver and silver compounds: environmental aspects, In: Concise International Chemical Assessment Document, 44, World Health Organi-zation, Geneva, 2002.
L. Kocurova, I.S. Balogh, L. Nagy, F. Billes, A. Simon, V. Andruch, Microchem. J. 99, 514 (2011)
Eisler R. Silver hazards to fish, wildlife and invertebrates: a synoptic review, US Department of the Interior, National Biological Service, Washington, DC, 1996p. 44.
X. Yang, Zh. Ji, X. Yang, G. Li, X. Liao, Saudi J. Biol. Sci. 24, 589–594 (2017)
M. Sikder, J.R. Lead, G.T. Chandler, M. Baalousha, Sci. Total. Environ. 618, 597–607 (2018)
R.K. Shah, Orien. J. Chem. 32, 499–507 (2016)
R. Cheng, L. Li, S. Ou, Y. Bu, L. Dai, Y. Xue, Roy. Soc. Ch. 6, 36218–36222 (2016)
Y. Yang, L. Luo, H.P. Li, Q. Wang, Z.G. Yang, C.L. Long, Talanta 161, 342–349 (2016)
R. Mikelova, J. Baloun, J. Petrlova, V. Adam, L. Havel, J. Petrek, A. Horna, R. Kizek, Bioelectrochem. 70, 508–518 (2007)
H. Zejli, J.L. Hidalgo-Hidalgo de Cisneros, I. Naranjo-Rodriguez, K.R. Temsamani, Talanta 71, 1594–1598 (2007)
E. Tomaszewska, K. Soliwoda, K. Kadziola, B. Tkacz-Szczesna, G. Celichowski, M. Cichomski, W. Szmaja, J. Grobelny, Microchem. J. 2013, 60 (2013)
P. Anekthirakun, A. Imyim, Microchem. J. 145, 470–475 (2019)
H. Tavallali, H. Malekzadeh, F. Dadvar, M. Tabandeh, M. Haghshenas, Arab. J. Chem. 10, S2090–S2097 (2017)
T. Daşbaş, Ş Sacmacı, S. Şahan, Ş Kartal, A. Ülgen, Talanta 103, 1 (2013)
F. Pena-Pereira, I. Lavilla, C. Bendicho, Anal. Chim. Acta 669, 1 (2010)
F. Pena-Pereira, I. Lavilla, C. Bendicho, Spectrochim. Acta B 64, 1–15 (2009)
M.A. Farajzadeh, S.M. Sorouraddin, M.R. Afshar Mogaddam, Microchim. Acta 181, 829–851 (2014)
H. Abdolmohammad-Zadeh, Z. Javan, Microchim. Acta 182, 1447–1456 (2015)
E. Tahmasebi, Y. Yamini, Microchim. Acta 181, 543–551 (2014)
T. Cetin, S. Tokalıoglu, A. Ulgen, S. Sahan, I. Ozenturk, C. Soykan, Talanta 105, 340–346 (2013)
Sh. Chen, Y. Sun, J. Chao, L. Cheng, Y. Chen, J. Liu, J. Environ. Sci. 41, 211–217 (2016)
S. Jafarvand, A. Bidari, P. Hemmatkhah, M.R. Milani Hosseini, Y. Assadi, Anal. Lett. 42, 2214–2231 (2009)
T. Daşbaşı, Ş Saçmacı, A. Ülgen, Ş Kartal, J. Ind. Eng. Chem. 28, 316–321 (2015)
A.N. Anthemidis, K.I.G. Ioannou, Talanta 84, 1215–1220 (2011)
A.R. Ghiasvand, F. Moradi, H. Sharghi, A.R. Hasaninejad, Anal. Sci. 21, 387–390 (2005)
Y. Su, T. Ren, Y. Lin, Ch. Zheng, Adv. Sample Prep. 3, 100033 (2022)
F. Omidi, M. Behbahani, S. Jamaleddin Shahtaheri, S. Salimi, Environ. Monit. Assess. 187, 361 (2015)
O. Heitzsch, K. Gloe, H. Stephan, E. Weber, Solvent Extr. Ion Exc. 12, 475–496 (1994)
A. Chisvert, S. Cárdenas, R. Lucena, Trends Anal. Chem. 112, 226–233 (2019)
T. Khezeli, A. Daneshfar, Trends Anal. Chem. 89, 99–118 (2017)
M. Ghorbani, M. Aghamohammadhassan, M. Chamsaz, H. Akhlaghi, T. Pedramrad, Trends Anal. Chem. 118, 793–809 (2019)
M. Sajid, M. Khaled Nazal, I. Ihsanullah, Anal. Chim. Acta 1141, 246–262 (2021)
A. Chisvert, J.L. Benedé, A. Salvador, Anal. Chim. Acta 1034, 22–38 (2018)
M.A. Farajzadeh, M. Sattari Dabbagh, J. Chromatogr. A 1627, 461389 (2020)
R. Fu, Y. Liu, Z. Lou, Z. Wang, S.A. Baig, X. Xu, J. Taiwan Inst. Chem. Eng. 62, 247–258 (2016)
D. Lv, Y. Liu, J. Zhou, K. Yang, Z. Lou, S.A. Baig, X. Xu, Appl. Surf. Sci. 428, 648–658 (2018)
K. Li, J. Cao, H. Li, J. Liu, M. Lu, D. Tang, RSC Adv. 6, 92186–92196 (2016)
F. Fang, L. Kong, J. Huang, S. Wu, K. Zhang, X. Wang, B. Sun, Z. Jin, J. Wang, X.-J. Huang, J. Hazard. Mater. 270, 1–10 (2014)
X. Dong, L.Q. Ma, Y. Li, J. Hazard. Mater. 190, 909–915 (2011)
A. Mukherjee, A. Zimmerman, W. Harris, Geoderma 163, 247–255 (2011)
L. G. Sillen, A.E. Martell, The Chemical Society, London, 1964, no. 17; 1973, no. 25.
D. D. Perrin, IUPAC Chemical Data Series no. 22, Pergamon Press, Oxford, 1979.
K. Parvizzad, S.M. Sorouraddin, M.A. Farajzadeh, J. Food. Compos. 112, 104654 (2022).
S.M. Sorouraddin, M.A. Farajzadeh, A. Hassanyani, M.R. Afshar Mogaddam, RSC Adv. 6, 108603 (2016)
Gh. Khayatian, Sh. Hassanpoor, J. Incl. Phenom. Macrocycl. Chem. 73, 151–159 (2012)
H. Tavallali, S. Yazdandoust, M. Yazdandoust, Clean: Soil, Air, Water 38, 242–247 (2010)
S. Rastegarzadeh, N. Pourreza, A. Larki, J. Ind. Eng. Chem. 24, 297–301 (2015)
Funding
The cost of the research was received by Saeed Mohammad Sorouraddin from University of Tabriz.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there is no conflict of interest.
Ethical approval
This article does not contain any studies with human or animal subjects.
Informed consent
Not applicable.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zendehdel, A.A., Sorouraddin, S.M. & Farajzadeh, M.A. In-situ formation of the adsorbent based on octadecylamine for the extraction of Ag+ ions from aqueous solutions and its determination by microinjection flame atomic absorption spectrometry. ANAL. SCI. 39, 1901–1908 (2023). https://doi.org/10.1007/s44211-023-00399-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s44211-023-00399-1